Vascular Calcification by Promoting Osteoblastic Differentiation of Vascular Smooth Muscle Cells
Received: 05-Sep-2022 / Manuscript No. asoa-22-77594 / Editor assigned: 07-Sep-2022 / PreQC No. asoa-22-77594 (PQ) / Reviewed: 21-Sep-2022 / QC No. asoa-22-77594 / Revised: 23-Sep-2022 / Manuscript No. asoa-22-77594 (R) / Accepted Date: 26-Sep-2022 / Published Date: 28-Sep-2022
Abstract
Vascular calcification is often associated with atherosclerotic lesions. Moreover, the process of atherosclerotic calcification has several features similar to the mineralization of skeletal tissue. Therefore, we hypothesized that vascular smooth muscle cells might acquire osteoblastic characteristics during the development of atherosclerotic lesions. In the present study, we investigated the effect of dexamethasone (Dex), which is well known to be a potent stimulator of osteoblastic differentiation in vitro, on vascular calcification by using an in vitro calcification model. We demonstrated that Dex increased bovine vascular smooth muscle cell (BVSMC) calcification in a dose- and time-dependent manner. Dex also enhanced several phenotypic markers of osteoblasts, such as alkaline phosphatase activity, procollagen type I carboxy-terminal peptide production, and cAMP responses to parathyroid hormone in BVSMCs. We also examined the effects of Dex on human osteoblast-like (Saos-2) cells and compared its effects on BVSMCs and Saos-2 cells. The effects of Dex on alkaline phosphatase activity and the cAMP response to parathyroid hormone in BVSMCs were less prominent than those in Saos-2 cells. Interestingly, we detected that Osf2/Cbfa1, a key transcription factor in osteoblastic differentiation, was expressed in both BVSMCs and Saos-2 cells and that Dex increased the gene expression of both transcription factors. These findings suggest that Dex may enhance osteoblastic differentiation of BVSMCs in vitro.
Keywords
Alkaline phosphatase; Core binding factor-α1; Atherosclerosis
Introduction
To clarify the mechanism of vascular calcification, we developed an in vitro calcification system in which diffuse calcification can be induced by culturing bovine vascular smooth muscle cells (BVSMCs) in the presence of β-glycerophosphate (β-GP). In this model, alkaline phosphatase (ALP), which is 1 of the markers for osteoblastic differentiation, is critical for vascular calcification and the expression of osteopontin mRNA, which increases during the development of calcification. Furthermore, we identified a local calcium-regulating system in which parathyroid hormone (PTH) related peptide plays an important role as an autocrine / paracrine regulator of vascular calcification [1].
Through recent evidence demonstrated by us and other investigators, we hypothesized that VSMCs might acquire osteoblastic characteristics during the development of atherosclerotic lesions [2].
Osteoblastic differentiation is a multistep process, proceeding through defined stages of maturation from a committed progenitor cell of mesenchymal origin capable of proliferation to a postproliferative osteoblast expressing bone phenotypic markers. However, the molecular basis of osteoblast- specific gene expression and differentiation remains unclear. Recently, a key regulatory transcription factor in osteoblastic differentiation, osteoblast-specific transcription factor-2 / core binding factor-α subunit 1 (Osf2/Cbfa1), has been identified. The Osf2/Cbfa1 gene generates 2 types of transcripts, osteoblast-specific and T cellspecific isoforms. In the mouse, the osteoblast isoform is different from the T-cell isoform in that the former contains a unique 87-amino acid sequence at its amino-terminal end. However, the precise roles of the 2 transcripts in osteoblastic differentiation still remain unclear. The homozygous Osf2/Cbfa1 (-/-) mouse shows a total lack of bone and a retention of the partially calcified cartilaginous skeleton.18 In humans, mutations of this gene cause cleidocranial dysplasia, an autosomal dominant skeletal disorder [3].
In this study, we investigated the effect of Dex on vascular calcification by using an in vitro calcification model. We first demonstrated that Dex increased calcium deposition in a time- and dose-dependent manner. In this process, Dex increased ALP activity, its mRNA expression, and procollagen type I C-peptide (PICP) production and influenced cAMP responses to PTH. Finally, we demonstrated that Dex promoted expression of the Osf2/Cbfa1 gene. These results suggest that Dex may stimulate vascular calcification by promoting osteoblastic differentiation of VSMCs [4-7].
Results
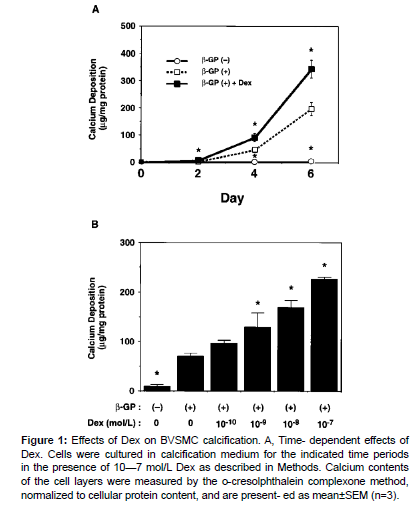
We first examined the effect of Dex on BVSMC calcification. As previously described, β-GP induced calcium deposition in a timedependent manner. In the presence of β-GP, Dex (10-7 mol/L) significantly increased calcium deposition compared with calcified controls at each time point (Figure 1). The calcium deposition in the Dex-treated group increased to 175% of the calcified control value on day 6. Likewise, Dex promoted calcium deposition in a dose-dependent manner on day 4, and the calcium deposition increased to 322% of the calcified control value at 10-7 mol/L. These results suggest that Dex increases BVSMC calcification [8].
Figure 1: Effects of Dex on BVSMC calcification. A, Time- dependent effects of Dex. Cells were cultured in calcification medium for the indicated time periods in the presence of 10—7 mol/L Dex as described in Methods. Calcium contents of the cell layers were measured by the o-cresolphthalein complexone method, normalized to cellular protein content, and are present- ed as mean±SEM (n=3).
ALP is known to be 1 of the phenotypic markers of osteoblastic differentiation. Because we reported that ALP plays an important role in this calcification system, we next examined the effect of Dex on ALP activity in BVSMCs. As a positive control of Dex’s effect, we utilized human osteoblast-like (Saos-2) cells. In the absence of β-GP, Dex (10-7 mol/L) enhanced ALP activity in a time-dependent manner, and ALP activity had increased to 222% of controls on day 6. On day 4, Dex dose-dependently increased ALP activity in the absence of β-GP, and the maximal effect (236% of control) was observed at 10-7 mol/L. In the presence of β-GP, Dex also increased ALP activity in BVSMCs, but the response to Dex was less prominent than that in its absence (Table 1). Furthermore, a greater response was observed in Saos-2 cells, both in the absence and presence of β-GP. Next, we examined the effect of Dex on expression of the ALP gene in BVSMCs. Dex dose-dependently promoted the expression of ALP mRNA at 48 hours, and the maximal effect was observed at 10-7 mmol/L (180% increase of control). Taken together, these results suggest that Dex may accelerate BVSMC calcification partially through enhancing expression of the ALP gene and its activity [9-12].
| BVSMCs | β-GP- | β-GP+ | β-GP- | β-GP+ | |
|---|---|---|---|---|---|
| Ctl Dex |
0.66±0.04 1.59±0.03* |
0.50±0.02 0.96±0.00* |
0.41±0.02 0.60±0.05* |
0.42±0.03 0.43±0.03 |
|
| Saos-2 cells Ctl | 3.96±0.94 | 2.87±0.86 | 0.47±0.05 | 0.20±0.01 | |
| Dex | 29.35±5.21* | 23.12±5.55* | 0.45±0.02 | 0.24±0.01 | |
Table 1: Effects of Dex on ALP Activities and PICP Production in BVSMCs and Saos-2 Cells.
Discussion
Because glucocorticoids are well known to be potent stimulators of osteoblastic differentiation, we utilized Dex, a potent synthetic glucocorticoid, to induce BVSMCs to acquire osteoblastic characteristics. As shown in this study, Dex enhanced not only in vitro calcification but also several phenotypic markers for osteoblastic differentiation in BVSMCs, such as ALP expression, type I collagen produc- tion, and cAMP responsiveness to PTH. Moreover, we confirmed the potency of Dex on osteoblastic differentiation by utilizing Saos-2 cells as a positive control. Regarding ALP activity and the cAMP response to PTH, the responsiveness of BVSMCs to Dex was less prominent than that of Saos-2 cells. The less potent effect of Dex on BVSMCs may be ascribed to a heterogeneous population of cells in the BVSMC culture. Interestingly, Dex exerted no effect on PICP production by Saos-2 cells, whereas Dex increased PICP secretion by BVSMCs in the absence of β-GP. Early phenotypic markers for osteoblastic differentiation such as PICP may not be affected by Dex in well-differentiated osteoblastic cells. However, the precise mechanism by which such differences of responsive- ness to Dex are induced remains to be clarified.
Several key factors in bone mineralization have been demonstrated in calcified lesions of arterial walls, such as matrix vesicles, BMP-2, osteopontin, matrix Gla protein, osteocalcin, and type I collagen. We previously dem- onstrated the significance of ALP, osteopontin, and PTH- related peptide in an in vitro model of vascular calcification by utilizing BVSMCs. Recently, a key regulatory factor in osteoblastic differentiation, Osf2/Cbfa1, has been identi- fied. BMP-7 induces expression of the osteoblastic isoform, followed by its enhancement of the osteocalcin gene in nonosteoblastic cells. Therefore, the Osf2/Cbfa1 gene is thought to be 1 of the “master genes” of as well as a molecular marker for osteoblastic differentiation. In this study, we showed the presence of the Osf2/Cbfa1 gene in cultured BVSMCs as well as in Saos-2 cells. Additionally, we cloned a 5’ partial sequence of the bovine osteoblast–specific Osf2/Cbfa1 transcript by reverse transcription– polymerase chain reaction by using total RNA from BVSMCs in preliminary experiments. This evidence suggests that cultured VSMCs may be committed to differentiate into osteoblastic cells under certain conditions. However, whether the tran- script detected in BVSMCs is the osteoblast-specific isoform remains to be confirmed. Furthermore, Dex enhanced the gene expression of Osf2/Cbfa1 in a time dependent manner in BVSMCs. Therefore, it is likely that Dex may promote osteoblastic differentiation of VSMCs by increasing the expression of the Osf2/Cbfa1 gene.
Linkage of phenotypic gene induction to the downregulation of proliferation is the hallmark of differentiation in numerous cell types.40 Some agents inhibiting the proliferation of osteoblast lineage cells, such as hydroxyurea, can induce osteoblastic differentiation. It is therefore possible that antiproliferative agents of VSMCs may induce osteoblastic differentiation under certain conditions. Moreover, 17β- estradiol has been reported to promote osteoblastic differentiation of bovine vascular cells and in vitro calcification without affecting cell growth. In this study, we examined the hypothesis that Dex may inhibit the proliferative capacity of BVSMCs, resulting in osteoblastic differentiation. Because Dex did not affect DNA synthesis in the presence of 15% FCS, Dex may directly induce osteoblastic differentiation of BVSMCs without affecting their growth.
The mechanisms of glucocorticoid action on atherogenesis remain to be evaluated. When applied as anti-inflammatory drugs at high doses, glucocorticoids suppress the development of atherosclerosis in experimental animals, despite enhancement of hypertriglyceridemia and hypercholesterolemia. Glucocorticoids have also been shown to inhibit the proliferation of cultured VSMCs and the thrombin- induced expression of growth factors. On the other hand, glucocorticoids are capable of decreasing the expression of hepatic LDL receptors,stimulating the net synthesis of apoB-100 and apoB-48 and decreasing their intracellular degradation. These changes are potentially atherogenic, and the strong correlation between an increased serum cortisol level in humans and the extent of coronary artery disease has also been documented. In this study, we have shown that Dex increases in vitro calcification by promoting osteoblastic phenotypes in BVSMCs. Taking into consideration that chronic treatment with glucocorticoids induces osteoporosis and that vascular calcification is often associated with osteoporosis, it is suggested that Dex may develop and exacerbate vascular calcification. Further studies are necessary to clarify the long term effect of glucocorticoid administration on the development of vascular calcification, especially calcified atherosclerotic plaque lesions.
References
- Polonsky TS, McClelland RL, Jorgensen NW, Bild DE, Burke GL, et al. (2010) Coronary artery calcium score and risk classification for coronary heart disease prediction. JAMA 303: 1610-1616.
- Arad Y, Goodman KJ, Roth M, Newstein D, Guerci AD (2005) Coronary calcification, coronary disease risk factors, C‐reactive protein, and atherosclerotic cardiovascular disease events: the St. Francis Heart Study. J Am Coll Cardiol 46: 158-165.
- Shafi S, Ansari HR, Bahitham W, Aouabdi S (2019) The Impact of Natural Antioxidants on the Regenerative Potential of Vascular Cells. Front Cardiovascu Med 6: 1-28.
- Ala-Korpela M (2019) The culprit is the carrier, not the loads: cholesterol, triglycerides and Apo lipoprotein B in atherosclerosis and coronary heart disease. Int J Epidemiol 48: 1389-1392.
- Esper RJ, Nordaby RA (2019) Cardiovascular events, diabetes and guidelines: the virtue of simplicity. Cardiovasc Diabetol 18: 42.
- Wityk RJ, Lehman D, Klag M, Coresh J, Ahn H, et al. (1996) Race and sex differences in the distribution of cerebral atherosclerosis.Stroke 27: 1974-1980.
- Qureshi AI, Caplan LR (2014) Intracranial atherosclerosis. Lancet 383: 984-998.
- Choi YJ, Jung SC, Lee DH (2015) Vessel Wall imaging of the intracranial and cervical carotid arteries. J Stroke 17: 238-255.
- Williams KJ, Tabas I (1995) The response-to-retention hypothesis of early atherogenesis. Arteriosder Thromb Vasc Biol 15: 551-561.
- Hansson GK (2005) Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med 352: 1685-1695.
- Klopper A (2021) Delayed global warming could reduce human exposure to cyclones. Nature 98: 35.
- Dichgans M, Pulit SL, Rosand J (2019) Stroke genetics: discovery, biology, and clinical applications. Lancet Neurol 18: 587-599.
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Citation: Mori K, Shioi A, Jono S, Nishizawa Y, Morii H (2022) Vascular Calcification by Promoting Osteoblastic Differentiation of Vascular Smooth Muscle Cells. Atheroscler Open Access 7: 185.
Copyright: © 2022 Mori K, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Share This Article
Open Access Journals
Article Usage
- Total views: 1843
- [From(publication date): 0-2022 - Mar 31, 2025]
- Breakdown by view type
- HTML page views: 1505
- PDF downloads: 338