Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
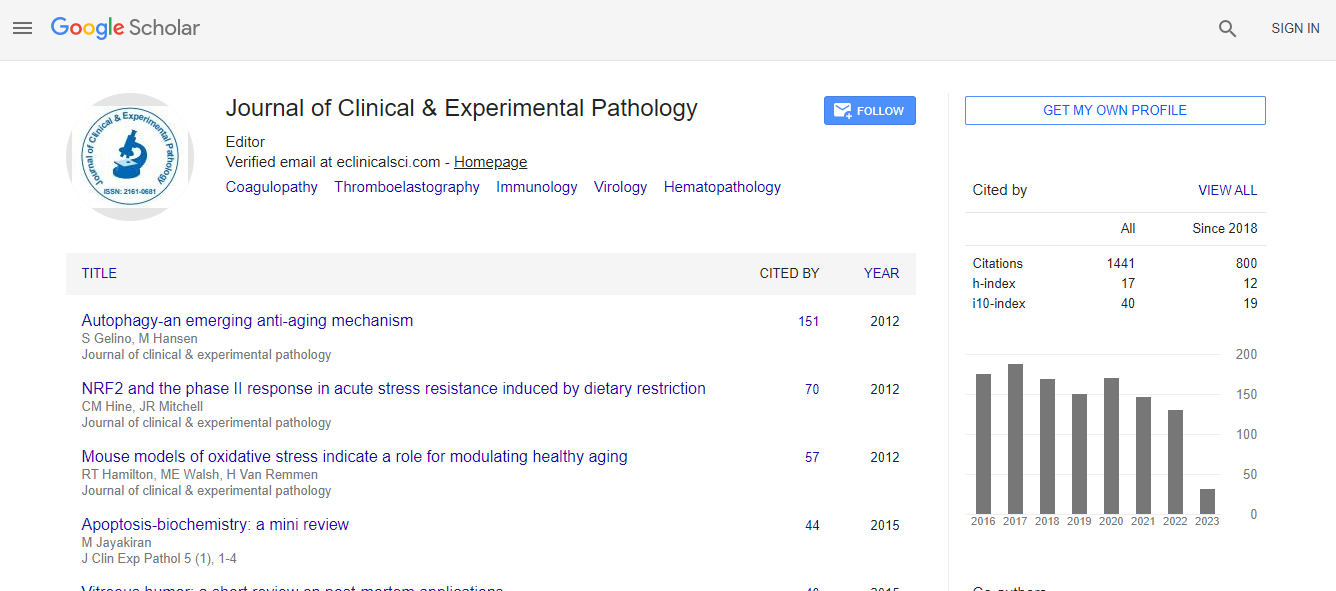
Google Scholar citation report
Citations : 2975
Journal of Clinical & Experimental Pathology received 2975 citations as per Google Scholar report
Journal of Clinical & Experimental Pathology peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Cosmos IF
- Ulrich's Periodicals Directory
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
- world cat
- journal seek genamics
- j-gate
- esji (eurasian scientific journal index)
Useful Links
Recommended Journals
Related Subjects
Share This Page
Hormone-immunotherapy in endocrine dependent metastatic breast cancer patients
Joint Event on 6th World Congress and Expo on Breast Pathology and Cancer Diagnosis & 20th International Conference on Medicinal Chemistry and Rational Drugs
Andrea Nicolini
University of Pisa, Italy
Keynote: J Clin Exp Pathol
Abstract
Hormone therapy is advised for ER+ metastatic breast cancer patients due to its efficacy concomitant with low toxicity however, in most patients the occurrence of resistance is a not well yet understood hurdle to overcome. In these patients, during clinical benefit (CB) from conventional anti-estrogens, the addition of cycles of sequential immunotherapy could prolong the benefit and delay the arising of acquired hormone resistance. In order to validate this hypothesis, in 1992 we started an open exploratory clinical trial. Forty-two of these patients in CB during first line anti-estrogen salvage therapy also received beta-interferon (INF-beta) 3,000,000 IU i.m./day 3 days/week, weeks 1-4 and successively recombinant interleukin-2 (IL-2) 3,000,000 IU s.c./day 3 days/week, weeks 5-8 until progression. The immunotherapy cycle lasted 10 weeks and the patient continued anti-estrogen alone during weeks 9-10, the 11th week being the first week of the successive cycle. At each control visit, routine laboratory examinations and serum measurement of a CEA-TPA-CA15.3 tumor marker (TM) panel were carried out, and an immunological assessment was made (total lymphocytes, CD4+, CD8+, NK cells, T-reg, IL-6, IL-10, IL-12, TNFa,TGFbeta1 and IFN-gamma.) The addition of INF-beta-IL-2 sequence significantly prolonged clinical benefit and overall survival from conventional antiestrogens. During CB as opposed to progression, a significant immune stimulation was observed. During CB also a significant CEA, TPA, CA15.3 decrease occurred 24��?72 h after interleukin-2 administration. At the progression a significant increase for CEA and for all 3 markers (standardized values) was found 24��?72 h after interleukin-2 administration. In patients who survived less than 5 years, the Treg cell increase occurred at a significantly shorter time interval than in those who survived longer than 5 years (20 vs 45.5 months, respectively; P = 0.001). To further confirm these promising results a multicenter prospective phase II trial is going to be launched by the Cancer Center Institute of Tuscany in Italy.Biography
Andrea Nicolini graduated (summa cum laude) at School of Medicine, University of Pisa in 1974. He received postgraduate diplomas at University of Pisa in Internal Medicine (1980), Pneumology (1984), and Nuclear Medicine (1986). His research interests include breast and gastrointestinal cancer and their metastases, tumour markers, post-operative follow-up, physiopathology, immunology and immunotherapy of cancer, and thyroid tumours.
E-mail: andrea.nicolini@med.unipi.it

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi