| Research Article |
Open Access |
|
| Sharma JN* and Al-Banoon A |
| Department of Applied Therapeutics, Faculty of Pharmacy, Health Sciences Centre, Kuwait University, PO Box 24923, Safat 13110, Kuwait |
| *Corresponding authors: |
Sharma JN
Department of Applied Therapeutics
Faculty of Pharmacy, Health Sciences Centre
Kuwait University, PO Box 24923
Safat 13110, Kuwait
E-mail: j.n.sharma@hsc.edu.kw |
|
| |
| Received July 15, 2012; Published September 23, 2012 |
| |
| Citation: Sharma JN, Al-Banoon A (2012) The Role of Inflammatory Mediator Bradykinin in Cardiovascular and Renal Diseases. 1: 142. doi:10.4172/scientificreports.142 |
| |
| Copyright: © 2012 Sharma JN, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. |
| |
| Summary |
| |
| Bradykinin and other related kinins appear to be important regulators of cardiovascular function. They are being increasingly noted as likely participants in actions of drugs that affect heart, kidney, and circulation. This phylogenetically ancient system of substrates, proteases, peptides, peptidases, and inhibitors has some responsibility in the regulation of local and perhaps systemic hemodynamics, vascular permeability, inflammatory response, activation of neuronal pathways, and movement of electrolytes, water, and metabolic substrates across epithelia and into other tissues. It has been shown that bradykinin acts through two receptors, B1 and B2 that differ in the mechanism by which they are regulated. The development and use of both B1 and B2 receptor antagonists as potential drug targets has been implicated in several pathophysiological conditions like hypertension, diabetes and other cardiovascular and renal disorders. Although bradykinin has multiple beneficial actions, some undesirable effects have been reported, such as, oedema, broncho constriction and angioedema; which can be converted by bradykinin antagonists. Finally, a number of other applications are still under clinical investigations for the applicability of bradykinin receptor antagonists in heart failure, sepsis and asthma. |
| |
| Introduction |
| |
| Bradykinin (BK) is a member of Kallikrein-Kinin System (KKS), a complex of two substrates (kininogens) activated by two enzymes (kallikreins) to produce four inflammatory mediators (kinins) that bind to two BK receptors, B1 and B2 [1]. |
| |
| B1 receptors are expressed in only few tissues under normal conditions and only in very small numbers. Several diseases are occurred with rapid induction of B1 receptors in specific tissues [2]. |
| |
| All tissues express B2 receptors which mediate activities of BK. BKinduced activation of B2 receptors causes relaxation of venular smooth muscle, hypotension, increased vascular permeability, contraction of smooth muscle of gut and airway leading to increased airway resistance, stimulation of sensory neurons, alteration of ion secretion of epithelial cells, production of Nitric Oxide (NO), and release of cytokines from leukocytes and eicosanoids from various cell types [3]. Because of this broad spectrum of activity, BK has been implicated in many pathophysiological conditions including pain, sepsis, asthma, symptoms associated with rhinoviral infection, rheumatoid arthritis, and other inflammatory diseases. Several BK receptor antagonists are being developed with major interest in treating these pathological conditions to block activation of BK receptors. In fact, using BK antagonist clinically to treat any of these disorders would represent novel therapy [4,5]. |
| |
| Furthermore, all members of KKS are located in cardiac muscle, and its deficiency may lead to cardiac dysfunction. Recently, many observations that are obtained from experimental animals of diabetes, hypertension, cardiac failure, ischemia, Myocardial Infraction (MI) and Left Ventricular Hypertrophy (LVH) have found that reducing activity of BK may be the reason for induction of cardiovascular related diseases in those cases [6]. So, activation of BK and/or its receptors by BK agonist can be an excellent candidate in treating hypertension, cardiovascular and renal diseases. In addition, BK agonists may also be available in the future as therapeutic agents for cardiovascular and renal disorders [7]. |
| |
| Kallikrein-Kinin System (KKS) |
| |
| Kinin is a general term for a group of polypeptides, found in blood, which ultimately produce BK [8]. By virtue of their ability to activate endothelial cells, leading to vasodilatation, increased vascular permeability, tissue-type plasminogen (t-PA) release, production of NO and mobilization of arachidonic acid, they participate in physiological (regulation of blood pressure, renal and cardiac functions) and pathological processes like inflammation [9]. |
| |
| Members |
| |
| KKS consists of large proteins, small polypeptides and group of enzymes that activate and deactivate the compounds. Proteins are High-Molecular Weight Kininogen (HMWK) and Low-Molecular Weight Kininogen (LMWK) [10]. |
| |
| Kininogens are precursors of kinins and substrates of kallikreins. They are present in plasma; lymph, and interstitial fluid, two kininogens are known to be found in plasma: a LMWK and HMWK. Both are acidic glycoproteins consisting of single polypeptide chain. About 15-20% of the total plasma kininogen is in HMW form. It is thought that LMWK crosses capillary walls and serves as substrate for tissue kallikrein, while HMWK is confined to bloodstream and acts as substrate for plasma kallikrein [1]. |
| |
| The polypeptides of KKS are BK and kallidin (KD). BK is produced when kallikrein releases it from HMWK, while, KD is released from LMWK by tissue kallikrein [11]. |
| |
| The enzymes are kallikreins, Carboxypeptidases, Angiotensin Converting Enzyme (ACE) and Neutral Endopeptidase (NEP) [7]. |
| |
| Kallikreins are glycoprotein enzymes present in plasma and in several tissues, including kidneys, pancreas, intestine, sweat glands, and salivary glands. They are serine proteases with active sites and catalytic properties similar to those of enzymes, such as, trypsin, plasmin, and elastase. |
| |
| Plasma kallikrein circulates in blood as precursor, prekallikrein, which is produced by the liver. Plasma kallikrein can be activated by trypsin, Hageman factor and possibly kallikrein itself. Some glandular kallikreins exist as prekallikrein; others are present in active forms. In general, biochemical properties of glandular kallikreins are quite different from those of plasma kallikreins [1]. |
| |
| Carboxypeptidases are present in two forms, N circulates and M is membrane-bound. They remove arginine residues at carboxylterminus (C-terminal) of BK and KD [11]. |
| |
| ACE also termed kininase II inactivates a number of peptide mediators, including BK [12]. |
| |
| NEP, also, deactivates kinins and other mediators [10]. |
| |
| Formation of kinins in plasma and tissues |
| |
| The pathway for formation and metabolism of kinins is shown in (Figure 1). Three kinins have been identified in mammals: BK, lysylBK (also known as KD), and methionyllysylBK. Their structures are shown below: |
| |
|
|
Figure 1: The Kallikrein-kinn system. Kininase II is identical to converting enzyme peptidyl dipeptidase. |
|
| |
| Arg-Pro-Pro-Gly-Phe-Ser-Pro-Phe-Arg |
| |
| 1 2 3 4 5 6 7 8 9 |
| |
| Bradykinin |
| |
| Lys-Arg-Pro-Pro-Gly-Phe-Ser-Pro-Phe-Arg |
| |
| LysylBK (KD, Lys-bradykinin) |
| |
| Met-Lys-Arg-Pro-Pro-Gly-Phe-Ser-Pro-Phe-Arg |
| |
| MethionyllysylBK (Met-Lys-bradykinin) |
| |
| Note that each kinin contains BK in its structure. Each kinin is formed from kininogen by the action of different enzyme. BK is released by plasma kallikrein, KD by glandular kallikrein and methionyllysylBK by pepsin and pepsin-like enzymes. The preferred substrate for plasma kallikrein is HMWK; whereas for tissue kallikrein is LMWK. Some KD is converted to BK by aminopeptidase. The three kinins have been found in plasma, but BK is the predominate one. All three kinins are also present in urine. KD is the major urinary kinin and is probably formed by the action of renal kallikrein. BK is generated from KD by renal aminopeptidase. MethionyllysylBK occurs in acidified urine: acid activates uropepsinogen, which then catalyzes release of methionyllysylBK from urinary kininogens [1]. |
| |
| Metabolism of kinins |
| |
| Kinins are metabolized rapidly (half life <15 seconds) by nonspecific exo- or endopeptidases, commonly referred to as kininases. Two plasma kininases have been well characterized. Kininase I, apparently synthesized in liver, is carboxypeptidase that released C- terminal arginine residue. Kininase Ðß is present in plasma and vascular endothelial cells throughout the body. It is identical to ACE; peptidyl dipeptidase. Kininase Ðß inactivates kinins by cleaving C- terminal dipeptide phenyl- alanyl-arginine. Like angiotensin I, BK is almost completely hydrolyzed during single passage through pulmonary vascular bed [11]. |
| |
| Bradykinin (BK) |
| |
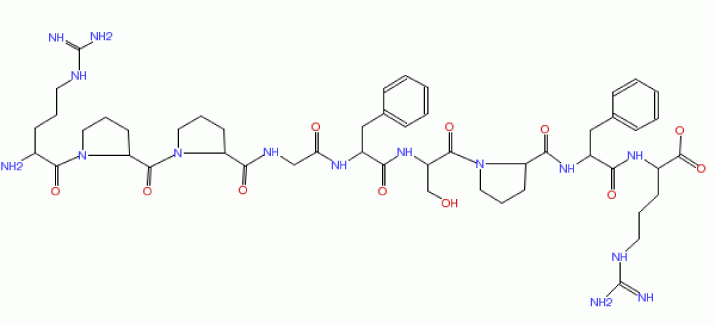
| BK is a naturally occurring neuropeptide (plasma protein). It is a pharmacologically active kinin, which is considered as either cardioprotective or proinflammatory agent [7]. BK is very similar to KD, which has same sequence but with additional N terminal lysine (KD possessing one additional amino acid residue) (Figure 2) [12,13]. |
| |
|
|
Figure 2: Struture of bradykinin and some bradykinin antagonists. The sites proteolytic cleavage for formation of kallidin and bradykinin by kallikrein from high-molecular-weight kininogen are shown in the upper half of the figure inactivation are shown in the lower half. The B2-receptor antagonist icatibant (Hoe 140) has a pA2 of 9 and the competitive B1-receptor antagonist des-Arg Hoe 140 pA2 of 8. The Hoe compounds contain unnatural amino acids: Thi, D-Tic and Oic, which are analogues of phenylalanine and proline. |
|
| |
| In humans and most mammals, term “kinin” refers to the nonapeptide BK, the decapeptide KD, and their C-terminal des-Arg metabolites [12]. |
| |
| History |
| |
| |
| BK was discovered by three Brazilian physiologists in 1948 as powerful hypotensive agent in animal preparations. BK was detected in blood plasma of animals after addition of venom of bothrops jararaca (Brazilian lanchehead snake), which was brought by Rosenfeld from Butantan Institute. This discovery was part of a continuing study on circulatory shock and proteolytic enzymes related to toxicology of snake bites, started by Rocha e Silva as early as 1939 [14]. |
| |
| Structure |
| |
| The amino acid sequence of BK is: Arg-Pro-Pro-Gly-Phe-Ser-Pro- Phe-Arg. Therefore, its empirical formula is: C50 H73 N15 O11 [6]. |
| |
 |
| |
| Synthesis |
| |
| An outline of formation of BK from HMWK in plasma by kallikrein is given in figure 3. Prekallikrein is present in plasma as inactive precursor of proteolytic enzyme kallikrein. Prekallikrein can be converted to active enzyme (which is serine protease) in a variety of ways. One of the physiological activators is Hageman factor (factor XII of blood clotting sequence). Hageman factor is normally in active form in plasma and is activated by contact with surface having a negative charge, such as collagen, basement membrane, bacterial lipopolysaccharides and urate crystals. As a result of increased vascular permeability that occurs in inflammation, Hageman factor, prekallikrein and kininogen leak out of vessels with plasma. Contact with the negatively charge surface promotes interaction of prekallikrein and Hageman factor and this leads to kinin generation, BK being clipped out of HMWK molecules by enzyme, which acts at two sites to release nonapeptide (Figure 2). Kallikrein can also activate complement system and can convert plasminogen to plasmin [9]. |
| |
|
|
Figure 3: The generation and breakdown of bradykinin. High-molecularweight kininogen (HMW-Kininogen) probably acts both as a substrate for kallikrein and as a cofactor in the activation of prekallikrein. |
|
| |
| In addition to the plasma kallikrein described above, there are other kinin –generation kallikreins found in pancreas, salivary glands, colon and skin. Tissue kallikreins act on both HMWK and LMWKs and generate mainly KD [1]. |
| |
| Inactivation |
| |
| The main enzymes that inactivate BK and related kinins are called kininases (Figure 2 and Figure 3). One of these, kininase II, is the same as ACE [10]. |
| |
| Kininase II is peptidyl dipeptidase that removes two C-terminal amino acid residues from kinin, thus inactivating it (Figure 2). The enzyme is bound to luminal surface of endothelial cells and it is found mostly in lung. It also cleaves two C-terminal residues from inactive peptide angiotensin I, converting it to active vasoconstrictor peptide angiotensin II. Thus, the enzyme inactivates vasodilator and activates vasoconstrictor [4]. |
| |
| Kinins are also inactivated by various less-specific kininases; one of these, carboxypeptidase present in serum (Figure 2), removes C-terminal arginine from BK, generating des-Arg 9-BK, which is a specific agonist of B1 receptor [3,6]. |
| |
| BK Receptors |
| |
| The biological actions of BK are mediated by specific receptors located on membranes of the target tissues. Two types of BK receptors, termed B1 and B2, which mediate very similar effects. Both are typical G-protein-coupled receptors [1]. |
| |
| B1 receptors appear to have limited distribution in mammalian tissues. The known functional roles for B1 receptors are determinate but may involve inflammation and long-lasting effects of BK, for example, collagen synthesis and cell multiplication [15]. By contrast, B2 receptors have widespread involvement which is consistent with multitude of biologic effects that are mediated by this receptor type [13]. |
| |
| B1-receptors are absent in most normal tissues, but are strongly inducible within few hours under conditions of inflammation and tissue damage; cytokines such as interleukin-1 (IL-1) are mainly responsible for this induction. B1- receptors respond to inactive BK metabolite (des-Arg9-BK), but not to BK itself, and are selectively blocked by various peptide antagonists. It is likely that B1-receptors play a significant role in inflammation and hyperalgesia [13]. B1 receptors, also, have been implicated in plasma extravasation, white blood cell activation and accumulation, and in control of blood pressure. B1 receptor activates phosphatidylinositol-spesific phospholipases C and possibly phospholipases A2 (PLA2) [16]. |
| |
| B1 receptor gene is composed of three exons. The entire coding region for receptor is contained within third exon. A variety of polymorphisms have been identified. One, G/C single nucleotide polymorphism, in the promoter region has been associated with diseases. Expression of C allele is higher than G allele, and patients with G allele have greater incidence of inflammatory bowel disease and endstage renal disease [3]. |
| |
| B1 receptor-mediated arachidonic acid release and prostaglandin (PG) synthesis are short-lived, which is similar to B2 receptors [10]. Activation of B1 receptors leads to elevation of intracellular free calcium activity by increasing calcium entry into the cell; this is different from B2 receptor which acts primarily to release bound intracellular calcium [1]. |
| |
| In animal models that their B1 receptor has been knockout; they develop normally and have normal blood pressure. On the other hand, when inflammatory stimuli are applied a dramatic reduction in accumulation and apoptosis of neutrophils and hypoalgesia have been reported [3,14]. |
| |
| B2-receptors are constitutively present in many normal cells and tissues and are activated by BK and KD, but not by BK metabolite (des- Arg9-BK). One exception is B2 receptor that mediates contraction of venous smooth muscle; this appears to be more sensitive to KD [10]. B2 receptors mediate broncho constriction, local blood flow regulation, hypotension, acute inflammatory reactions, pain, and hyperalgesia. Like B1 receptors, B2 receptors activate phosphotidylinositol-specific Phospholipase C. In addition, in most tissues, B2 receptor activation results in production of PGs and other arachidonic acid metabolites [13,14]. |
| |
| B2 receptor gene is composed of three exons, of which exon 2 and exon 3 provide coding region for receptor. Promoter region for B2 receptor contains a single nucleotide polymorphism; T/C. C allele has been demonstrated to be independent risk factor for essential hypertension in several ethnic groups [1,3]. |
| |
| B2 gene expression level is constitutive [9]. Activation of B2 receptor by binding of BK results in rapid internalization of receptor protein by endocytosis, occurring within few minutes. Internalization causes cessation of biological activity of receptor. Thus, activation of particular B2 receptor lead to transient increase in intracellular calcium activity and short-lived PG release [11]. |
| |
| In comparison to B1 receptors, activation of B2 receptors generates an increased intracellular calcium activity from release of calcium from intracellular stores [12]. |
| |
| Studies with B2 receptor knockout animals have shown that they developed normally. However, when these animals are fed highsalt diet, severe hypertension occurs. It also has been noticed that Renin-Angiotensin System (RAS) was abnormal (KKS and RAS are antagonistic hemodynamic regulatory systems that play important roles in blood pressure homeostasis) and abnormal renal development happened. Besides some cardiac defects as well as chronically elevated heart rate where identified [16]. |
| |
| The physiological effects of B1 receptors |
| |
| BK-B1-receptors have roles in different systems. Stimulation of B1-receptor can cause vasodilatation in blood vessels. B1-receptor has been shown to precondition heart against ischemic events and protect it from arrhythmias [4]. In addition, they are involved in renal function by affecting both natriuresis and glomerular filtration [17]. B1-receptors, also, contributed in pathogenesis of diabetes [6]. The roles of B1-receptors in inflammation are in leucocyte recruitment, initiation of inflammatory responses as well as physiology of pain. Recently, it has been determined that B1-receptors are mitogenic in fibrotic tissue [15]. |
| |
| The physiological effects of B2-Receptors |
| |
| B2-receptors produce many effects on a number of tissues. In vasculature and cardiovascular system, they can lead to vasoconstriction or vasodilatation, through either stimulation or inhibition of growth in parenchymal tissues, reduce infract size and precondition heart against ischemic events. Moreover, they have been shown to be antiarrhythmic in heart and antithrombotic in vasculature. In addition, B2 receptors improve myocardial demand of oxygen in heart failure by attenuating endothelial dysfunction [7,16]. Where, in diabetes they affect glucose metabolism either directly or by interaction with insulin [18]. BK and its receptors play a role in alimentary tract. They affect smooth muscle cells of duodenum, ileum, and cecum, causing either relaxation or contraction. B2-receptors have been implicated in the pathogenesis of asthma as they cause chloride secretion and bronchoconstriction. They, also, affect function of reproductive organs and bladder by inducing smooth muscle contraction in vas deferens, uterus, and bladder. B2- receptors have been involved in the pathophysiology of pain, sepsis, inflammation, hyperalgesia, rhinoviral infection, and rheumatoid arthritis [3]. |
| |
| The Effect of BK in Cardiovascular System |
| |
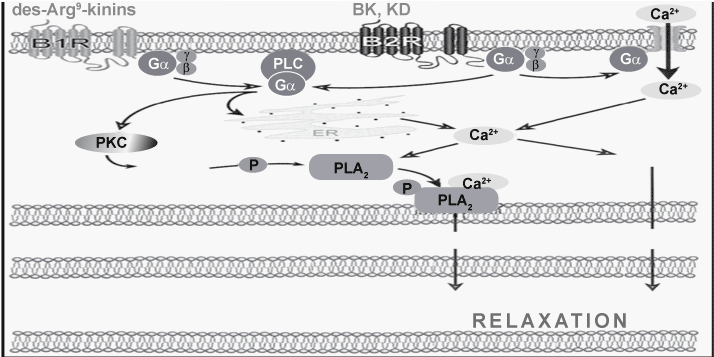
| BK is known for its multiple effects on cardiovascular system and particularly by its vasodilatation and plasma extravasation properties, leading to an inflammatory response [9]. Vasodilatation is normally mediated by B2 receptor, but under inflammatory conditions, B1 receptor up-regulation mediates kinin-induced vasodilatation and hypotension [19]. BK-related peptides act as vasodilators through endothelial cells from which secondary mediators are released to affect vascular smooth muscle [20]. In humans, cardiovascular actions of kinins (Figure 4) are mainly correlated to preformed B2 receptor stimulation (leading to NO and PG formation) and contribution of B1 receptor is not detectable [21]. NO is derived from L-arginine by endothelial NOS (eNOS). NO diffuses from endothelium to smooth muscle where it activates guanylate cyclase. NO-independent ion channel are also suspected to mediate endothelial-dependent vasorelaxation [22]. PG is also released by kinins from endothelial cells, properly via cytosolic calcium-sensitive isoform of PLA2 and stimulates cyclic Adenosine Monophosphate (cAMP) production in smooth muscle cells [21,22]. These physiological effects of kinins are potentially useful to treat hypertension and ischemic disorders and to maintain renal function (as KKS plays a role in handling salt excess) [9]. |
| |
|
|
Figure 4: Kinin receptors and their signaling pathways. Schematic representation of B1 and B2 receptors and the second messengers released by their activation. PLC: phospholipase C; ER: endothelial reticulum; DAG: diacylglycerol; IP3: inositol 1, 4, 5-triphosphate; PLA2: phospholipase A2; NO: nitric oxide; eNOS: endothelial NO synthase. |
|
| |
| Local generation of kinins or inhibition of their degradation and resulting B2 receptor stimulation could be of interest in reducing blood pressure or promoting cardioprotective effects [19]. |
| |
| On the other hand, B1 receptor activation has been shown to exert protective effect after cardiac ischemia in mice [19]. |
| |
| The Effects of BK in Cardiovascular Diseases |
| |
| Tissue kallikrein produces the potent vasodilator kinin (BK) from kininogen substrate, which binds to kinin receptor and triggers wide spectrum of biological effects [9]. The levels of tissue kallikrein are reduced in human and animal models with hypertension, cardiovascular and renal diseases [19]. A single injection of human tissue kallikrein gene in transgenic hypertensive mice resulted in prolonged lowering of blood pressure and attenuating hypertrophy, fibrosis in heart and kidney [20]. Furthermore, enhanced KKS levels after gene transfer exerted beneficial effects, with protection against cardiac remodelling, renal injuries, restenosis, and cerebral infraction in normotensive animal models without hemodynamic changes, indicating direct actions of kallikrein independent of its ability to lower blood pressure [21]. Moreover, KKS exhibited pleiotropic effects by inhibiting apoptosis, inflammation, hypertrophy, and fibrosis, and promoting angiogenesis and neurogenesis in heart, kidney, brain, and blood vessels [22]. The effects of kallikrein were mediated by BK B2 receptor, as specific B2 receptor antagonist, icatibant, abolished actions of kallikrein [19]. These results indicate that BK, through B2 receptor activation and NO formation, can protect against oxidative damage in cardiovascular and renal diseases and ischemic stroke [23]. And may uncover new drug targets for prevention and treatment of heart failure, vascular injury, end-stage renal disease and stroke in humans [24]. |
| |
|
|
| |
| Hypertension |
| |
| The pharmacological actions of BK in controlling systemic blood pressure were vasodilatation in most areas of circulation, reduction of total peripheral vascular resistance and regulation of sodium excretion from kidney [22]. When BK is injected into renal artery, it causes diuresis and natriuresis by increasing renal blood flow [24]. These consequences of BK have been attributed to PG release in renal circulation [21-23]. The role of KKS in hypertension was established with observations that urinary kallikrein excretion is significantly decreased in hypertensive patients. This led to the suggestion that reduced urinary kallikrein excretion might result from a defect in kinin generation in hypertensive situations [23]. |
| |
| BK and cardioprotection |
| |
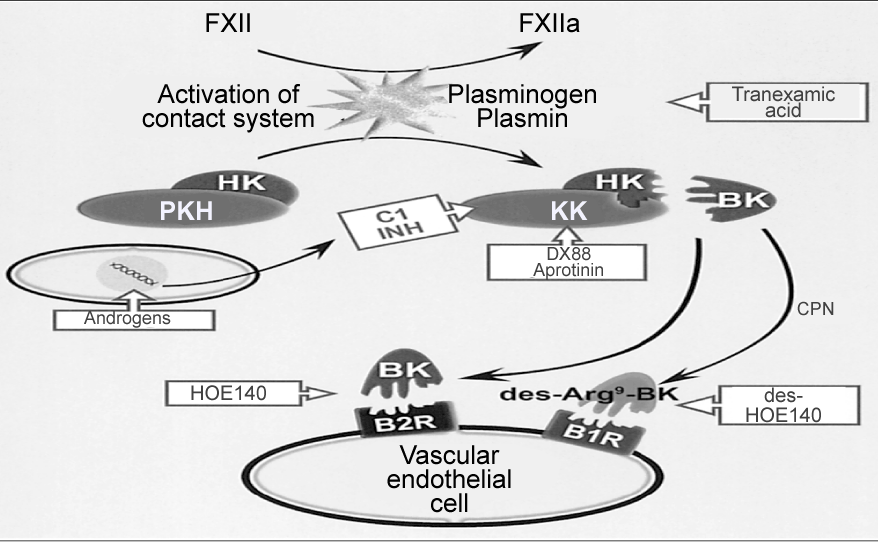
