Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
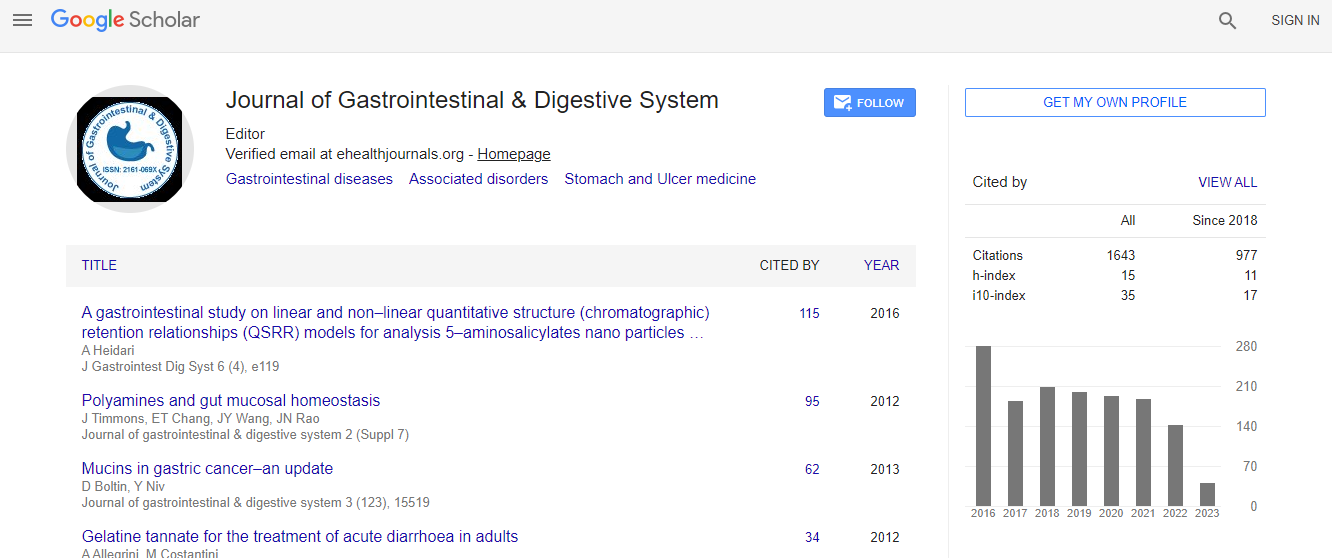
Google Scholar citation report
Citations : 1636
Journal of Gastrointestinal & Digestive System received 1636 citations as per Google Scholar report
Journal of Gastrointestinal & Digestive System peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Pediatric population a miniature adult - A delusion
International Conference on Pediatric Gastroenterology and Pediatric Practices
Arti R Thakkar
University of North Carolina at Chapel Hill, USA Baddi University of Emerging Sciences & Technology, India
Posters & Accepted Abstracts: J Gastrointest Dig Syst
Abstract
Pediatric population represents a changing and dynamic population due to the ontogenetic changes that occur during their development. As it has been observed that, pediatrics does not deal with miniature adults, with reduced doses and has its own independent range and horizon. So prescribing the medication for pediatric population is a unique challenge as their anatomy and physiology is completely different from adults. In particular, age-related changes in gastrointestinal (GI) physiology (transit time, pH, GI fluid composition, specifically bile salt concentrations) and in intestinal/hepatic drug metabolizing enzymes, as well as formulation parameters (dose volume, solubility) can contribute to differential absorption of orally administered drugs in children versus adults. For example, oral bioavailability of the potent antifungal lipophilic drug, voriconazole, in children (2-10 years) is almost half that in adults (45-65% versus 96%). The present study describes the facts and current scenario in formulation development aspects for pediatric populations and determining various factors responsible for variable bioavailability of pediatric medicaments in children. Absorptive transport of pediatric drugs was measured across intestinal tissue using Using-type diffusion chamber as a function of Fasted-State Simulated Intestinal Fluid (FaSSIF) with variable bile salt concentration. Dissolutions studies were determined of the model drugs as a function of volume and media with variable bile salt concentrations. It was found that dissolution and absorptive transport of model drugs has reduced rate of dissolution and variable absorptive transport. Thus, normalization of the adult dose may lead to poor and unsafe estimates of the pediatric dose.Biography
Email: artirthakkar@gmail.com

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi