Research Article Open Access
Variations of Plant Macronutrients and Secondary Metabolites Content in Response to Radionuclides Accumulation
| Hegazy AK1,3*, Al-Rowaily SL2, Kabiel HF3, Faisal M1 and Emam MH3 | |
| 1Department of Botany and Microbiology, College of Science, King Saud University, Riyadh, Saudi Arabia | |
| 2Department of Plant Production, college of Food and Agricultural Sciences, King Saud University, Riyadh, Saudi Arabia | |
| 3Botany Department, Faculty of Science, Cairo University, Giza, Egypt | |
| Corresponding Author : | Hegazy AK Department of Botany and Microbiology College of Science, King Saud University Riyadh, Saudi Arabia Tel: +966541792717 Fax: +96614675833 E-mail: akhegazy@yahoo.com |
| Received: December 21, 2012; Accepted: April 15, 2013; Published: April 17, 2013 | |
| Citation: Hegazy AK, Al-Rowaily SL, Kabiel HF, Faisal M, Emam MH (2013) Variations of Plant Macronutrients and Secondary Metabolites Content in Response to Radionuclides Accumulation. J Bioremed Biodeg 4:185. doi:10.4172/2155-6199.1000185 | |
| Copyright: © 2013 Hegazy AK, et al. This is an open-a ccess article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Related article at Pubmed Pubmed  Scholar Google Scholar Google |
|
Visit for more related articles at Journal of Bioremediation & Biodegradation
Abstract
Current knowledge on the basic radionuclide tolerance indicates that plants may develop mechanisms preventing the damaging effects of radionuclide stress. The radionuclides content was determined in the Mediterranean coastal black sand soil and in the edible portions of the four food crop plant species, viz., Eruca sativa, Lycopersicon esculentum, Pasidium guajava and Mangifera indica. Biochemical and nutritional constituents were assessed to elucidate the possible effect of radionuclides on the edible portions of the plants. The ability of the four study plant species to accumulate Uranium and Thorium in their edible portions from the coastal black sand soil is higher than that absorbed from the inland agricultural soils. The nutritional constituents were affected by the high concentrations of Uranium and Thorium in the plant tissues.
| Keywords |
| Nutritive value; Antioxidants; Phenolic compounds; Flavonoids; Uranium; Thorium |
| Introduction |
| Soil has always been important to humans and their health, providing a resource that can be used for shelter and food production [1]. Some elements are essential mineral nutrients for plants, with a requirement throughout life and its absence produces specific deficiency symptoms. In contrast all trace elements are toxic if their intake through ingestion or inhalation is excessive. In particular Ag, As, Be, Cd, Hg, Pb and some of the daughter products of Uranium are good examples of potentially harmful elements that have no proven essential functions in humans, and are known to have adverse physiological effects at relatively low concentrations. Despite this, known causal relationships between health problems and the elements in human foods derived from the immediate soil environment are limited [1]. |
| In some parts of the world, population growth and movement, industrial development and food security have resulted in pressure to use agricultural lands containing relatively high levels of radioactivity, for instance in the monazite areas of India and Brazil, and in parts of Iran with 226Ra anomalies where exposures up to tens of mSv, and in extreme cases 100 mSv, occur annually [2] and in many localities of black sand along the Mediterranean coast of Egypt [3-5]. |
| In Egypt, the occurrences of black sand deposits are known at the Nile mouth near Damietta and Rosetta and have been worked for their heavy mineral content such as magnetite, ilmenite, zircon and monazite [6,7]. Uptake of radionuclides from contaminated soil represents a significant pathway of human radiation exposure, either due to the direct consumption of cereals, fruits and vegetables or, indirectly, following consumption of milk and meat from animals fed on contaminated vegetable matter [8]. The understanding the radioactive mineral uptake by crop plants and the knowledge of its distribution in different organs is important in the agricultural management, to avoid its possible negative health effects. |
| The geochemistry of Uranium and Thorium is of particular interest for radiological studies. Fundamental knowledge of biogeochemical behavior of these metal and processes involved in their environmental migration are also of particular importance for power generation, water supply, agriculture and environmental protection and remediation [9]. Environmental contamination caused by radionuclides, in particular Uranium (U) and its decay products, is a serious problem worldwide [10]. Uranium is the heaviest naturally occurring element, found in small amounts in soil, rock, surface water and groundwater. Uranium can enter the human body through inhalation, ingestion, or penetration through the skin [11]. Ingestion from food or water is the main source of internally deposited Uranium in humans. It is both an alpha-emitter with a radiotoxic potential and a heavy metal with a chemotoxic potential. Chemical toxicity of U is predominantly caused by the aqueous hexavalent uranyl ion [UO2]2+ due to its high reactivity with oxygen binding centers. Hence, it resembles Ca2+ and Mg2+, but with U complexes of higher stability are formed. [UO2]2+ has a strong affinity for phosphate moieties and sugar alcohol groups of nucleotides and polynucleotides and as such, causes DNA damage [10]. Yet, a wide range of organisms in both terrestrial and aquatic environments take up U including plants, bacteria, algae, and fungi [12]. |
| The major health effect of depleted or natural Uranium exposure has been reported to be chemical kidney toxicity rather than a radiation hazard [11]. However, little is known about the effects of long-term Uranium exposure in humans. In animals, studies described effects of chronic exposure to Depleted Uranium (DU) through drinking water on the central nervous system, on reproduction or on xenobiotic detoxification [11]. It was reported that vitamin D3 metabolism, involved in mineral homeostasis and bone mineralization, was affected after a long-term exposure with environmental doses of DU or another radionuclide 137Cesium [11]. In plants, effects of U on biomass, root growth, root cell viability and interactions with mineral nutrients are reported [13]. Variable responses in plants are induced upon U exposure, which are linked with the differences in Uranium bioavailability [4,5]. Recent studies show an increase in the production of Reactive Oxygen Species (ROS) and consequent stimulation of antioxidant defenses in response to U exposure similar to that observed upon exposure of plants to redox-active metals like Cu [10]. However, the effect of U on the plant’s oxidative defense system is yet to be thoroughly investigated. |
| Current knowledge on the ubiquitous basic metal tolerance indicates that plants share several common mechanisms preventing the damaging effects of the metallic stress instead of developing proteins that could resist the heavy metal effects. These mechanisms involve reduced metal uptake, oxidative defense, metal chelation, repair of stress-damaged proteins and vacuolar compartmentalization [14]. Many nutritional factors are widely considered to be critical for human health. Among them, free radicals have been of concern as one of the factors contributing to chronic degenerative disease [15]. Usually the human body has mechanisms for eliminating the free radicals by some nutrients in the diet that have antioxidant activities. The dietary antioxidant has been defined as a substance in commonly consumed foods that significantly decreases the adverse effects of chemically reactive species on normal physiological functions in humans [15]. In recent years, an interest has been focused on antioxidant vitamins C and E, phenolics and carotenoids due to their ability to scavenge active oxygen species and free radicals [16]. Vegetables and fruits containing vitamin E, beta-carotene, and lycopene constitute natural sources of antioxidants. Antioxidants function to decrease DNA damage, reduce lipid peroxidation, and inhibit malignant transformation or cell proliferation [17]. |
| Crop plant species or fruit trees raised in radionuclide contaminated soil may take up radionuclides and accumulate them in their edible portions. These absorbed radioactive elements may affect negatively or positively the biochemical and phytochemical constituents of the plants that can be used as a defense mechanism to control the undesirable effects of toxic elements. The present study was conducted to assess the radionuclides uptake by four commonly cultivated crop species in the Mediterranean coastal black sand and inland soils. The physiological and biochemical response that may be associated with radionuclides uptake in the edible portions of the study plant species was assessed. |
| Materials and Methods |
| Study site |
| The present study was conducted in the Mediterranean coastal black sand deposits Nile delta, Egypt. The site is located around Lake El-Burullus between longitudes 31° 5′ E and latitudes 31° 35′ N. Black sand deposits are rich in heavy minerals e.g. zircon (ZrSiO4), rutile (TiO2) and ilmenite (FeTiO3). They are economically important and are characterized by high concentrations of Thorium (232Th) and Uranium (238U) in their crystalline structure [3,18]. Inland sites were selected in the main agricultural land of Nile delta as control for comparison with the coastal black sand. |
| Study species |
| Two crop plant species namely Rocket (Eruca sativa Mill.) and Tomato (Lycopersicon esculentum L.) and the two common fruit trees Guava (Psidium guajava L.) and Mango (Mangifera indica L.) were used in this study. |
| Determination of radionuclides content |
| The soil and the edible portions of the study plant species were collected from the coastal black sand and from inland cultivated site. The concentrations of Uranium and Thorium in the edible portions of the study plant species were measured by spectrophotometric technique [19], using high resolution Inductively Coupled Plasma Mass Spectrometer (ICP-MS technique) model Jeol-JMS-PLASMAX2, at the Central Laboratory for Elemental and Isotopic Analysis, Nuclear Research Center, Atomic Energy Authority, Egypt. Plant samples were oven dried at 105°C for 24 hours. Upon removal from the oven, the samples were stored in desiccators and thoroughly ground into fine powders. Exact 500 mg for each sample were dissolved in a mixture of mineral acids using a microwave digestion system and then heated to dryness. The residue was dissolved in 20ml of 2% HNO3 (40 times dilution). The measured Uranium and Thorium concentrations in soil and plant samples were obtained in ppm and converted to unite of Bq Kg-1 dry weight by multiplying the value of Uranium by 12.34 and multiplying the value of Thorium by 4.04. |
| Phytochemical analysis |
| The edible portions of the plant materials were collected in plastic bags and transferred in ice box to the laboratory within three hours after collection. The material was handily cleaned and washed with distilled water before further processing and analysis. |
| Macronutrients content |
| Moisture was determined according to Association of Official Analytical Chemist [20], 5g of air dried plant sample were accurately weighed in porcelain crucible, and then dried in an oven at 105°C until constant weight was obtained. The loss in weight was calculated and reported as percent moisture. |
| For ash determination, exact 2g of air dried plant sample were heated in a crucible at 100°C until water was expelled. The crucible was then left in a muffle furnace at 550°C to a constant weight. The weight of the residue was calculated and expressed as percent ash [20]. |
| Total protein content was calculated by multiplying the total organic nitrogen by 6.25. Total nitrogen was determined by kjeldahl method [21]. The dried sample (1g) was digested with concentrated sulfuric acid in the presence of digestion catalyst (a mixture of copper sulfate and anhydrous sodium sulfate, 1:10). The digestion was carried out using Kjeldatherm; Gerhardt, laboratory instrument. After digestion, the solution was treated with excess NaOH (50ml, 50%). The ammonia was received into (50ml, 20%) boric acid and titrated with 0.1 N sulfuric acid. Titration was carried out using Vapodest 50s; Gerhardt, laboratory instrument. Crude fats were extracted and determined using Soxtherm; Gerhardt, laboratory instrument according to Association of Official Analytical Chemist [20]. Ten grams dried plant sample was extracted using chloroform methanol (2:1 v/v). The extract was dried over anhydrous sodium sulfate, and then the solvent was removed by heating at 80°C under vacuum. The residue was cooled, weighed and expressed as percent lipid. |
| For fiber determination, a known weight of the ground sample (2g) was boiled in 1.25% H2SO4 for 30 minutes and filtered then thoroughly washed with hot distilled water. The residue was boiled with 1.25% NaOH solution for 30 minutes, and then filtered. The residue was washed with distilled water followed by ethyl alcohol and acetone, then dried at 100°C to constant weight. The ash content was determined and subtracted from the dry weight of treated material to give the fiber content [20]. |
| Regarding total carbohydrate determination, a known weight (0.5g) of the dried sample was placed in a test tube, and then sulfuric acid (10ml-1 N) was added. The tube was placed overnight in an oven at 100°C. The solution was then filtered into measuring flask and completed to the mark with distilled water. The total hydrolysable carbohydrates were determined calorimetrically with the phenol-sulfuric acid method according to Dubois M [22]. |
| The energy content was estimated by multiplying the percentages of crude protein, crude fat and digestible carbohydrates by their respective Atwater factors 4, 9 and 4, respectively [23,24]. The obtained value represents the nutritive value. |
| Macroelements |
| Analysis was made according to Wu et al. [25], by using a known weight (0.5g) of the dried sample and wet digestion was conducted using a microwave oven (Advanced Microwave Digestion System. ETHOS1). Total content of macroelements were determined in the digested solution using inductively coupled plasma emission spectrometry (ICP 6000 Series; Thermo Scientific) according to Allen et al. [26]. |
| Vitamin A content |
| According to Neeld and Pearson [27], 2g fresh plant material was mixed with 2ml 95% ethanol followed by 3ml petroleum ether and shaked for two minutes for extraction of vitamin A. After centrifugation supernatant was placed in a cuvatte, then the absorbance was determined at 450 nm against petroleum ether blank. Petroleum ether was evaporated to dryness in a water bath then the residue was taken up in 0.1ml chloroform and 0.1ml acetic anhydride was added. Vitamin A was read at 620 nm against a blank consisting 0.1ml chloroform and 1ml trifluoroacetic acid reagent. Vitamin A in the sample was calculated as following: μg vitamin A/100g fresh weight plant sample=OD620- (OD450×0.3)×337, Whereas OD=Optical Density. |
| Vitamin C content |
| Vitamin C was determined according to Iqbal [28]. To accurately weighted 1g of each sample, 10ml of 0.05M oxalic acid solution was added then the solution was placed under shade for 24h to extract vitamin C. After filtration, the filtrate was transferred to 25ml volumetric flask then 2.5ml of 0.05 M oxalic acid solution was added. Separately meta phosphoric acid with acetic acid, 5% sulphuric acid and ammonium molybdate solution were added to the sample then the volume was made up 25ml with distilled water. The sample was then analyzed for vitamin C at 760nm compared with the standard L-ascorbic acid. |
| Total phenolics |
| Total phenolic content was determined by the Folin-Ciocalteu method described by Meda A [29]. Two grams of fresh plant material was mixed with 2.5ml of 0.2 N Folin-Ciocalteu reagent for 5 min, then 2ml of 75g/l sodium carbonate was then added. After incubation at room temperature for 2h, the absorbance of the reaction mixture was measured at 760 nm against methanol blank. Gallic acid was used as standard to produce the calibration curve. |
| Total flavonoids |
| Total flavonoids in 1g sample were extracted with 10ml 80% ethanol for 20 min with continuous shaking [30]. After centrifugation for 10 min, the supernatant was collected. Exact 0.5ml of extract was mixed with 1.5ml of 95% ethanol, followed by 0.1ml of 10% aluminum chloride, 0.1ml of 1M potassium acetate and 2.8ml distilled water. After incubation at room temperature for 30 min, the absorbance of the reaction mixture was measured at 415 nm. The flavonoid content was calculated using a standard calibration of rutin solution. |
| Antioxidant activity |
| The antioxidant activity was determined by the free radical scavenging effect of the plant extract assessed by the decolouration of the methanolic solution DPPH (1,1 diphenyl-2-picryl hydrazyl) radical according to Lee et al. [31]. Dried plant sample was dissolved in methanol then 5ml of DPPH solution added. Different concentrations of the plant extract were prepared. Methanolic DPPH was used as control. After 30 min, the absorbance was measured at 517 nm. Rutin was used as reference free radical scavenger at the same dose. The antioxidant activity was expressed as: DPPH decolouration %=100×(A1-A2 /A2), whereas A1 is the absorbance of the control and A2 the absorbance in the presence of the tested extracts. |
| Total chlorophyll |
| The determination was carried out according to the method reported by Holden [32]. The fresh sample (0.5g) was grounded in a mortar with acetone in the presence of little amount of calcium carbonate then filtered. The residue was washed with acetone several times until the washing liquid was colourless. The absorbance was measured at 663 and 645 nm against 80% aqueous acetone as blank. The concentration was calculated from the following equation. |
| Chlorophyll a (mg/g)=((12.3×A663-0.86×A645)/ a×100×w) v |
| Chlorophyll b (mg/g)=((19.3×A645-3.6×A663)/ a×100×w) v |
| Whereas A663 and A645 are the absorbance at 663 and 645 nm; a is the length of light path in cell; w is the fresh weight in gram and v is the volume inml. |
| Total carotenoids |
| Carotenoids were extracted from the plant samples and determined spectrophotometrically following [33]. One gram of each sample was saponified overnight with 0.5 N KOH at room temperature. The saponified fraction was then transferred to separating funnel and extracted several times with diethyl ether. The extract was washed with distilled water and 0.5 N KOH then dried over anhydrous sodium sulfate. A known volume of ether extract was evaporated at room temperature then 5ml petroleum ether added. Carotenes were measured at 440 nm against petroleum ether blank. |
| β-Carotene and lycopene |
| The β -carotene and Lycopene were extracted using the method described by Tee and Lim [34]. From the extract, 2.5ml was mixed with 40ml methanol containing 1g potassium hydroxide. From the concentrated extract, 2ml were evaporated under running nitrogen, re-dissolved in 2ml acetone, passed through a 0.45 μm millipore membrane and 30 μl aliquots were injected into the HPLC system (Hewlett Packard, HP 1090 liquid chromatography). |
| Results |
| Radionuclides content |
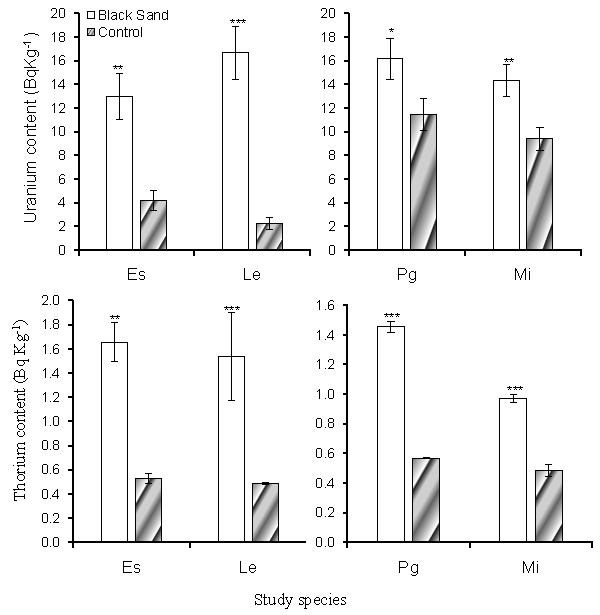
| Uranium and Thorium concentrations of the study soil are summarized in table 1. The concentrations of Uranium and Thorium were significantly higher in the coastal black sand soil than in the inland soils. The Uranium concentration in the black sand soil where E. sativa and L. esculentum cultivated were 76.919 ± 3.105 and 95.429 ± 3.105 BqKg-1 dry weight respectively, which was significantly higher than that in the inland soil where values attained 45.247 ± 1.885 and 46.892 ± 2.468 BqKg-1 dry weight respectively. The Uranium concentrations in the coastal black sand soil supporting P. guajava was 86.791 ± 1.885 BqKg-1 dry weight, while in the inland soil, was 60.877 ± 1.425 BqKg-1 dry weight. The same trend of results was observed with the concentration of Thorium. Concentrations of Uranium and Thorium determined in the edible portions of the study species are shown in figure 1. Uranium and Thorium concentrations in the plants cultivated in the coastal black sand soil generally attained values higher than that in the plants cultivated in the inland soil. In the leaves of E. sativa Uranium and Thorium concentrations were 4.196 ± 0.494 and 0.525 ± 0.008 BqKg-1 dry weight respectively in the plants cultivated in the inland soil but these concentrations significantly increased to 12.957 ± 2.221 and 1.657 ± 0.364 BqKg-1 dry weight respectively in the plants cultivated in the coastal black sand soil. Similarly, Uranium and Thorium concentrations in fruits of L. esculentum, P. guajava and M. indica from coastal black sand soil were significantly higher than their concentrations in the plants cultivated in the inland sites. |
| Proximate composition |
| The macronutrients content of the study plant species are shown in table 2. Crude fibers attained value 0.737 ± 0.025gm /100gm dry weight in leaves of E. sativa cultivated in the inland site, while in the coastal black sand site, the value increased to 0.844 ± 0.018gm/100gm dry weight. This is associated with the uptake of high level of Uranium and Thorium and their accumulation in edible portions of the plants. This was also observed in the nutritive values which increased from 27.279 ± 0.902 KCal in plants in the inland site to 32.461 ± 0.68 KCal in plants in the coastal black sand. In L. esculentum, the chemical composition didn’t showed significant difference between the values in plants from the inland soil and plants from coastal black sand, except for crude protein which significantly decreased. The total carbohydrates and nutritive value significantly increased with the increase of Uranium and Thorium content in the edible portions of the plants cultivated in the coastal black sand site. For P. guajava cultivated in the inland soil, the moisture content reached 88.469 ± 0.163% and significantly decreased to 83.582 ± 1.236% with the increased Uranium and Thorium content in the coastal black sand site. In contrary, the remaining chemical composition showed significant increase with increased Uranium and Thorium in the plants (Table 2). For M. indica, the measured proximate composition increased with uptake of more Uranium and Thorium in the coastal black sand soil. The moisture content reached 86.074 ± 0.071% in plants cultivated in the inland soil, while decreased to 82.334 ± 0.202% in the coastal black sand site. |
| Macroelements content |
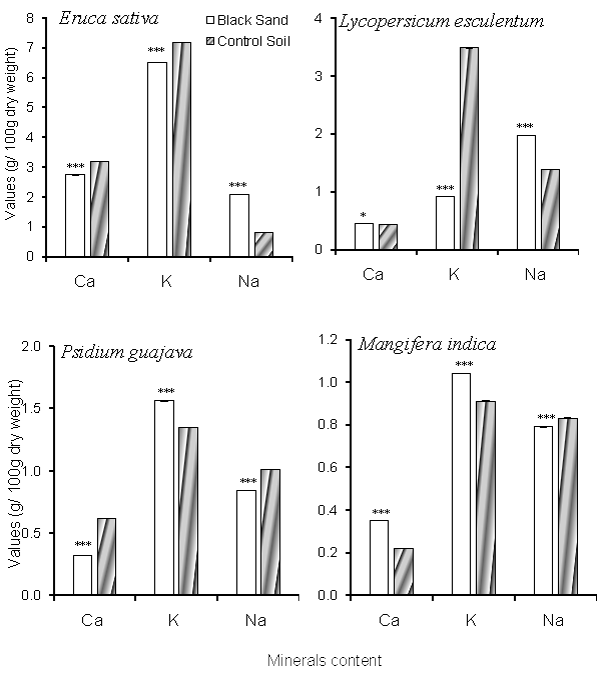
| The edible portions content of Ca, K and Na of the study plant species are shown in figure 2. As indicated from the results, K content in edible portions of study crop plant species (E. sativa and L. esculentum) decreased with Uranium and Thorium uptake while in the edible portions of the study fruit trees, values increased with accumulation of more Uranium and Thorium. In contrast to K content, Na content increased in the crop plant species and decreased in the fruit trees with uptake of more Uranium and Thorium in plant tissues. Calcium content didn’t show constant trend with the increase of Uranium and Thorium content of the plants. |
| Vitamins composition |
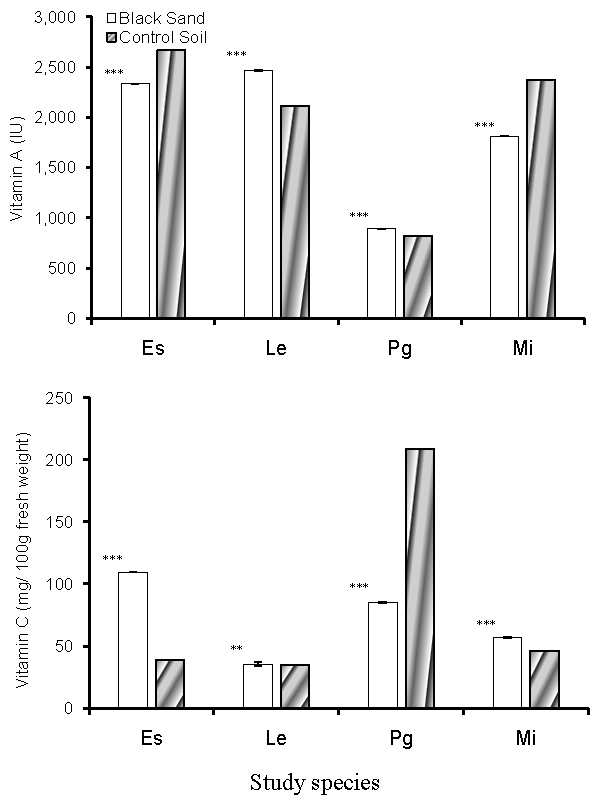
| The content of vitamin A and vitamin C in the edible portions of the study plant species are shown in figure 3. Vitamin A content has significantly increased with the increased amounts of Uranium and Thorium in the tissues of L. esculentum and P. guajava cultivated in the inland soils, with values reached 2113.38 ± 2.09 and 822.70 ± 1.00 IU in both species respectively. For plants cultivated in coastal black sand soil, vitamin A content reached 2468.24 ± 1.00 and 892.29 ± 1.00 IU in both species respectively. In E. sativa and M. indica, vitamin A content decreased in plants from black sand soil. Concentration of vitamin C (Figure 4) increased in the plants cultivated in the coastal black sand soil, which is associated with increased accumulation of Uranium and Thorium in the plant tissues. |
| Secondary compounds content |
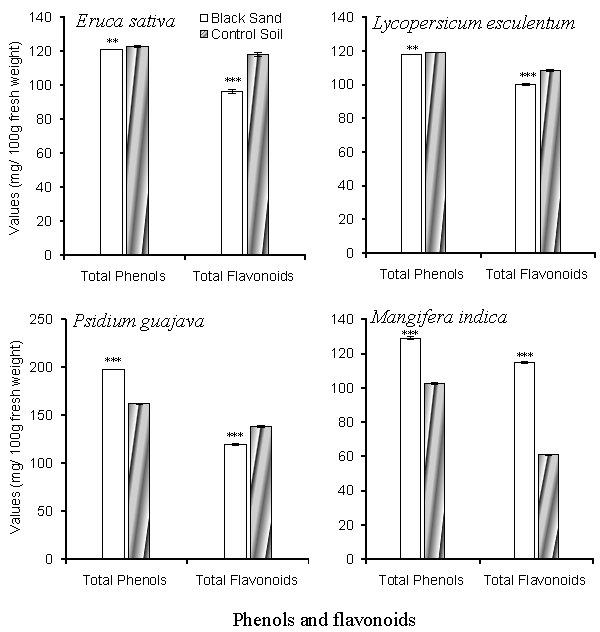
| Total phenolic compounds and total flavonoid content are shown in figure 4. Total phenolic compounds varied between 102.521 ± 0.507 mg/100gm fresh weight in fruits of M. indica to 161.98 ± 0.186 mg/100gm fresh weight of P. guajava fruits cultivated in the inland soil. In the coastal black sand soil, the total phenolic compounds increased to 129.123 ± 0.85 mg /100gm fresh weight in M. indica and 197.996 ± 0.22 mg/100gm fresh weight in P. guajava. As for E. sativa and L. esculentum, the total phenolic compounds showed no significant difference between coastal and inland sites. Total flavonoids content attained lower values in plants cultivated in the coastal black sand site than plants in the inland sites. |
| Antioxidant activity |
| As shown in figure 5, the antioxidant activity measured as DPPH free radical scavenging activity, decreased with the increase of Uranium and Thorium content of plant tissues. For P. guajava, the antioxidant activity attained value 6.722 ± 0.003% in plants cultivated in the inland site and value 7.062 ± 0.003% in plants of the coastal black sand site. |
| Total chlorophylls and carotenes content |
| Total chlorophylls in leaves of E. sativa (Table 3) increased from 727.00 ± 2.00 mg/100gm fresh weight in plants from the inland site to 952.00 ± 2.00 mg/100gm fresh weight in plants from coastal black sand site. Total carotenoids content showed significant increase with the increase of Uranium and Thorium content of the plant edible portions. Only for M. indica, the total carotenoids decreased with Uranium and Thorium uptake from 9.481 ± 0.003 to 7.234 ± 0.001 mg/100gm fresh weight in the plants of the coastal black sand site. β-carotene and lycopene in L. esculentum exhibited values 3.135 ± 0.01 and 9.520 ± 0.03 mg/100gm fresh weight respectively in plants cultivated in the inland sites. For plants cultivated in the coastal black sand soil, the values increased to 4.128 ± 0.01 and 10.773 ± 0.003 mg/100gm fresh weight, respectively. |
| Relationship between uranium content and biochemical constituents |
| The relationship between Uranium content of the plant and its biochemical constituents showed positive linear relationship with total carbohydrates, nutritive value, vitamin C, phenolic compounds, flavonoids, antioxidant activity and beta-carotene contents (Figure 6). Only negative relationship attained with the total protein content. |
| Discussion |
| The concentrations of Uranium and Thorium accumulated in the edible portions of the study plant species cultivated in the coastal black sand soil were significantly higher than that in the inland soil. Similarly [3] detected higher level of Uranium and Thorium in the tissues of wild plants raised in the black sand soil than those in the inland soil. Physiological responses of higher plants to environment have been studied extensively over the world. Abiotic environmental stresses are linked with all the living process of higher plants and greatly influence plant growth and productivity [4,35,36]. Although some metals are necessary for biological processes, they are toxic at high concentrations. This is due to their oxidative capacity to form free radicals and their ability to replace essential metals in enzymes, interrupting their normal activity. Other metals are not essential and accumulate in different organisms and become toxic even at low concentrations [37]. |
| The macronutrients in the study species revealed that high Uranium and Thorium content in the plant tissues was associated with significant increase in total carbohydrates, ash, crude fibers, crude fats and nutritive values of the four study species. These results are in agreement with results obtained by Gang et al. [35], who found increase in the total soluble sugars with the increase of Uranium and Thorium concentration in the leaves of Capparis spinosa. Total protein content was found to decline due to uptake of more U and Th in plants from coastal black sand soil. This finding is supported by Srivastava et al. [10] who reported decline in the level of total soluble protein in Hydrilla verticillata after exposure to Uranium in comparison to the control treatment. This is in contrary to the results of Bajpai et al. [38] that revealed a positive correlation between protein content and the heavy metal content of the plant. Inhibition of protein accumulation induced by higher concentrations of heavy metals may attribute to the toxicity of these metals on the enzymatic reactions responsible for protein biosynthesis [39,40]. Increase of Uranium and Thorium content in the edible portions of the study species caused general decline in the content of Ca, K and Na. This observation can be explained by the presence of a competition for uptake between radionuclides and other captions present in the soil such as Zn, Mg, K, Na and Ca [41]. |
| Vitamin C content was negatively affected with high U and Th content in the plant tissues. As expected from its antioxidant based function in plant metabolism, the level of vitamin C is responsive to a wide variety of environmental stress factors including radioactive elements [42]. Similarly, the total flavonoids content was found to decrease in the plants from coastal black sand soil with the absorption of high amounts of Uranium and Thorium. Similarly, study of Morsy and Afifi [43] on some wild plant species grown on natural radioactive soils revealed that Uranium has a negative effect on the flavonoids content. In contrast to minerals, vitamin C and flavonoid content, the phenolic compounds significantly increased with increased amounts of Uranium and Thorium in plants. That was observed from the relationship between phenolic compounds content in the four study plant species and concentrations of Uranium in the edible portions of these plants. This finding is in accordance with that obtained by Srivastava et al. [10], where total phenolic compounds showed significant increase in response to Uranium exposure. |
| Phenolic compounds, among the most widely occurring groups of phytochemicals, are of considerable physiological and morphological importance in plants as they play an important role in growth, reproduction, and protection against pathogens and predators [42,44]. Phenolic compounds exhibit a wide range of physiological properties and have been associated with the health benefits [44]. The antioxidant activity of phenolic compounds is due to their ability to scavenge free radicals, donate hydrogen atoms or electron, or chelate metal cations. The role of antioxidants in preventing oxygen radicaland hydrogen peroxide-induced cytotoxicity and tissue damage is becoming increasingly recognized. The importance of the antioxidant constituents of plant materials in the maintenance of health and protection against diseases [45]. |
| The total chlorophyll and carotenoids content are positively correlated with Uranium and Thorium content of the study plant species. Also, β-carotene and Lycopene content were positively influenced by elevated amounts of Uranium and Thorium in plant tissues. Our results are in agreement with the results of Bajpai et al. [38] who found that carotenoids have significant positive correlation with metal accumulation. Alternatively, the results obtained by Huang GY [46] proved remarkable decrease in chlorophyll a, chlorophyll b, chlorophyll (a+b) and carotenoids content in the leaves of mangrove plants exposed to multiple heavy metals. Carotenoids are essential components for the photosynthetic apparatus in plants, which participate in the adaptation of plastids to changing environmental conditions and prevent photo-oxidative damage of the photosynthetic apparatus [46]. Previous studies have reported an increase [47] or decrease [48] in the carotenoids content of plants in response to metal stress. They are present as micro-components in fruits and vegetables and represent important dietary sources of vitamin A [49]. Carotenoids accumulation is often regarded as one of the mechanisms to counteract stresses [50]. |
| Several stress factors including exposure to toxic metals can lead to an oxidative stress state that inhibits a range of enzyme activities in plants, in particular those of the Calvin cycle [51]. Pollutant metals disturb the oxidative balance and thus an important palliative measure is the induction of antioxidants [52]. The induction of antioxidant compounds and enzymes is of great importance to increase tolerance of plants against different kinds of stress [53]. Carotenoids accumulation is often regarded as one of the mechanisms to counteract stress in organisms [50]. The role of β-carotene as 1O2 quencher was established in plants and algae [54]. A significant rise in carotenoids content as noticed under toxic metal stress might offer protection to the chlorophyll and photosynthetic membrane from photooxidative damage [55]. |
| The antioxidant activity of fruits and vegetables is an important aspect of their nutritional value since antioxidant molecules have a critical role in the detoxification of free radicals in both plants and humans [56]. It’s well known that amount of each antioxidant in tomato fruits is strongly influenced by varietal differences (genetic influence), in addition to agronomical, geographical and environmental parameters [57]. Lycopene, the red isoprenoid pigment of tomato, constitutes about 80-90% of total carotenoids content [57]. Recent epidemiological studies have shown that supplementation of diets rich in lycopene is associated with reduced risk of many chronic diseases, such as cancer and heart diseases [16]. The antioxidant activity of lycopene has been extensively evaluated based on its ability to scavenge free radicals in cell culture and in animal models. Increased DNA damage and elevated levels of lipid peroxidation in γ-irradiated hepatocytes are accompanied by a decrease in the free radical scavengers. Pretreatment of lycopene resulted in increased levels of these antioxidants. These may be attributed to lycopene's ability to act as an antioxidant and singlet oxygen quencher [58]. |
| Various interactions are known to occur when plants are exposed to unfavorable concentrations of more than one element [59]. As indicated from the present study the tissue concentrations of highvalue metabolites, such as vitamin C, phenolic compounds, flavonoids, Lycopene and β-carotene and other antioxidant compounds are variable under environmental stresses that may elicit the biosynthesis/ catabolism of stress responsive molecules [56]. |
| In conclusions, the study of radionuclides content in crop plants cultivated in the coastal black sand soil revealed their ability to accumulate high concentrations of Uranium and Thorium in the edible portions. The biochemical responses indicated that some nutritional contents of the plants including protein, moisture, mineral, vitamin C and total flavonoids contents showed negative correlation with Uranium and Thorium content. The carbohydrates, crude fats, crude fibers, nutritive value, total phenolic compounds, total chlorophylls, total carotenoids, β-carotene and lycopene were significantly enhanced by high Uranium and Thorium concentration in the plant tissues. The results indicated that each of biochemical constituents of the test plants were affected by elevated level of radionuclides in plants. The variations between enhancement and suppression of the phenolic compounds content of the study plant species can be considered as antioxidant defense mechanism of cells in order to control the redox state of the cell containing high radionuclides concentrations. |
| Acknowledgements |
| We thank the support of King Saud University, Deanship of Scientific Research, College of Science Research center. |
References
|
Tables and Figures at a glance
| Table 1 | Table 2 | Table 3 |
Figures at a glance
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 |
 |
 |
 |
| Figure 4 | Figure 5 | Figure 6 |
Relevant Topics
- Anaerobic Biodegradation
- Biodegradable Balloons
- Biodegradable Confetti
- Biodegradable Diapers
- Biodegradable Plastics
- Biodegradable Sunscreen
- Biodegradation
- Bioremediation Bacteria
- Bioremediation Oil Spills
- Bioremediation Plants
- Bioremediation Products
- Ex Situ Bioremediation
- Heavy Metal Bioremediation
- In Situ Bioremediation
- Mycoremediation
- Non Biodegradable
- Phytoremediation
- Sewage Water Treatment
- Soil Bioremediation
- Types of Upwelling
- Waste Degredation
- Xenobiotics
Recommended Journals
Article Tools
Article Usage
- Total views: 14709
- [From(publication date):
March-2013 - Apr 03, 2025] - Breakdown by view type
- HTML page views : 10028
- PDF downloads : 4681
