| Rapid Communication |
Open Access |
|
| Marli Camassola* and Aldo J.P. Dillon |
| Institute of Biotechnology, University of Caxias do Sul 1130 Francisco Getúlio Vargas Street, Caxias do Sul, RS, Brazil |
| *Corresponding author: |
Marli Camassola
Institute of Biotechnology
University of Caxias do Sul 1130 Francisco Getúlio Vargas Street
Caxias do Sul, RS, Brazil
Zip Code 95070-560
Tel: 55 54 32182100 Ext:2681
Fax : 55 54 3218 2149
E-mail: mcamassola@gmail.com |
|
| |
| Received June 23, 2012; Published June 31, 2012 |
| |
| Citation: Camassola M, Dillon AJP (2012) Cellulase Determination: Modifications to Make the Filter Paper Assay Easy, Fast, Practical and Efficient. 1: 125. doi:10.4172/scientificreports.125 |
| |
| Copyright: © 2012 Camassola M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. |
| |
| Abstract |
| |
| There has been a notable increase in interest about cellulases in recent years due to their numerous potential applications, including the hydrolysis of cellulose in lignocellulosic biomass to produce reducing sugars for the development of second-generation ethanol. However, the traditional cellulase assays are tedious, time-consuming and labor intensive. What is more, they require large amounts of reagents and produce large quantities of toxic effluents? The Filter Paper Assay (FPA) is the standard measure of cellulase activity, according to the International Union of Pure and Applied Chemistry. Although it can be reproduced in most laboratories, with some practice, this method has long been known for its complexity and many manual manipulations. In this study, adaptations were made to the FPA to create a low-cost, easier, faster, less labor intensive and less polluting alternative, while maintaining similar results to the standard method. |
| |
| Keywords |
| |
| Cellulase determination, Filter Paper Assay, Practical and efficient method |
| |
| Introduction |
| |
| Cellulose is a major renewable form of carbohydrate, with approximately 1011 tons synthesized annually. It is an unbranched β-1,4-linked homopolymer of glucose [1], but cellulose samples of different origins vary widely in their chain lengths and in the degrees of interaction between chains. As a raw material, cellulose has currently the greatest potential to produce bioethanol; however, cellulose must be hydrolyzed to obtain fermentable sugars and the cellulolytic enzymes are central to the processing of biomass for bioethanol production. |
| |
| A cellulolytic enzyme is a complex system of enzymes, comprising endoglucanase (endo-1,4-β-D-glucanase, EC 3.2.1.4), exo-glucanase (1,4-β-D-glucan-cellobiohydrolase, EC 3.2.1.91) and β-glucosidase (β-D-glucoside glucanohydrolase, cellobiase, EC 3.2.1.21), which act synergistically to degrade cellulosic substrate [2,3]. |
| |
| Many reports use the Filter Paper Activity/Assay (FPA) to determine the quantity of cellulolytic enzymes required to hydrolyze lignocellulosic biomass. The FPA was initially proposed by Mandels et al. [4] as a simple, reproducible and quantitative method that predicts enzyme action under practical saccharification conditions. This method is very useful, as demonstrated by Eveleigh et al. [5] in a republication of the original article. The FPA, however, remains a very laborious and time-consuming method, requiring many manual manipulations and producing large quantities of toxic effluents. |
| |
| Decker et al. [6] proposed an attractive method for automated filter paper activity/assay determination, but the results obtained by these authors were not similar to those obtained with the standard method proposed by Mandels et al. [4]. The standard method, performed manually, resulted in a FPA of 38.6 FPU/mL in the commercial cellulase preparation. The same cellulase mixture on Whatman n. 1 filter paper with other cellulose substrates, assayed in microtiter plates, resulted in a varied range of activities. In the microtiter plate-based assays performed using the Cyberlabs C400 robots, the commercial cellulase preparation resulted in an apparent FPA activity of 60.4 FPU/ mL on filter paper (disks with an average weight of 2.65 mg), 35.2 FPU/ mL on SigmaCell-20, 34.7 FPU/mL on Solka-Floc, 27.8 FPU/mL on Avicel PH101 and 14.1 FPU/mL on cotton linters. |
| |
| The aim of this study was to improve the method proposed by Mandels et al. [4] and Eveleigh et al. [5], thus creating an easier, faster, less labor intensive and less polluting alternative. |
| |
| Concomitant with the development of this new method, it was also used the methodologies proposed by Decker et al. [6] and Xiao et al. [7]. However, the liquid/solid (buffer plus enzyme and substrate) ratios proposed in the standard method were retained. By using the simple adjustments suggested in this paper, it is possible to reduce reagent expenses by ten-fold, as well as reducing preparation time by at least 75% while obtaining similar results to the standard method. |
| |
| Materials and Methods |
| |
| Reagents and materials |
| |
| The total cellulase activity was determined using Whatman n.1 filter paper. |
| |
| The reducing sugar reagent was the standard FPA DNS reagent, which contained 283.2 mL deionized water, 2.12 g 3, 5-dinitrosalicylic acid, 3.96 g NaOH, 61.2 g Rochelle salts (sodium potassium tartrate), 1.52 mL phenol and 1.66 g sodium metabisulfite. This solution was prepared as described by Miller GL [8]. |
| |
| Anhydrous glucose (2.0 mg/mL) was used as the reducing sugar stock standard and it was diluted to produce a standard curve. |
| |
| Enzymes |
| |
| This study used enzyme preparations derived from Penicillium echinulatum 9A02S1 (DSM 18942) and Trichoderma reesei (RUT C30) that were produced at the University of Caxias do Sul (Caxias do Sul, RS, Brazil) using lab-scale fermenters and solid state culture [9-14]. The submerged media used for cellulase production contained: 0.2 g/L-1 KH2PO4; 0.13 g/L (NH4)SO4; 0.03 g/L CO(NH2)2; 0.03 g/L MgSO4.7H2O; 0.03 g/L CaCl2; 0.5 mg/L FeSO4.7H2O; 0.156 mg/L MnSO4.H2O; 0.14 mg/L ZnSO4.7H2O; 0.14 mg/L CoCl2; 1% (w/w) microcrystalline cellulose or sugar cane bagasse; and 0.1% (w/w) microbiological grade peptone. The inoculum contained 105 spores·mL-1 and was prepared in a medium containing 0.5% (w/w) wheat flour. Cellulase production was conducted for 4 days at 28°C and 150 rpm. |
| |
| For solid state culture, Wheat Bran (WB) Sugar Cane Bagasse (SCB) and Steam Explosion Sugar Cane Bagasse (SESCB) were used as the support and main carbon sources. The culture media consisted of WB and SCB. Solid state cultures were incubated in static tray bioreactors closed with a gauze-covered cotton wool plug containing 50 g dry mass of production media and 50 mL basal salt solution containing: 20 g/L KH2PO4; g/L (NH4)2SO4; 3 g/L CO(NH2)2; g/L MgSO4.7H2O; 3 g/L CaCl2; 500 mg/L FeSO4.7H2O; 156 mg/L MnSO4.H2O; 140 mg/L ZnSO4.7H2O and 20 mg/L CoCl2. |
| |
| All flasks were autoclaved at 120°C for 30 min. Each flask was then inoculated with sufficient conidial suspension to give a final concentration of 1×106 conidia per gram dry mass of production media. The moisture of the media was adjusted to 67% with distilled water. The flasks were then incubated at 28°C and 90% humidity for 4 days. To extract the enzymes after incubation, the contents of each flask were separately added to a 2 L-Beaker flask containing 1 L 50 mM sodium citrate buffer (pH 4.8). The contents were mixed and incubated with agitation for 30 min at 4°C and then filtered. This filtrate was used for the enzyme assays. |
| |
| Filter Paper Assay Protocols |
| |
| All values of enzyme activity are averages obtained from assays that were performed in triplicate. The enzymes were analyzed using three methods: 1) the standard method as described by Mandels et al. [4] and Eveleigh et al. [5]; 2) the 60 μl microplate assay as described by Xiao et al. [7]; and 3) the “Reduced method”, as proposed by this paper. |
| |
| Figure 1 shows the steps involved in the determination of FPAs. All steps were adapted, but the proportions described in the standard method were preserved. |
| |
|
|
Figure 1: Comparison of the steps for FPA determination between the Standard method, proposed by Mandels et al. and Eveleigh et al. and the adapted method (Reduced method). |
|
| |
| Enzyme units |
| |
| One unit (U) enzyme activity was defined as the amount of enzyme required to liberate 1 μmol reducing sugar from the appropriate substrate per min under the assay conditions. The results are shown in U·mL-1 for submerged and solid state cultivation. |
| |
| Statistical tests |
| |
| The results were statistically analyzed using Prism GraphPad software and analysis of variance with Tukey’s post-test with p<0.05. Origin software was also used to conduct t-tests, when necessary. |
| |
| Results and Discussion |
| |
| The tests were conducted by employing enzymes produced in submerged and solid state cultures by the fungi P. echinulatum 9A02S1 and Trichoderma reesei Rut C30. |
| |
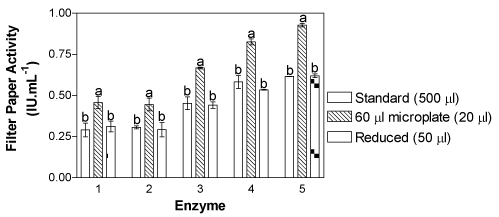
| In Figure 2, the data obtained using the five enzymes and examined with all three methods is shown. It appears that the method performed using a smaller final volume (60 μL), as proposed by Xiao et al. [7], showed higher activity for all enzymes tested. The activities were statistically higher than the standard and the reduced methods. |
| |
|
|
Figure 2: Comparison of Penicillium echinulatum cellulase activity measured using the standard FPA, the 60 μl microplate FPA and the reduced FPA (proposed in this paper) methods. Enzymes 1 and 2 were produced in a submerged culture using cellulose and sugar cane bagasse as the inductor and carbon source, respectively. Enzymes 3, 4 and 5 were produced in solid state cultures using only wheat flour (WB), sugar cane bagasse plus WB (1:1) and steam explosion sugar cane bagasse plus WB (1:1), respectively. Methods with the same lower case letter for the same enzyme do not differ statistically by Tukey’s test (p > 0.05). Methods with the same capital letters for the same enzyme are statistically similar, according to the t-test (p > 0.05). The values in brackets in the legend indicate the volume of enzyme used for testing. |
|
| |
| When comparing the results from the standard and the reduced methods, a statistic similarity (using Tukey’s test) is noted, except for enzyme 3, which was produced in solid state culture using WB. However, when activities obtained with the standard and reduced methods were only compared for this enzyme, there was no significant difference (using the t-test). |
| |
| |
| When comparing the different FPA methods using T. reesei Rut C30 enzymes, the microplate assay once more showed the highest mean activities, and these values were significantly higher than those produced through the other two methods. Nevertheless, the standard and reduced methods produced statistically similar data, as shown in Figure 3. |
| |
|
|
Figure 3: Comparison of Trichoderma reesei Rut C30 activities measured using the standard FPA, the 60 μl microplate FPA and the reduced FPA (proposed in this paper). Enzymes 1 and 2 were produced in submerged cultures using cellulose and sugar cane bagasse, respectively. The enzymes 3, 4 and 5 were produced in solid state cultures using only wheat flour (WB), sugar cane bagasse plus WB (1:1) and steam explosion sugar cane bagasse plus WB (1:1), respectively. Methods with the same lower case letter for the same enzyme do not differ statistically by Tukey’s test (p > 0.05). The values in brackets in the legend indicate the volume of enzyme used in testing. |
|
| |
| The FPA has been widely used to measure total cellulase activity, since Mandels et al. [4] proposed a number of standard procedures for the measurement of cellulase activity. Whatman n.1 Filter paper is used as the standard substrate because it is readily available and inexpensive (Figure 4). What is more, assays performed with this filter paper provide reliable and reproducible results [15]. However, the Mandels FPA is time consuming, labor intensive, and requires large amounts of reagents. Thus, the method proposed by Xiao et al. [7] was performed using a total volume of 60 μl, as described in Table 1. Nevertheless, the data obtained with this method were different from those of the standard method; perhaps, because it did not use the same ratio of reaction volume to filter paper. |
| |
|
|
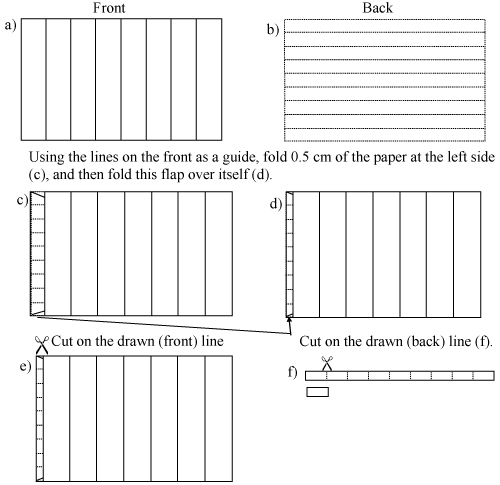
Figure 4: Preparation of Whatman n.1 filter paper strips.
Draw parallel lines on the front of the sheet, leaving exactly 1 cm between them. On the back, draw parallel lines leaving 0.6 cm between them, as shown in Figure 2 (a) and Figure 2 (b). |
|
| |
|
|
Table 1: Comparison of the different reaction volumes and components of the filter paper assays. |
|
| |
| In Table 1, the relative liquid/solid area and relative liquid/solid weight for the assays is shown. When comparing the relative liquid/ solid area or relative liquid/solid weight for the Standard FPA and 60- μl FPA, it was noted that the method proposed by Xiao et al. [7] used a lower quantity of liquid (buffer and enzyme) in relation to the amount and area of substrate. This justifies the higher activities obtained when using the microplate methodology. Xiao et al. [7] attempted to use the same ratio of reaction volume to filter paper area for their microplatebased assay, as it is used for the standard FPA, but similar results were not obtained. |
| |
| The data obtained through the method here proposed (Reduced FPA) is the result of simple modifications, which allowed the same liquid-to-substrate ratio used in the standard method. |
| |
| Decker et al. [6] also reported a miniature-scale FPA. However, the detectable filter paper activities were 56% higher than those obtained with the standard FPA. Interestingly, in this study, the average activity obtained for the enzymes analyzed by the 60-μL FPA microplate were 56.12% (P. echinulatum) and 48.60% (T. reesei) higher than those obtained with the standard FPA. However, with the Reduced FPA method, water evaporation from the 96-well plate was prevented after 60 min incubation at 50°C and 5 min incubation at 100°C in the water bath (data not shown), because the plate lids were closed with a silicone seal, which remained closed at 50°C. |
| |
| The seal allowed to centrifuge the plates, thus reincorporating the liquid that condensed on the undersurface of the lid during the incubations. Although the lid will come off at 100°C, it remains over the well and inhibits evaporation. It is possible to close the lid subsequently, by applying light pressure. In addition, the plates were incubated in environments with high humidity. |
| |
| Conclusions |
| |
| The reproducibility of the method proposed here was carefully evaluated in preliminary experiments. Although the collected data indicate the possibility of realizing measurement of total cellulases in smaller volumes than those proposed in the standard method, and in a less laborious way, the ideal methodology is still a controversial issue in the literature. The search for cellulolytic microorganisms, carbon sources and inductors, and the optimization process for cellulase production demand a large number of assays. For these reasons, the use of a practical, less polluting and less costly method is of fundamental importance for science and the environment. |
| |
| |
| References |
| |
- Tahoun MK, Ibrahim AA (1999) Conversion of natural cellulosic substrates into fermentable sugar by recombinant fungi strains Zeitschrift für Untersuchung der Lebensmittel 208: 65-68.
- Singhania RR, Sukumaran RK, Patel AK, Larroche C, Pandey A (2010) Advancement and comparative profiles in the production technologies using solid-state and submerged fermentation for microbial cellulases. Enzyme Microb Technol 46: 541-549.
- Chandra M, Kalra A, Sangwan NS, Gaurav SS, Darokar MP, et al. (2009) Development of a mutant of Trichoderma citrinoviride for enhanced production of cellulases. Bioresour Technol 100: 1659-1662.
- Mandels M, Andreotti R, Roche C (1976) Measurement of saccharifying cellulase. Biotechnol Bioeng Symp6: 21-33.
- Eveleigh DE, Mandels M, Andreotti R, Roche C (2009) Measurement of saccharifying cellulase. Biotechnol Biofuels 2: 21.
- Decker SR, Adney WS, Jennings E, Vinzant TB, Himmel ME (2003) Automated filter paper assay for determination of cellulase activity. Appl Biochem Biotechnol 105-108: 689-703.
- Xiao Z, Storms R, Tsang A (2004) Microplate-based assay to measure total cellulase activity. Biotechnol Bioeng 88: 832-837.
- Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31: 426-428.
- Camassola M, Bittencourt LR, Shenem NT, Andreaus J, Dillon AJ (2004) Characterization of the cellulose complex of Penicillium echinulatum. Biocatal Biotransf 22: 391-396.
- Dillon AJ, Zorgi C, Camassola M, Henriques JA (2006) Use of 2-deoxyglucose in liquid media for the selection of mutant strains of Penicillium echinulatum producing increased cellulase and ß-glucosidase activities. Appl Microbiol Biotechnol 70: 740-746.
- Camassola M, Dillon AJ (2007) Effect of methylxanthines on production of cellulases by Penicillium echinulatum. J Appl Microbiol 102: 478-485.
- Camassola M, Dillon AJ (2007) Production of cellulases and hemicellulases by Penicillium echinulatum grown on pretreated sugar cane bagasse and wheat bran in solid-state fermentation. J Appl Microbiol 103: 2196-2204.
- Camassola M, Dillon AJ (2009) Biological pretreatment of sugar cane bagasse for the production of cellulases and xylanases by Penicillium echinulatum. Ind Crops Prod 29: 642-647.
- Camassola M, Dillon AJ (2010) Cellulases and xylanases production by Penicillium echinulatum grown on sugar cane bagasse in solid-state fermentation Appl Biochem Biotechnol 162: 1889-1900.
- Coward-Kelly G, Aiello-Mazzari C, Kim S, Granda C, Holtzapple M (2003) Suggested improvements to the standard filter paper assay used to measure cellulase activity. Biotechnol Bioeng 82: 745-749.
|
| |
| |