
Page 133
Notes:
conferenceseries
.com
Volume 10, Issue 8 (Suppl)
J Proteomics Bioinform, an open access journal
ISSN: 0974-276X
Structural Biology 2017
September 18-20, 2017
9
th
International Conference on
Structural Biology
September 18-20, 2017 Zurich, Switzerland
Francesca Massi, J Proteomics Bioinform 2017, 10:8(Suppl)
DOI: 10.4172/0974-276X-C1-0100
RNA-binding domain disordermodulates the RNAdestabilizing activity in the TTPfamily of proteins
Francesca Massi
University of Massachusetts Medical School, USA
D
espite the importance of RNA-binding proteins to gene regulation, our understanding of how their structure and dynamics
contribute to their biological activity is limited. In this study, we focus on two related RNA-binding proteins—TTP and
TIS11d—that regulate the stability of mRNA transcripts encoding key cancer-related proteins, such as tumor necrosis factor-
α
and vascular endothelial growth factor. These two proteins display differential folding propensity in the absence of RNA, despite
sharing a high sequence identity. We identified three residues located at the C-terminal end of an
α
-helix that determine the
folding propensity of the RNA-binding domain in the apo state. We also showed that stabilization of the structure of the RNA-
binding domain is associated with differences in RNA-binding activity
in vitro
and increased RNA-destabilizing activity in the
cell. Phylogenetic analysis indicates that this family of proteins has only recently evolved to be able to modulate its biological
activity through its dynamic structure. To investigate how three residues determine the folding and stability of the TZF domain
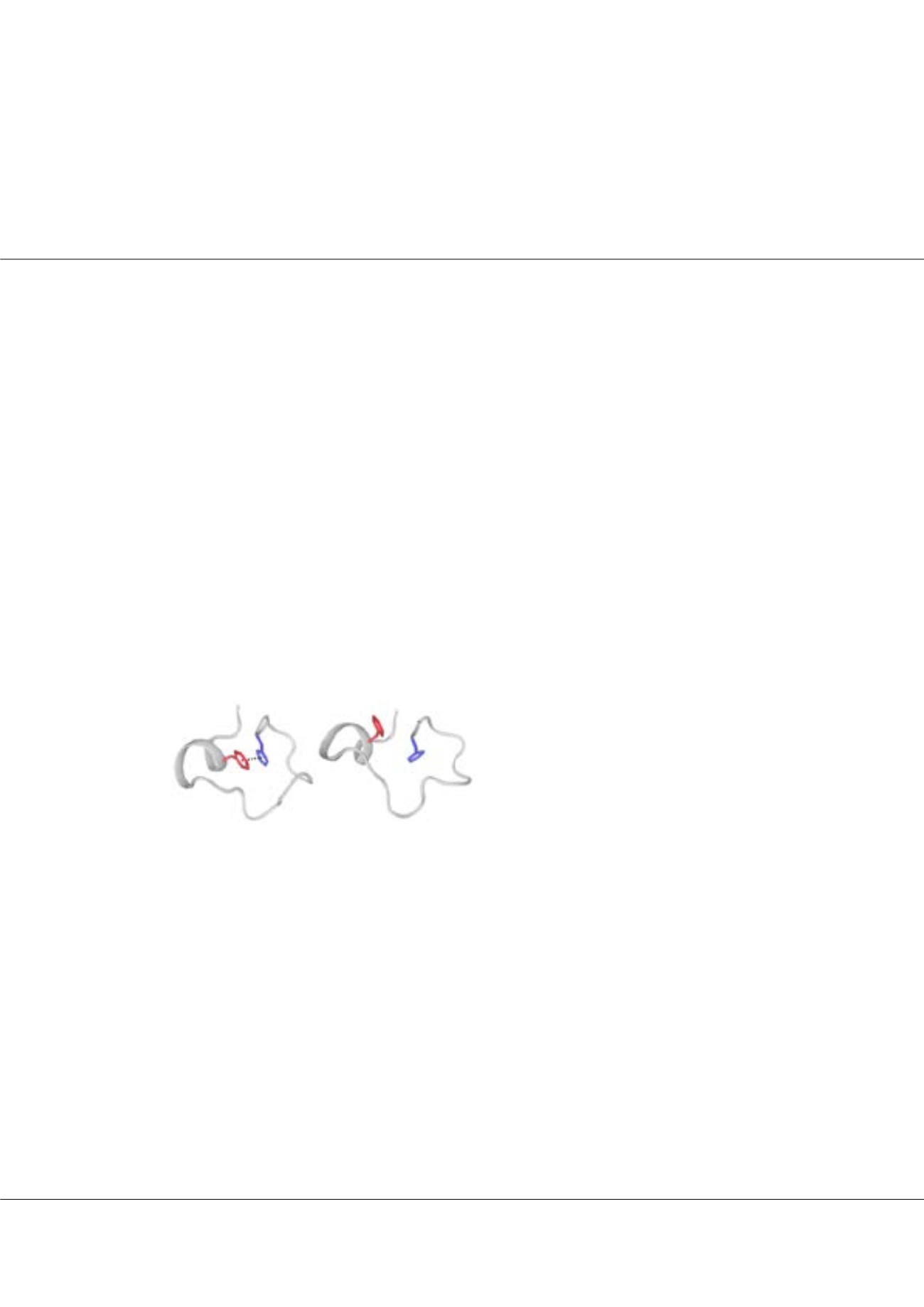
we used molecular dynamics and NMR spectroscopy. We observed that a π-π stacking between the side chains of a conserved
phenylalanine and the zinc coordinating histidine is essential to maintain the correct tetrahedral geometry between the three
cysteines, the histidine and the zinc ion. A hydrogen bond in the C-terminal zinc finger of TIS11d is important to keep the
phenylalanine in proximity of the imidazole ring of the zinc coordinating histidine in a conformation that allows for stacking
of the side chains. Lack of this hydrogen bond in TTP is responsible for the reduced zinc affinity of the C-terminal zinc finger.
Sequence alignment shows that this phenylalanine residue is highly conserved. These results suggest that most CCCH-type
zinc finger proteins employ π-π interactions to stabilize the structure of the TZF domain.
Biography
Francesca Massi is an Associate Professor in the Department of Biochemistry and Molecular Pharmacology at the University of Massachusetts Medical School.
She received her Ph.D. in Chemistry with John E. Straub from Boston University and was a postdoctoral fellow with Arthur G. Palmer at Columbia University. Her
research interests include protein function and dynamics studied with NMR spin relaxation experiments and computer simulations.
francesca.massi@umassmed.eduFigure1:
The stacking of the aromatic rings of the
conserved Phe and of the zinc coordinating His
stabilizes conformation of the His in a rotameric state
compatible for zinc-binding.


















