The Influence of Foods and Beverages on Polyethylene Glycol Marker Detection in Urine
Received: 26-Dec-2018 / Accepted Date: 24-Jan-2019 / Published Date: 31-Jan-2019 DOI: 10.4172/2155-6105.1000376
Abstract
Purpose: In the fight against drug abuse, Polyethylene Glycol (PEG) markers have become an established method to prevent false negative results by means of donor urine or own clean urine that has been stored beforehand. Until now, little is known about the influence of meal composition, time interval between meal and PEG marker ingestion, and acute urine dilution on PEG marker detection.
Aims: In the present study we investigated the detectability of monodisperse PEG markers under field conditions with variable meal compositions, caloric intakes, and drink behaviors.
Methodology: 448 subjects (272 females and 176 males) took part in the study. Six study centers recruited subjects for 2 runs differing in the time interval between capsule marker intake and urine sampling (40 and 60 mins). Two different meal compositions (either carbohydrate or fat-protein rich) and three different delays between meal completion and marker intake were tested (30, 60 and 120 min.). Food and beverage intake could be made ad libitum but were protocolled. Capsules containing either a PEG 8/PEG 10 or a PEG8/PEG 12 combination with 150 mg, each served as PEG markers. PEG concentrations from PEG 8 to PEG 12 were determined by liquid chromatography mass spectrometry. Individual thresholds for positive PEG detection were used by means of the concentrations of non-administered PEGs.
Main results: Both the caloric and the fluid intake significantly influenced PEG marker concentrations in urine. The 40 mins resorption phase led to a detection rate between 81% and 100%. All subjects were tested positive for PEG markers following the 60 mins resorption phase.
Conclusion: The marker detection following a PEG marker containing capsule is a valid method, when individual thresholds and a marker resorption phase of 60 mins are taken into account. That holds true independently of the time interval between meal and marker intake, meal composition, and beverage volume.
Keywords: Urine; PEG; food; Beverage; Creatinine; Drug abuse; Doping
Introduction
Drug abuse tests are a common feature of many areas such as probation service, addiction treatment, road traffic safety, workplace testing, and doping testing in sports [1-7], To this end, the detection of previous drug exposure can be performed, in principle, in blood, saliva, sweat, hairs, and urine [8-10]. However, the check for drug or doping abuse is predominantly performed in urine, mainly due the long experience, the well-known windows of detection and its simple and non-invasive collection [8]. Unfortunately, urine sample manipulation is a serious problem in clients who have an interest in false negative results. There are primarily two ways to achieve false negative results, namely dilution and substitution [11-14]. The first can be realized either by drinking excessive amounts of water, taking diuretics or adding water to the urine sample. For the second, stored own and clean urine, donor or synthetic urine are used. Even the internet market provides users, for example with a so called “Whizzinator” kit [15] containing a false penis, clean urine, its storage bag, and a heater pack to keep the urine at body temperature.
During recent years, tagging of urine with orally ingested low molecular weight Polyethylene Glycols (PEG) prior to urine sample collection has become an established method to prevent false negative results [16-20]. In this case, under supervised conditions the client swallows a defined PEG combination and, after a latency period, the client urinates without direct visual control. This method in combination with a sophisticated analytical procedure impedes false positive and negative results [20]. Simultaneously, it prevents the mental stress that many clients and supervisors feel when directly exposed to direct observation during urination [21-23]. Moreover, supervision can even lead to the clinical disorder of paruresis [23].
In order to exclude a false negative result with respect to PEGdetection, the critical time interval between oral PEG intake and urination has to be known. In a recent investigation it could be shown that 40 mins after an oral ingestion of a PEG containing capsule, the urine PEG concentrations were significantly elevated [19]. Using a time interval of 60 mins, the urine of all subjects could be positively tested for the PEG marker [19]. However, that study was performed with a limited number of subjects and without any control of subject´s eating and drinking behavior prior to PEG ingestion. The influence of a preceding meal cannot be neglected since it is well known that the ingestion of food and beverages evokes gastrointestinal effects such as changes in gastric retention phase and absorption within the small intestine, which probably may influence the rate and extent of absorption of any orally administered substance and, therefore, indirectly the PEG-marker concentration in urine. Compared to an isocaloric high-carbohydrate soup, a high-fat soup slowed gastric emptying [24]. After a high fat meal, the peak plasma concentration of the proteasome inhibitor Ixazomib was delayed by about 3 hours and a 69% reduction in peak plasma concentration [25]. Similar results could be obtained for the pharmacological chaperone Johnson et al. [26]. Aside from the kind and composition of macronutrients, meal volume [27], the total energy content [28], and timing of meal in relation of timing of a drug/marker administration [27] may play a role.
In order to determine the effects of meal composition, the time between meal completion and PEG-marker administration, and the time-interval between PEG-marker intake and urine sampling, the present study was conducted with a representative number of subjects aiming in the urinary detection rate of PEG-markers as the primary outcome. The influences of composition of an acute meal and acute urine flow on PEG concentrations served as secondary outcomes.
Methods
General information
The present study followed all the relevant national regulations and the tenets of the Declaration of Helsinki and was approved by the ethical committee of the University of Duisburg-Essen, Germany (Number 16-7266-BO).
The multi-center prospective study consisted of six local centers. Each center conducted two separated runs with the identical food administration (Chapter 2.3) but different time intervals between PEG-marker intake and urination (40 min vs. 60 min) and different subjects. Prior to the empirical part, all staffs were thoroughly informed about the implementation and goals of the study. Each center was requested to include at least 30 and at most 50 subjects per run.
Subjects
Participants were recruited by local announcements. The inclusion criteria were older than 18 years and the written informed consent after oral and written information about the course of the study. The only exclusion criterion was pregnancy. In total, 448 subjects (272 females and 176 males) took part in the study. Their anthropometric data were as follows: age 31+12.3 years, height 173+13.2 cm, and weight 74.0+16.6 kg.
Schedule
Subjects entered the centre in the morning after an overnight fasting and emptied the bladder. From this urine a 10 mL sample was taken for baseline analysis of PEG and creatinine concentrations. Thereafter, either a Carbohydrate-rich (CM) or a Fat-protein-rich Meal (FM) was offered. CM was based on a breakfast and consisted of rolls with various toppings. All subjects were allowed to eat ad libitum but had to log the number of portions.
Calculation of energy content of food and beverage
The calculation of the energy content of the food was based on the composition of the macromolecules and the weight of one serving. A single roll combined with its toppings was energetically composed to a content of about 1000 KJ. FM consisted of a lunch, where subjects could choose between defined portions of gratin, ragout or a combination thereof. Each portion was also equivalent to about 1000 KJ. In addition, subjects were allowed to drink ad libitum water, coca, soft drinks, or fruit juice. With the exception of water, each drink (200 mL) was calculated with 320 KJ based on the carbohydrate content.
Study-groups
For both time intervals between PEG-intake and urine sampling (40 and 60 min), six groups were investigated differing in meal composition (FM vs. CM) and/or time interval between completion of the meal and PEG-intake (30, 60 and 120 min). Figure 1 shows the study groups and the number of subjects in each group.
Analytical procedures
Marker composition
The marker capsule consisted of two times 150 mg polyethyleneglycol. Two marker combinations were applied containing either PEG 8+PEG 10 or PEG 8+PEG 12. The molecular weights account to 370 g/mol (PEG 8), 458 g/mol (PEG 10), and 546 g/mol (PEG 12). Each marker capsule was delivered in a small glass container. The peelable barcode contained the marker combination (Figure 2).
Analysis of PEG concentrations
The analyst was blinded for the marker composition. Aside from PEG 8, PEG 10, and PEG 12 the polyethylene glycols PEG 9 (414 g/ mol) and PEG 11 (502 g/mol) were also detected. The analytical procedure to determine PEG concentrations have been described in detail previously (LIT). In brief: PEG concentrations in urine were analyzed on a Shimadzu LCMS 8050 with a LC Nexera 2 from Shimadzu and computed by means of the LabSolutions software from Shimadzu. The PEGs used as standards were provided by CSChromatography (Germany) and the internal standard Dimethoxytetraethyleneglycol by Sigma-Aldrich (Germany). All other solvents and chemicals were of LCMS-grade purchased from Roth (Germany).
Detection threshold of PEG markers
The detection threshold of PEG markers (DTPEG) were set individually. To this end, the concentrations of the non-administered PEGs (PEGna; either PEGs 9, 10, 11 or PEGs 9, 11, 12) were calculated from the urine after marker intake according to the formula:
DTPEG=mean PEGna+3×standard deviation PEGna.
A sample was regarded as positive, if the urine concentrations of both markers were above threshold. A borderline positive result corresponded to one marker concentration above threshold.
Analysis of creatinine concentration
Creatinine concentrations in urine (CREA) were analysed on an ADVIA 1800 analyser. The CREA method is based on the reaction of picric acid with creatinine in an alkaline environment as described in the original procedure according to Jaffe. Prior to the start of analysis, samples were centrifugated at 4°C for 15 min at 4000 RPM. For calibration a Siemens Healthcare Diagnostics Chemistry Calibrator (REF 09784096) was used.
Statistics
For the data description and evaluation, Microsoft Excel 15 (Microsoft Corporation, Redmond, USA) and IBM SPSS Statistics 24 (IBM Corporation Amonk, USA) were used. If not otherwise stated, mean+standard deviation are given in text and tables, mean+standard error are presented in figures.
A two-factor variance analysis (factors “composition of meal” and “delay”) was applied to the PEG 8 and PEG 10 or PEG 12 marker concentrations. In case of significant effects of the factors multiple comparisons of means were tested by a Sidak.
The influence of acute urine flow (measured as quotient of CREA before and CREA after) and total caloric intake on PEG 8 and PEG 10 or PEG 12 marker concentrations was tested by multiple linear regression. The regression coefficient was tested by a t-test.
A p value of <0.05 was considered significant.
Results
PEG concentrations
Prior to marker ingestion the basal mean PEG concentrations were in the range between 0.35+0.39 μg/mL and 1.23+1.34 μg/mL with no detectable influence of PEG molecular weight. Figure 3 presents the (PEG 8) and the concentrations of the second marker, respectively. Except for group 1, the 60 min resorption phase led to significantly higher PEG concentrations in urine. Neither a significant influence of meal composition nor of the time interval between finishing of meal and PEG capsule intake on PEG concentrations could be detected. The correlation between PEG 8 and the second marker concentration was very high with a Pearson R of 0.97, a slope of 1,004 and an intersection of -8.0 (Figure 4).
Although the inter-individual comparison of baseline concentrations of PEG yielded huge differences ranging from 0.00 μg/mL to 2.42 μg/mL ([PEG 8] 0.07-2.42 μg/mL; [PEG 9] 0.05-2.27 μg/mL; [PEG 10] 0.00-2.40 μg/mL; [PEG 11] 0.00-2.33 μg/mL; [PEG 12] 0.03-2.02 μg/mL), the intra-individual concentrations of PEG 8 to PEG 12 were in comparable ranges. After marker administration, the two marker concentrations significantly increased in all groups.
Influence of caloric intake and urine flow on marker concentrations
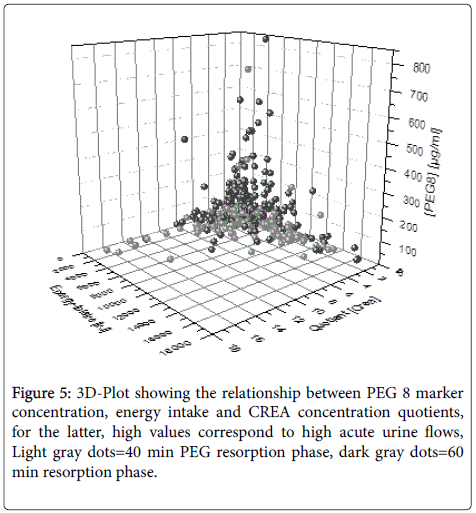
The caloric intake ranged between 0 KJ and 16.200 KJ, the quotient between basal CREA and CREA after marker ingestion ranged from 0.09 to 16.21. The multiple linear regression analysis yielded significant influences of both caloric intake and urine flow on marker concentrations for the 60 min but not for the 40 min resorption phase. Figure 5 depicts the combined influence of both parameters with highest marker concentration for low caloric intake and low urine flow values.
Detection threshold and detection rate for PEG marker concentrations
The individual detection thresholds for PEG marker had a mean of 4.3+10.3 μg/mL with a huge range from 0.1 μg/mL to 121.4 μg/mL. For the 40 min. resorption phase, the detection rate ranged from 81% to 100% within groups with a tendency of higher values with increasing time interval between finishing of meal and marker intake. However, the 60 min. resorption interval led to a 100% detection rate in all groups (Table 1).
| Group | Total | Detected | Borderline | Not detected | |||
|---|---|---|---|---|---|---|---|
| Number | Absolute | Percent | Absolute | Percent | Absolute | Percent | |
| G1-40 | 46 | 40 | 87 | 1 | 2 | 5 | 11 |
| G2-40 | 31 | 25 | 81 | 4 | 13 | 2 | 6 |
| G3-40 | 33 | 31 | 94 | 2 | 6 | 0 | 0 |
| G4-40 | 40 | 33 | 83 | 4 | 10 | 3 | 7 |
| G5-40 | 42 | 42 | 100 | 0 | 0 | 0 | 0 |
| G6-40 | 39 | 37 | 95 | 0 | 0 | 2 | 5 |
| G1-60 | 36 | 36 | 100 | 0 | 0 | 0 | 0 |
| G2-60 | 39 | 39 | 100 | 0 | 0 | 0 | 0 |
| G3-60 | 36 | 36 | 100 | 0 | 0 | 0 | 0 |
| G4-60 | 35 | 35 | 100 | 0 | 0 | 0 | 0 |
| G5-60 | 38 | 38 | 100 | 0 | 0 | 0 | 0 |
| G6-60 | 34 | 34 | 100 | 0 | 0 | 0 | 0 |
Table 1: Detection rates of PEG marker concentration after 40 min, (upper block) and 60 min resorption phase (bottom block).
Discussion
The main finding of the present study is that when using PEGmarker containing capsules, PEG detection in urine is valid when a time interval of 60 min. between marker intake and urine sampling is taken into account. A hundred percent detection holds for all conditions tested, i.e. low and high urinary flows, a huge range of caloric intake prior to marker intake, different time intervals between meal consumption and marker intake, and the meal composition.
The present data are in line with a recent kinetic study of PEG marker [19], where a nonlinear, reciprocal correlation between urine flow and PEG concentrations was obtained. In that study, 30 min. after oral marker intake the PEG concentrations in urine already reached statistically significant increases but it took 60 mins to get elevated concentrations in all subjects. Our data also confirm the results of Jones et al. [17] who found a 10% detection gap of PEG markers in urine for an up to 40 mins resorption phase. Since the oral intake of PEG containing capsules and PEG markers in a fluid form may lead to different gastro-intestinal resorption delays, the present results are not actually comparable to the findings of Huppertz et al. [29], who proposed a minimum resorption phase of 30 mins for liquid PEGmarkers.
In the present investigation, highest marker concentrations were obtained in subjects with low energy and fluid intake. The latter was confirmed by the quotient of creatinine concentrations before and after PEG administration. In contrast to the statistically significance of the combined influence of energy intake and fluid ingestion on PEG marker concentration, we did not find any significant impact of meal composition or time interval between meal completion and oral marker intake on marker concentration. However, none of these parameters was able to depress marker concentration below the individual detection threshold 60 min. after marker intake.
Already the baseline urine prior to marker administration contained PEG in all subjects. That can be explained with the fact that PEG is applied in everyday life such as a galenic media in pharmaceutical and cosmetic products. Moreover, the US Food & Drug Administration allows PEGs with a molecular weight between 200 and 9500 g/mol as a binder and lubricant in tabs used for food as well as an adjuvant in non-nutritive sweeteners, vitamin and mineral preparations. Quantitative different expositions to PEG in everyday life may have led to the obtained huge inter-individual differences of basal PEG concentrations. However, the intra-individual basal PEG concentrations of all analyzed PEG specimen (PEG 8 to PEG 12) were in a similar range. Therefore, an individual threshold after PEG administration as proposed in a former [19] and validated in the present study appears to be independent from the basal concentrations as well as from the influences of foods and beverages.
In an opioid replacement therapy setting, clients regarded the regular external urine control mainly as a supporting tool to stay away from drugs but the sight control itself was considered horrible, humiliating, and demeaning [21]. Some patients reported that memories of earlier misuse become present. In a recent study, Baum et al. [30] evaluated that the supervision procedure during urine sampling does not only affect clients but burdens both clients and supervisors. In the same setting, the introduction of PEG markers without sight control improved drug screenings at the psychological level as well as from safety and economical points of view. For safety reasons, the concrete PEG marker composition must not be anticipated by the client. To this end, multiple combinations of PEG markers are needed. In the present study we used combinations of two markers. Of course, combinations of other PEG specimen, a single marker, or a multiple combination can be selected.
Conclusion
In the field of drug and doping abuse detection, the use of PEG markers is a valid method when individual thresholds and a 60 mins resorption phase are taken into account. It makes the use of donor urine of any source impossible and prevents psychological stress originating from sight control during sample collection.
References
- Guide for physicians on substitution-based treatment for opiate addicts (2018) German.
- Zurhold H (2013) Medical rehabilitation of drug patients according to § 35 BtMG ("Therapy instead of punishment")-effectiveness and trends: Wirksamkeit und Trends. Abschlussbericht. German.
- Kermani EJ, Castaneda R (2006) Psychoactive substance use in forensic psychiatry. Am J Drug Alcohol Abuse 22:1-27.
- US Department of Transportation (2018) Urine specimen collection guidelines for the workplace drug testing programs (49CFR Part 40).
- Department of Health and Human Services (2017) Urine specimen collection handbook for federal agency workplace drug testing programs.
- International Standards-Testing and Investigation (2017) World anti-doping code.
- Melanson SEF, Clarke WA, Fragoza K, Gilligan C, Holdren R, et al. (2018) Debates in pain management testing. Clin Chem 64:769-776.
- Allen KR (2011) Screening for drugs of abuse: Which matrix, oral fluid or urine? Ann Clin Biochem 48: 531-541.
- Gryczynski J, Schwartz RP, Mitchell SG, O'Grady KE, Ondersma SJ (2014) Hair drug testing results and self-reported drug use among primary care patients with moderate-risk illicit drug use. Drug Alcohol Depend 14: 44-50.
- Lafolie P, Beck O, Blennow G, Boreus L, Borg S, et al. (1991) Importance of cretinine analysis of urine when screening for abused drugs. Clin Chem 37: 1927-1931.
- Kapur B, Hershkop S, Koren G, Gaughan V (1999) Urine fingerprinting: detection of sample tampering in an opiate dependency program. Ther Drug Monit 21: 243-250.
- Thevis M, Geyer H, Mareck U, Sigmund G, Henke J, et al. (2007) Detection of manipulation in doping control urine sample collection: A multidisciplinary approach to determine identical urine samples. Anal Bioanal Chem 388: 1539-1543.
- Accurso AJ, Lee JD, McNeely J (2017) High prevalence of urine tampering in an office-based opioid treatment practice detected by evaluating the norbuprenorphine to buprenorphine ratio. J Subst Abuse Treat 83: 62-67.
- Gauchel G, Huppertz B, Feiertag H, Keller R (2003) Clinical use of polyethylene glycols as marker substances and determination in urine by liquid chromatography. J Chromatogr B Analyt Technol Biomed Life Sci 787: 271-279.
- Jones JD, Atchinson JJ, Madera G, Metz VE (2015) Need and utility of a polyethylene glycol marker to ensure against urine falsification among heroin users. Drug Alcohol Depend 153: 201-206.
- Christensen K (2014) Shifting the paradigm. A blueprint for value recognition and process improvement in the US drug testing market.
- Baum K, Einwächter S, Bibl M, Huppertz B (2017) Urine kinetics of low molecular polyethylene glycols following an oral capsule ingestion. J Appl Life Sci Int 15: 1-6.
- Huppertz B, Bartling C, Baum K (2018) Adulteration of urine samples, discovery, and mitigation. J Appl Life Sci Int 16: 1-8.
- Monwell B, Bülow P, Johnson B (2018) The pros and cons of urine tests in opioid maintenance treatment: A study of patient´s experiences. Heroin Addict Clin Probl 20: 5-15.
- Strahler K, Elbe AM (2007) Want-but cannot. The problem of doping control. Leistungssport 4: 35-38.
- Hammelstein P (2002) For the Practioner - From practice for practice. Verhaltenstherapie 12: 224-227.
- Cecil JE, Francis J, Read NW (1999) Comparison of the effects of a high-fat and high-carbohydrate soup delivered orally and intragastrically on gastric emptying, appetite and eating behaviour. Physiol Behav 67: 299-306.
- Gupta N, Hanley MJ, Venkatakrishnan K, Wang B, Sharma S, et al. (2016) The effect of a high-fat meal on the pharmacokinetics of Ixazomib, an oral proteasome inhibitor, in patients with advanced solid tumors or lymphoma. J Clin Pharmacol 56: 1288-1295.
- Johnson FK, Mudd PN, Janmohamed SG (2015) Relative bioavailability and the effect of meal type and timing on the pharmacokinetics of migalastat in healthy volunteers. Clin Pharmacol Drug Dev 4: 193-202.
- Singh BN (1999) Effects of food on clinical pharmacokinetics. Clin Pharmacokinet 37: 213-255.
- Sutton SC, Nause R, Gandelman K (2017) The impact of gastric pH, volume, and emptying on the food effect of ziprasidone oral absorption. AAPS J 19: 1084-1090.
- Huppertz B, Gauchel G, Feiertag H, Schweizer H, Krieger H, et al. (2004) Urine labeling with orally applied marker substances in drug substitution therapy. Clin Chem Lab Med 42: 621-626.
- Baum K, Schüler Springorum M, Huppertz B (2018) Process reliability and psychological stress in urine sample collection for drug testing: A pilot study. J Appl Life Sci Int 19: 1-12.
Citation: Einwächter S, Huppertzb B, Bibl M, Baum K (2019) The Influence of Foods and Beverages on Polyethylene Glycol Marker Detection in Urine. J Addict Res Ther 10:376. DOI: 10.4172/2155-6105.1000376
Copyright: © 2019 Einwächter S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 3787
- [From(publication date): 0-2019 - Feb 22, 2025]
- Breakdown by view type
- HTML page views: 3132
- PDF downloads: 655