
Volume 8
Journal of Biotechnology and Biomaterials
ISSN: 2155-952X
Biomaterials 2018
March 05-06, 2018
Page 76
conference
series
.com
March 05-06, 2018 | Berlin, Germany
3
rd
Annual Conference and Expo on
Biomaterials
Kunio Ishikawa, J Biotechnol Biomater 2018, Volume 8
DOI: 10.4172/2155-952X-C1-087
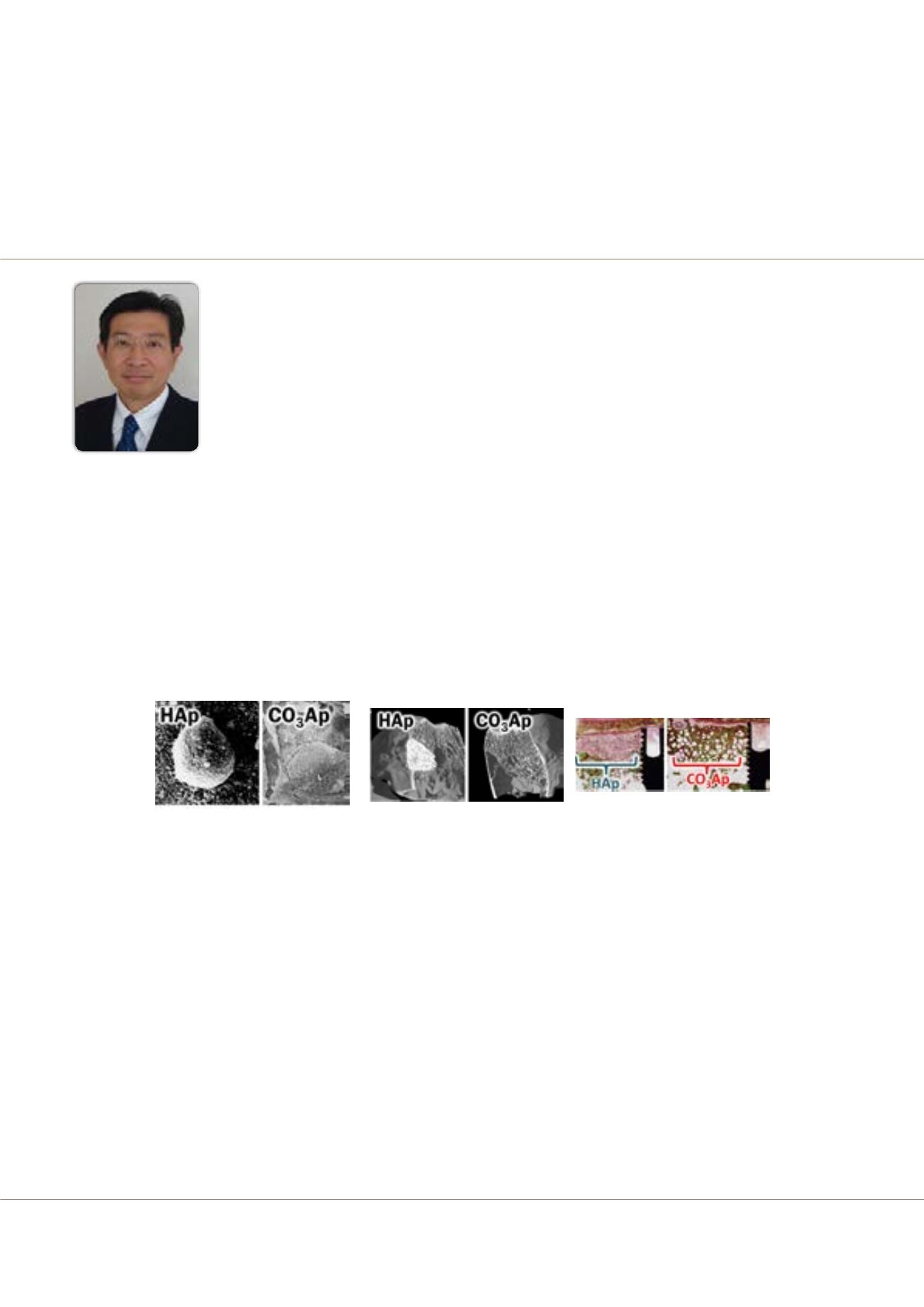
Carbonate apatite: Next generation artificial bone replacement
A
lthough bone apatite is the carbonate apatite (CO
3
Ap) that contains 6-9 wt% carbonate in apatitic structure, hydroxyapatite
(HAp) has been used as one of typical artificial bone substitute since CO
3
Ap powder start to decompose at 400°C, thus
cannot be sintered. We have found that CO
3
Ap block can be fabricated by compositional transformation though a dissolution–
precipitation reaction using precursors such as calcium carbonate and tricalcium phosphate. Although HAp is not resorbed by
osteoblasts, CO
3
Ap thus fabricated was resorbed by osteoclasts similar to bone. As a result of osteoclastic resorption, CO
3
Ap is
replaced by bone whereas HAp remained as it is at the bone defects. CO
3
Ap up-regulate differentiation of osteoblasts even when
compared to HAp. Figure 3 show the typical Villanueva Goldner staining of HAp and CO
3
Ap when used for the reconstruction of
bone defect made at the beagle dog mandible. Both HAp and CO
3
Ap demonstrated excellent tissue response. However, amount of
the bone formed at the bone defect was much larger in the case of CO
3
Ap when compared to HAp.
Figure 1: SEM images of HAp and CO
3
Ap when osteoclastic cells were incubated on their surfaces.
Figure 2: Micro-CT images of HAp and CO
3
Ap when used for the reconstruction of rabbit femur bone defect. 24 months after implantation.
Figure 3: Villanueva Goldner staining of HAp and CO
3
Ap when used for the reconstruction of bone defect at the beagle dog mandible. Three months after implantation
Recent publications
1. Ishikawa K, Kawachi G, Tsuru K and Yoshimoto A (2017) Fabrication of calcite blocks from gypsum blocks by
compositional transformation based on dissolution–precipitation reactions in sodium carbonate solution. Mater Sci &
Eng C, 51:389–393.
2. Shariff K A, Tsuru K and Ishikawa K (2017) Fabrication of dicalcium phosphate dihydrate-coated β-TCP granules and
evaluation of their osteoconductivity using experimental rats. Materials Science and Engineering C, 75:1411-1389–393.
Kunio Ishikawa
Kyushu University, Japan
















