Review Article Open Access
Interaction between Amyloid Beta Toxicity and the PI3K Pathway in Alzheimer's Disease
Hojin Choi and Seong-Ho Koh*
Department of Neurology, Hanyang University Guri Hospital, Korea
- *Corresponding Author:
- Seong-Ho Koh
Department of Neurology, Hanyang University Guri Hospital
153, Gyeongchun-ro, Guri-si
Gyeonggi-do, 11923 Korea
Tel: +82-31-560-2260
E-mail: ksh213@hanyang.ac.kr
Received date: September 20, 2016; Accepted date: October 05, 2016; Published date: October 12, 2016
Citation: Choi H, Koh SH (2016) Interaction between Amyloid Beta Toxicity and the PI3K Pathway in Alzheimer’s Disease. J Alzheimers Dis Parkinsonism 6:269. doi:10.4172/2161-0460.1000269
Copyright: © 2016 Choi H, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Alzheimers Disease & Parkinsonism
Abstract
Alzheimer’s disease (AD) is the most common disease causing dementia. Amyloid beta (Aβ) is well known to play the most important roles in the pathogenesis. Aβ induces neuronal cell death and neuroinflammation. It has been published that Aβ affects numerous intracellular signaling pathways. The phosphatidylinositol-3-kinase (PI3K) pathway associated with neuronal cell survival and various neural stem cell functions. In this review, the interaction between Aβ and the PI3K pathway will be discussed.
Keywords
Alzheimer’s disease; Amyloid beta; PI3K; Neurons; Neural stem cells
Abbreviations
PI3K: Phosphatidylinositol 3-kinase; GSK-3β: Glycogen synthase kinas-3β; VEGF: Vascular endothelial growth factor; GS: Glycogen synthase; CoQ10: Coenzyme Q10; â�?�?: Phosphorylated form; Ser: Serine
Introduction
Alzheimer’s disease (AD) is a chronic neurodegenerative disease that is the cause of 60% to 70% of cases of dementia. The most common early symptom is difficulty in remembering recent events. As the disease advances, symptoms can include problems with language, disorientation (including easily getting lost), mood swings, loss of motivation, not managing self-care, and behavioral issues. Though AD has been studied for over several decades, there are still crucial problems in its diagnosis, prevention and treatment.
When it comes to pathogenesis, however, amyloid beta (Aβ) has been considered as a main factor: namely, Aβ monomers, Aβoligomers, and Aβ fibrils play very important roles in the pathogenesis of AD [1]. Once Aβ monomers start to form oligomers in the brain, astrocytes and microglial cells become activated and a lot of inflammatory cytokines are released by Aβ oligomers. Subsequently, abnormal protein-folding activities, mitochondrial dysfunction, and neuronal cell death are followed. As the result, neurodegenerative change of AD is started in the brain [2].
The Amyloid Cascade Hypothesis and its Weakness in AD Patients
The amyloid cascade hypothesis is still one of the most reliable pathophysiological processes of AD until now. It is well known that Aβ oligomers provoke oxidative stress and cytotoxicity, so there is growing interest in using antioxidants to treat this disorder. To be more detailed, Aβ produces hydrogen peroxide (H2O2) through metal ion reduction, with expression of thiobarbituric acid-reactive substances, and this mechanism is probably caused by hydroxyl radicals as well as nitro-oxidative stress [3- 5]. These processes are well known to induce the damage of neuronal cells and neural stem cells. Therefore, it is clear that some of the cytotoxicity of Aβ oligomers is associated with oxidative stress.
In the basis of this amyloid cascade hypothesis, numerous clinical trials for AD using anti-amyloid treatment have been performed, but all of the anti-amyloid treatment that reached Phase III clinical trials has failed even though some of them effectively clear Ab deposits [3]. These failures have raised questions about the role of Aβ and anti-amyloid treatment in AD, and then, we might have to consider other pathophysiological processes of AD and other strategies for its treatment.
The PI3K Pathway in Neurons or Neural Stem Cells
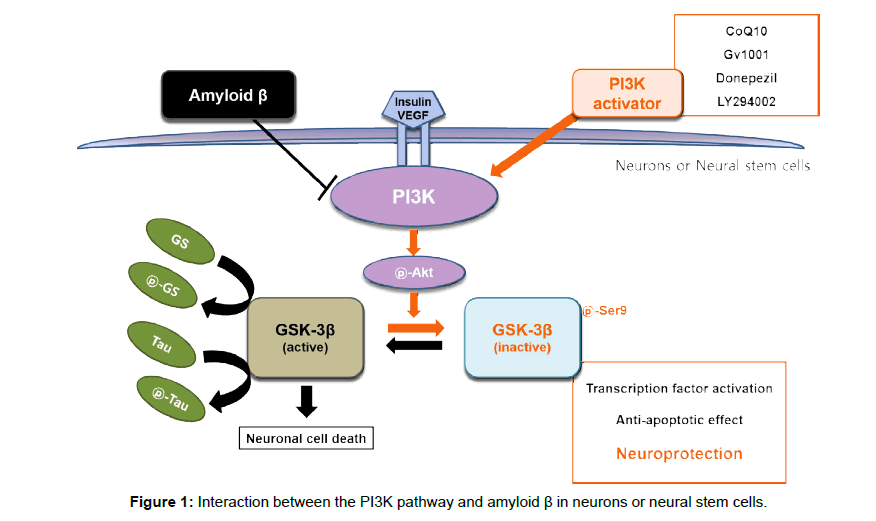
The phosphatidylinositol 3-kinase (PI3K) pathway is known to be important in neuronal cell survival. It is the major intracellular signal pathway responsible for the transmission of anti-apoptotic genes and the control of cell survival, and it is activated by some neuroprotective signals [6,7]. This pathway also plays a key role in control of oxidative stress and adult neurogenesis [8] and a pivotal role in control of amyloid metabolism and tau phosphorylation in AD brains [9-12]. Describing in detail of the PI3K pathway, various neuroprotective signals activate PI3K, and then activated PI3K phosphorylates its downstream target, Akt/protein kinase B. Phosphorylated Akt (pAkt) directly affects Bcl- 2 associated death promoter (BAD)/Bcl-2, caspase 9, IkB kinase, and Forkhead-related transcription factor. pAkt also inhibits GSK-3β by phosphorylating it at Ser9. Glycogen synthase kinas-3β (GSK-3 β) involves in glycogen metabolism, regulates the function of metabolic, structural, and signaling proteins, such as activator protein-1, cyclic AMP response element binding protein, nuclear factor of activated T cells, heat shock factor-1, and β-catenin. Activation of GSK-3β, which activates the mitochondrial death pathway, increases the release of cytochrome c from mitochondria. Released cytochrome c activates caspase-9 and caspase-3, and cleaves PARP, resulting in apoptosis [13,14]. Thus, inactivation of the phosphorylated GSK-3β by pAkt is important for neuronal cell survival. In addition, it was reported that the PI3K pathway is essential for the self-renewal of embryonic stem cells (ESCs) and activation of this pathway is significant for maintaining pluripotency in ESCs [15]. Several studies showed that the phosphatase and tensin homolog deleted on chromosome 10 (PTEN), an antagonist of PI3K, negatively regulates NSCs proliferation, survival, and selfrenewal both in vivo and in vitro. Contrary to PTEN, components of the PI3K pathway were reported to be involved in the self-renewal of NSCs [16,17] (Figure 1).
Interaction between the PI3K Pathway and Amyloid β in Neurons or Neural Stem Cells
We have published that Aβ induces neurotoxicity by inhibiting the PI3K pathway in neuronal cells or neural stem cells and a direct PI3K activator has neuroprotective effects through activation of the PI3K pathway in Aβ-induced neuronal cell death and we also confirmed that several neuro-protectants protect those cells against Aβ-induced injury by activating the PI3K pathway. For example, it was found in our previous study that Coenzyme Q10, Donepezil, GV1001, and LY294002 (a PI3K activator) protect against amyloid beta-induced neuronal cell death by activating the P13K pathway [18-21]. In the other study, we also confirmed that Coenzyme Q10 restores Aβ-inhibited proliferation of neural stem cells through the activation of the pathway [22]. Other previous studies have shown that abnormal activities of the PI3K pathway are found in people at risk of developing AD. These abnormal activities are known to be associated with an increased production of Aβ and a decreased ability to clear Aβ, together resulting in excessive Aβ. Moreover, Aβ has been also described to affect the activities of the pathway: Aβ oligomers activate GSK-3β blocking the activities of the pathway and increasing hyperphosphorylation of tau directly associated with cognitive decline [23-25].
Conclusion
In conclusion, because AD is a multifactorial disease and lots of pathogenic mechanisms are involved in AD, we need to investigate the molecular pathways associated with AD in much more detail. Based on the results, we should develop the new approaches for adjuvant therapies. One more important thing is that new therapies have to be started from the presymptomatic or early AD state considering that all the clinical trials targeting full-blown or advanced AD have been failed. Taken together of our previous results, activation of the PI3K pathway can be one of new therapeutic strategies for AD. Thus, how to activate the PI3K pathway without any side effects should be developed for the treatment of AD patients and then its activation has to be used for AD patients in the presymptomatic or early state.
Acknowledgement
This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (2015R1A2A2A04004865).
References
- Shim YS, Morris JC (2011) Biomarkers predicting Alzheimer’s disease in cognitively normal aging. J ClinNeurol 7:60-68.
- Karran E, Mercken M, De Strooper B (2011) The amyloid cascade hypothesis for Alzheimer’s disease: an appraisal for the development of therapeutics. Nat Rev Drug Discov 10:698-712.
- Godyn J,Jonczyk J, Panek D, Malawska B (2016) Therapeutic strategies for Alzheimer's disease in clinical trials. Pharmacol Rep 68:127-138.
- Butterfield DA (2002) Amyloid beta-peptide(1-42)-induced oxidative stress and neurotoxicity: implications for neurodegeneration in Alzheimer's disease brain. A review. Free Radic Res 36:1307-1313.
- Cenini G, Cecchi C, Pensalfini A, Bonini SA, Ferrari-Toninelli G, et al. (2010) Generation of reactive oxygen species by beta amyloid fibrils and oligomers involves different intra/extracellular pathways. Amino Acids 38:1101-1116.
- Koh SH, Lo EH (2015) Therole of the PI3K pathway in the regeneration of the damaged brain by neural stem cells after cerebral infarction.J ClinNeurol 11:297-304.
- Cantley LC (2002) Thephosphoinositide 3-kinase pathway. Science 296: 1655-1657.
- Varela-Nallar L, Aranguiz FC, Abbott AC, Slater PG, Inestrosa NC (2010) Adulthippocampalneurogenesis in aging and Alzheimer’s disease. Birth Defects Res C Embryo Today 90: 284-296.
- Kirschenbaum F, SC Hsu, B Cordell, JV McCarthy (2001) Glycogen synthase kinase 3-beta regulates presenilin 1 C-terminal fragment levels. J BiolChem 276: 30701-30707.
- Mudher A, Chapman S, Richardson J, Asuni A, Gibb G, et al. (2001) Disheveled regulated the metabolism of amyloid precursor protein via protein kinase C/mitogen-activated protein kinase and c-Jun terminal kinase. J Neurosci 21: 4987-4995.
- Sang H, Z Lu, Y Li, B Ru, W Wang, et al. (2001) Phosphorylation of tau by glycogen synthase kinase 3beta in intact mammalian cells in�?uence the stability of microtubules. NeurosciLett 312: 141-144.
- Varela-Nallar L, Aranguiz FC, Abbott AC, Slater PG, Inestrosa NC (2010) Adult hippocampal neurogenesis in aging and Alzheimer’s disease. Birth Defects Res C Embryo Today 90: 284-296.
- 13.Frame S, P Cohen, RM Biondi (2001) A common phosphate binding site explains the unique substrate speciï¬city of GSK3 and its inactivation by phosphorylation. Mol Cell 7: 1321-1327.
- Frame S, P Cohen, RM Biondi (2001) A common phosphate binding site explains the unique substrate speciï¬city of GSK3 and its inactivation by phosphorylation. Mol Cell 7: 1321-1327.
- Pap M, GM Cooper (2002) Role of translation initiation factor 2B in control of cell survival by the phosphatidylinositol 3-kinase/Akt/glycogen synthase kinase 3beta signaling pathway. Mol Cell Biol 22: 578-586.
- Watanabe S, Umehara H, Murayama K, Okabe M, Kimura T, et al. (2006) Activation of Akt signaling is sufï¬cient to maintain pluripotency in mouse and primate embryonic stem cells. Oncogene 25: 2697-2707.
- Groszer M, Erickson R, Scripture-Adams DD, Lesche R, Trumpp A, et al. (2001) Negative regulation of neural stem / progenitor cell proliferation by the Pten tumor suppressor gene in vivo. Science 294: 2186-2189.
- Li J, Simpson L, Takahashi M, Miliaresis C, Myers MP, et al. (1998) The PTEN/MMAC1 tumor suppressor induces cell death that is rescued by the Akt/protein kinase B oncogene. Cancer Res 58: 5667-5672.
- Lee KY, SH Koh, MY Noh, SH Kim,YJ Lee (2008) Phosphatidylinositol-3-kinase activation blocks amyloid beta-induced neurotoxicity. Toxicology 243: 43-50.
- Noh MY, SH Koh, Y Kim, HY Kim, GW Cho, et al. (2009) Neuroprotective effects of donepezil through inhibition of GSK-3 activity in amyloid-beta-induced neuronal cell death. J Neurochem 108: 1116-1125.
- Choi H, Park HH, Koh SH, Choi NY, Yu HJ, et al. (2012) Coenzyme Q10 protects against amyloid beta-induced neuronal cell death by inhibiting oxidative stress and activating the P13K pathway. Neurotoxicology 33:85-90.
- Park HH, Lee KY, Kim S, Lee JW, Choi NY, et al. (2014) Novel vaccine peptide GV1001 effectively blocks β-amyloid toxicity by mimicking the extra-telomeric functions of human telomerase reverse transcriptase. Neurobiol Aging 35:1255-1274.
- Choi H, Park HH, Lee KY, Choi NY, Yu HJ, et al. (2013) Coenzyme Q10 restores amyloid beta-inhibited proliferation of neural stem cells by activating the PI3K pathway. Stem Cells Dev 22:2112-2120.
- O'Neill C, Kiely AP, Coakley MF, Manning S, Long-Smith CM (2012) Insulin and IGF-1 signalling: longevity, protein homoeostasis and Alzheimer's disease.BiochemSoc Trans 40:721-727.
- O' Neill C (2013) PI3-kinase/Akt/mTOR signaling: impaired on/off switches in aging, cognitive decline and Alzheimer's disease.ExpGerontol 48:647-653.
Relevant Topics
- Advanced Parkinson Treatment
- Advances in Alzheimers Therapy
- Alzheimers Medicine
- Alzheimers Products & Market Analysis
- Alzheimers Symptoms
- Degenerative Disorders
- Diagnostic Alzheimer
- Parkinson
- Parkinsonism Diagnosis
- Parkinsonism Gene Therapy
- Parkinsonism Stages and Treatment
- Stem cell Treatment Parkinson
Recommended Journals
Article Tools
Article Usage
- Total views: 13771
- [From(publication date):
October-2016 - Apr 03, 2025] - Breakdown by view type
- HTML page views : 12715
- PDF downloads : 1056