| Cardiac failure and ischemia: The local and systemic administration of BK can increase coronary blood flow and improve myocardial metabolism [22]. The binding of kinins to endothelial B2-receptors causes release of NO and PG, exerting vasodilator, anti- ischemic, and anti-proliferative effects, and preserving myocardial stores of energyrich phosphates and glycogen [20]. Kinins contribute to maintenance of cardiovascular homeostasis by opposing vasoconstrictor activity of angiotensin II [21]. On the other hand, kinins are continuously released during cardiac hypoxia and ischemia. They act as cardioprotective agents in perfusion and participate in the process of ischemic precondition [22]. Furthermore, kallikrein gene transfer improved cardiac function, and reduced myocardial infract size, incidence of ventricular fibrillation and apoptosis after acute ischemia-reperfusion via activation signal transduction pathways generating NO and PG [1] [Figure 6]. |
| |
|
|
Figure 6: Pharmacological targets to modulate the kallikrein-kinin activity. Tranexamic acid inhibits fibrinolysis and DX88 and C1INH inhibit the serine activity of plasma kallikrein, although androgens stimulate the synthesis of C1INH. B1 and B2 antagonists block the activation of their respective receptors. |
|
| |
| LVH: BK can counter the accretion of LVH in rats with hypertension. This anti-hypertrophic effect of BK was blocked by B2-receptor antagonist and NOS inhibitor. Thus, BK has a role in protecting heart against developing LVH by releasing NO [22]. Therefore, deficiency in components of KKS in heart may be the reason of myocardial dysfunction in maintaining high blood pressure and cardiac LVH [2]. |
| |
| Stroke: KKS has a potential protective role in stroke, because it reduced stroke-induced mortality, blood pressure, and aortic hypertrophy [3]. In addition, KKS significantly lowered ischemiainduced neurological deficits, cerebral infract volume and apoptosis, while promoting survival and migration of glial cells into ischemia core in haemorrhagic cerebral ischemic rat [24]. |
| |
| The BK Role in Renal System |
| |
| Tissue KKS is one of the major regulators of renal circulation, as it regulates arterial pressure, renal hemodynamic, and electrolyte excretion [17]. The present study showed that transgenic mice over expressing human BK B2 receptor are hypotensive and exhibited augmented renal hemodynamic, as manifested by increased renal blood flow, glomerular filtration rate, urine flow, urinary potassium excretion and pH [19]. Enhanced renal functions were accompanied by increased vasorelaxation factors, such as, NO, cyclic Guanosine Mono Phosphate (cGMP), and cAMP in kidney and urine [9]. So, these findings suggest that BK effects in renal system, which are, protection of kidney from renal injuries, improvement of renal function, natriuresis, and diuresis depend on NO synthesis [9,17]. |
| |
| Role of BK in Renal Diseases |
| |
| KKS and renal protection |
| |
| Current study indicates a novel role of KKS in protection against salt- and drug-induced renal injury by inhibiting oxidative stress and inflammation [24]. This was documented by giving intravenous infusion of urinary kallikrein to animal models; in which KKS reduced renal damage, diminished inflammatory cell accumulation, and enhanced renal function without affecting blood pressure [17]. Moreover, it is not only attenuated but also reversed salt-induced renal fibrosis in renal interstitium and vasculature and glomerular hypertrophy and restored NO production [16]. These protective effects of KKS were antagonised by icatibant [18]. |
| |
| Diabetic nephropathy |
| |
| Experimental investigations demonstrated that KKS may be a therapeutic target in preventing and treating diabetic nephropathy. In fact, ACE-inhibitors which inhibit degradation of BK lowered development of diabetic nephropathy in experimental animals and clinical settings. The role of KKS in diabetes is further supported by findings that diabetic nephropathy is worsened in diabetic mice lacking BK B2 receptors [25]. |
| |
| BK Agonists and Antagonists |
| |
| The properties of agonists and antagonists are summarized in Table 1 for B1 receptor and Table 2 for B2 receptor [9]. |
| |
|
|
Table 1: Pharmacological and clinical application of kinin B1-receptor agonists and antagonists. |
|
| |
|
|
Table 2: Pharmacological and clinical application of kinin B2-receptor agonists and antagonists. |
|
| |
| Antagonists for B1 receptor were discovered almost 10 years before B2 receptor; thus receptor nomenclature is justified by the fact that it was the first to be pharmacologically fully defined [19]. |
| |
| Some kinin antagonists have also been discovered as natural compounds: Martinelline, Pyrroloquinoline alkaloid isolated from plant Martinelline iquitosensis, is the most remarkable example [18]. |
| |
| The Undesirable Effects of BK |
| |
| Patients who present genetic deficiency in C1 inhibitor (C1 INH), which is protease inhibitor that is responsible for release of vasoactive BK, suffer from Hereditary Angio Edema (HAE). BK is probably the main but not the sole mediator accountable for increased vascular permeability that results in Angio Edema (AE). In fact, some researchers reported that generation of BK is associated with activation of fibrolysis during acute attacks of HAE. A model of HAE contributed to support hypothesis that BK mediates HAE. In this model, gene coding for C1 INH demonstrated increased permeability and depletion of HMWK when treated with specific plasma kallikrein inhibitor or B2 receptor antagonist, increased vascular permeability was completely reversed (Figure 6) [26]. |
| |
| Despite their clinical effectiveness, ACE-Inhibitors can cause chronic and acute side effects. The side effects that do occur are primarily related directly or indirectly to increased kinins since ACE is also kininase. These side effects include non-productive cough, angioneurotic oedema, and anaphylactoid reactions [19]. |
| |
| The cause and mechanism of ACE-Inhibitor-induced dry cough is not known, but increased local concentration of kinins (BK) or PGs may be important. Kinins may induce bronchial irritation and cough via enhanced production of PGs which may then stimulate afferent C-fibres in airways and sensitize bronchial contractility [27]. In addition, local accumulation of BK may lead to activation of proinflammatory peptides and a local release of histamine, inducing cough reflex hypersentivity [9]. |
| |
| Novel Development in BK System |
| |
| Roles of KKS in inflammation have been investigated and reviewed extensively [3]. Many diseases such as chronic inflammatory pain, oedema, asthma and sepsis have their basis in inflammatory response; development of novel antagonist drugs targeted at B1 and B2 receptors provides novel therapeutic opportunity. Clinical development of these drugs is at early stage, with few human clinical studies reported until now and mainly based on peptide compounds [4]. The potential therapeutic applications of kinin receptor ligands (not always antagonists) include cardiovascular and renal disorders, inflammation, pain, diabetes, asthma, and perhaps cancer [14]. Examples of using novel BK antagonists to prevent and/or treat cardiovascular diseases, include, treatment of HAE. The purpose of this treatment is to inhibit release of vasoactive peptide, BK, or to block its proinflammatory effects (Figure 6). This can be achieved by using Serine proteases inhibitors: C1 INH and Aprotinin are two seprins used in treatment of HAE. They have high affinity for plasma kallikrein and plasmin. Inhibition of plasma kallikrein, which triggers the release of BK during contact system activation, could lead to decreased release of BK during HAE attack [18]. |
| |
| The other example is Omapatrilat, which is vasopeptidase inhibitor (VPi). VPi possess ability to inhibit simultaneously two membranebound zinc metalloproteases, ACE and NEP, with similar nanomolar inhibitory constants. This dual inhibitor has been evaluated clinically for treatment of hypertension, heart failure, and renal disease. It is appeared to be a potent antihypertensive agent with favourable effects on cardiac function in heart failure patients [9]. |
| |
| Staphylococcus aureus (S. aureus) is a major pathogen of grampositive septic shock and associated with consumption of plasma kininogen. The examination of vascular leakage (VL) activity of two cysteine proteinases that are secreted by S. aureus showed that both induced VL in BK B2-receptor-dependent-manner. S. aureus also produced VL activity from human plasma, apparently by acting directly on kininogens to release BK. Collectively, these data suggest that production of BK is new mechanism of S. aureus virulence and bacterial shock. Therefore, BK-receptor antagonists could be used to treat this disease [28]. |
| |
| In humans, BK induces potent bronchoconstriction and cough when inhaled in asthmatic patients and it causes rhinitis-like symptoms when instilled into the nose. Furthermore, BK is generated in human nasal secretions during rhinoviral infections and allergic rhinitis. On the basis of these findings, therapeutic potential role of kinin B2 receptor antagonists has been hypothesized for treatment of airways inflammatory pathologies associated with hyper responsiveness to BK, such as, chronic bronchial asthma, or with release of BK, like, perennial and seasonal allergic rhinitis [29]. |
| |
| Icatibant (HOE 140), a widely used peptide B2 receptor antagonist, has been found to significantly improve the ventilatory function in humans with asthma when administrated in an aerosol form. The mode of action of this drug was not related to an acute bronchodilator action, but rather to a long-term anti-inflammatory effect [3]. |
| |
| |
| References |
| |
- Yousef GM and Diamandis EP(2007) The New Human Tissue Kallikrein Gene Family:Structure, Function and Association to Disease. Endocri Rev 22: 184-204.
- Sharma JN (2007) Does the Kinin System Mediate in Cardiovascular Abnormalities. J Clin Pharm 24: 34-36.
- Chakravarty S, Mavunkel BJ, Andy R and Kyle DJ (2004) Non-Peptidic Bradykinin Antagonists from a Structurally Directed Non-Peptide Library. https://www.netsci.org
- Dendorfer A and Wolfrum S (2003) Bradykinin. https://encyclopediathefreedictionary.com
- Sharma JN, Zubaid M, Khan BZ, Yousof AP, Asmawi MZ (2004) Pathophysiological Activities of the Kallikrein-Kinin System with Emphasis on the Cardiovascular Disorders. Inflammopharmacology 8: 219-241.
- Kuoppala A, Lindstedt KA, Saarinen J, Kovanen PT, Kokkonen JO (2000) Inactivation of Bradykinin by Angiotensin-Converting Enzyme and by Carboxypeptidase N in human plasma. Am J Physiol Heart Circ Physiol 278: 1069-1074.
- Campbell DJ, Krum H and Ester MD (2005) History and a Review of the Kinin System. https://www.patentstorm.us
- Hill RG and Oliver KR (2007) Neuropeptide and Kinin Antagonists. Exp Pharmacol 177: 181-216.
- Moreau ME, Garbacki N, Molinaro G, Brown NJ, Marceau F, et al. (2005) The kallikrein-kinin system: current and future pharmacological targets. J Pharmacol Sci 99: 6-38.
- Miles A (2006) The Kinin System: A History and Review of the Kinin System. https://www.nature.com
- Wennemuth G (2003) Peptides, Bradykinin. https://homepage.ntlworld.com
- Carr M (2003) Migraine and Bradykinin. https://www.anapec.com
- Kuga T, Mohri M, Egashira K, Hirakawa Y, Tagawa T (1997) Bradykinin-induced vasodilation of human coronary arteries in vivo: role of nitric oxide and angiotensin-converting enzyme. J Am Coll Cardiol 30: 108-112.
- Wiki C (2007) Description of Bradykinin. https://www.ch.ic.ac.wk
- Medeiros R, Cabrini DA, Ferreira J and Mori MA(2004) Bradykinin B1 receptor expression induced by tissue damage in the rat portal vein: a critical role for mitogen-activated protein kinase and nuclear factor-kappaB signaling pathways. Circ Res 94: 1365-1382.
- Dendorfer A, Wolfrum S and Dominiak P (1999) Pharmacology and Cardiovascular Implications of the Kinin-Kallikrein System. Jpn J Pharmacol 79: 403-426.
- Wang D, Yoshida H, Song Q, Chao L, Chao J (2000) Enhanced Renal Function in Bradykinin B2 Receptor Transgenic Mice. Am J Physiol Renal Physiol 278: 484-491.
- Burch RM (2004) Antibodies to des-Arg9-Bradykinin and Bradykinin Receptors. https://www.acris-antibodies.com
- Margolius HS (1995) Theodore Cooper Memorial Lecture. Kallikreins and kinins. Some unanswered questions about system characteristics and roles in human disease. Hypertension 26: 221-229.
- Scholkens BA (1996) Kinins in the Cardiovascular System. Immunopharmacology 33: 209-216.
- Margoluis HS (2005) The Kallikrein-Kinin System, Renal Function and Hypertensive Disease. Prog Biochem Pharmacol 17: 116-122.
- Chen ZL, Deddish PA, Becker RP and Erdos EG (2006) Cardiovascular Properties of the Kallikrein-Kinin System. Exp Biol 20: 2261-2270.
- Chao J, Bledsoe G, Yin H, Chao L (2006) The tissue kallikrein-kinin system protects against cardiovascular and renal diseases and ischemic stroke independently of blood pressure reduction. Biol Chem 387: 665-675.
- Chaol J and Chaol L (2005) Kallikrein-Kinin in Stroke, Cardiovascular and Renal diseases. Exp Physiol 90: 291-298.
- Riad A, Zhuo JL, Schultheiss HP, Tschöpe C (2007) The Role of the Renal Kallikrein-Kinin System in Diabetic Nephropathy. Curr Opin Nephrol Hypertens 16: 22-26.
- Carretero OA (2005) Vascular Remodeling and the Kallikrein-Kinin System. J Clin Invest 115: 588-591.
- Norman MK and Burton DR (2000) Major Side Effects of ACE Inhibitors. Diabetes Care 14: 34-39.
- Imamura T, Tanase S, Szmyd G, Kozik A, Travis J, et al. (2006) Induction of Vascular Leakage through Release of Bradykinin and a Novel Kinin by Cysteine Proteinases from Staphylococcus aureus. J Exp Med 201: 1669-1676.
- Valenti C, Cialdai C, Giuliani S, Lecci A, Tramontana M (2005) MEN16132, a Novel Potent and Selective Non-peptide Kinin B2 Receptor Antagonist: In Vivo Activity on Bradykinin-Induced Bronchoconstriction and Nasal Mucosa Microvascular Leakage in Anesthetized Guinea Pigs. J Pharmacol Exp Ther 315: 616-623.
|
| |
| |