Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
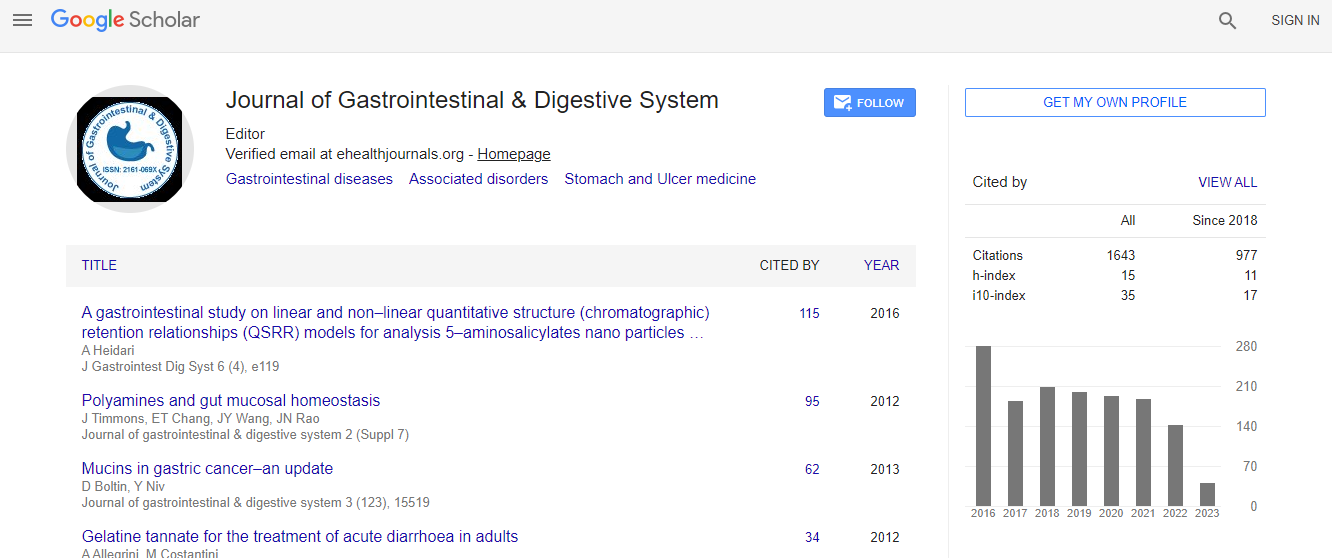
Google Scholar citation report
Citations : 1636
Journal of Gastrointestinal & Digestive System received 1636 citations as per Google Scholar report
Journal of Gastrointestinal & Digestive System peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Hepatotoxicity and related risk factors of severe hepatotoxicity among HIV-1 infected individuals initiated on highly active antiretroviral therapy in Cameroon
14th Annual Congress on Gastroenterology & Hepatology
Lem Edith Abongwa, Anthony Kebira Nyamache, Fokunang Charles, Judith Torimiro, Nshom Emmanuel, Irenee Domkam and Paul Okemo
University of Bamenda, CameroonKenyatta University, KenyaChantal Biya International Reference Centre for Research on HIV/AIDS Prevention and Management, CameroonUniversity of Yaounde , CameroonMbingo Baptist Hospital, Cameroon
Posters & Accepted Abstracts: J Gastrointest Dig Syst
Abstract
Introduction & Aim: Hepatotoxicity due to Highly Active Antiretroviral Therapy (HAART) has gained prominent attention since it can be affected by many factors. The aim of this study was to determine the prevalence of hepatotoxicity and related risk factors of severe hepatotoxicity following HAART initiation. Methods: One hundred (100) naive HIV-1 patients were recruited and followed up for 24 weeks. They were placed on either Tenofovir (TDF)+Lamivudine (3TC)+Efavirenz (EFV) or Zidovudine (AZT)+Lamivudine+Nevirapine (NVP) or Zidovudine+Lamivudine+Efavirenz regimen. Venous blood samples were collected to measure trans-aminotransferases (ALT and AST) and Alkaline Phosphatase (ALP), using colometric enzymatic reaction which were used to classified hepatotoxicity based on age and sex. Results: A total of 38 (38%) and 55 (55%) patients presented with hepatotoxicity while 15% and 28% of patients of them had severe hepatotoxicity at 4 and 24 weeks respectively. Serum levels of all enzymes increased significantly (p<0.05) with increased treatment duration. Univariate analysis revealed that the risk factor of developing severe hepatotoxicity was significantly (p<0.05) greater in patients <30 years, males, low BMI, low monthly income earners and patient on AZT+3TC+NVP regimen. While multivariate analysis showed that age <30 years, Low BMI, low monthly income and the use of AZT+3TC+NVP was an independent risk factor. Conclusion: Low BMI, <30 years, low monthly income and the use of AZT+3TC+NVP regimen were identifiable risk factors for the development of severe hepatotoxicity. As such these factors should be considered as an important strategy by clinicians in preventing the hepatotoxicity.Biography
E-mail: lemedith19@gmail.com

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi