Make the best use of Scientific Research and information from our 700+ peer reviewed, Open Access Journals that operates with the help of 50,000+ Editorial Board Members and esteemed reviewers and 1000+ Scientific associations in Medical, Clinical, Pharmaceutical, Engineering, Technology and Management Fields.
Meet Inspiring Speakers and Experts at our 3000+ Global Conferenceseries Events with over 600+ Conferences, 1200+ Symposiums and 1200+ Workshops on Medical, Pharma, Engineering, Science, Technology and Business
Editorial Open Access
GMO Foods: Regulatory Mechanism and Challenges in India
| Sheelendra BM* | |
| Assistant Professor, Lovely Professional University, 144401, Punjab | |
| Corresponding Author : | Sheelendra M.Bhatt Assistant Professor Lovely Professional University Punjab, 144401 Tel: 1800 102 4431 E-mail: drsmbhatt@gmail.com |
| Received: August 20, 2015; Accepted: August 21, 2015; Published:August 22, 2015 | |
| Citation: Sheelendra BM (2015) GMO’s Foods: Regulatory Mechanism and Challenges in India. J Bioremed Biodeg 6:e165. doi:10.4172/2155-6199.1000e165 | |
| Copyright: © 2015 Sheelendra BM. This is an open-a ccess article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
| Related article at Pubmed, Scholar Google | |
Visit for more related articles at Journal of Bioremediation & Biodegradation
| Keywords |
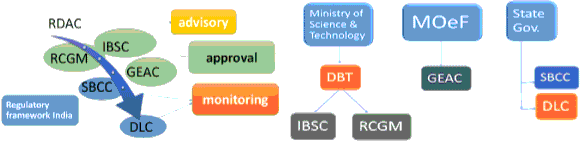
| COP: The Conference of the Parties; GMOs: Genetically modified organism; DBT: Department of science and technology; MOef: Ministry of environment and forest; GEAC: Genetic Engineering Appraisal Committee; SBCC: State Biotechnology Coordination Committees; DLC: District Level Committees; RCGM: Review Committee on Genetic Manipulations; WTO: World trade organization (Figure 1) [1]. |
| Biosafety and GMO��?s |
| The battle is still among people who oppose or supports GMO’s product. Nevertheless feeding of mounting population is main concern. That’s why Public Interest Litigation (PIL) on GMOs products release were kept in front of Supreme Court in 2014 and surprisingly, the report of scientific members were in support of GMOs. Therefore, its case study is very important. Bt-cotton was released in 2002 in Gujarat where, there was report of 24% decrease in pesticide consumption [2] but some author assumed that bt-cotton was responsible for the death of Vidarbha farmers [3]. Recently Forbes magazine report shows many features behind failure of not responding GMO crops such as soil condition, and biggest problem of farmers not understanding GMOs and thus selling too low to byers 2015 report [4]. |
| If we look data of increasing GM crops in developing countries then its around 26% in 2001 while China has approved around 31 GM crops [5]. Now need to change the mind of people. Current Modi government now supporting large scale trials of GMOs products and has allowed to grow various transgenic crops such as transgenic, cotton, rice, mustard, maize (corn), brinjal and chickpea. This may change the future of crops since India is second largest producer of crops and 50% oil is produced alone by Indian famers [1]. Anti GMO groups protested heavily against launch of Bt-brinjal in 2010 India but now it becomes fourth largest GMO crop in world after United States, Brazil and Argentina. |
| Application of biotechnology seems to be one of the essential option to foster benefits but environmentalist opposing on the ground of its future impact. Genetic engineering allow to modify the crops genetics intentionally and thus any feature can be added to these products called as GMOs. GMOs may be plant, maybe animal, or may be the microbes. GMOs may even be created after protoplast fusion or direct gene transfer from unrelated organism. One might be familiar with lateral and horizontal gene transfer. The Cartagena protocol on biosafety finally issued various guidelines in 2000 in view of GM related biohazards. Transgene added may be beneficial in killing some insect but also may kill some other beneficial insect. Biosafety testing is performed often in proper containment facility to avoid any harmful impact in the environment. The level of risk is decide by the category of infectious agent and then work is performed as per guideline of Biosafety level (BSL1-4) which is being monitored by various levels of authority from time to time. Field trials are also monitored which is being conducted after the notification and as per RCGM approved protocols [6]. Capacity building at institutional level is major issue in India. For GMO foods its essential to follow Codex standard (as per WTO guidelines) and practices Hazard Analysis and Critical Control Point (HACCP).Risk assessment is performed at every level of GM food release and likewise state or district level committee monitors post risk assessment based on the health aspects such as their allergenicity causing capacity, toxicity and disease causing capacity (Figure 2). |
| The gene behavior in the environment is difficult to predict. People worldwide have more fear about GMOs than natural food crops. They are basically concern about the future where novel gene insertion can cause origin of new weeds, or spread of new allergens or Bacteria develop resistance to antibiotics and thus new toxicity and diseases in the society [7]. Biosafety encompasses all the guidelines and mechanism related to Genetically Modified Organisms (GMOs). |
| Understanding Biosafety Regulation |
| There is complex chain of regulatory Framework In India for purpose of biosafety which has been depicted in (figure 1). DBT, department of science and technology and ministry of environment and forest (MOef) is the two main regulatory body. |
| DBT has been setup under Ministry of science and technology while GEAC has been setup under MOeF. There are various committee setup under the said ministry. Their main work is to regulate the various activities from research activity to storage or trials of GMOs in India. These committees have six statutory authority also. |
| DBT has right to appoints the members of the committees. The GEAC implements any decision based on report of the State Biotechnology Coordination Committees (SBCC) and District Level Committees (DLC) after any events of post release. Other committees are (1) The Institutional Biosafety Committees (IBSC), (2) Review Committee on Genetic Manipulations (RCGM) and both involve in implementation of guidelines. Recombinant DNA Advisory Committee (RDAC) issue guidelines for working over R-DNA technology. The main objective of National biosafety framework is to (1) Regulate all the process related GMOs in all state to Ensures safety of GMO. |
| “Rules for the Manufacture, Use, Import, Export and Storage of Hazardous Microorganisms/ Genetically Engineered Organisms or Cells, 1989 (Rules, 1989)” notified under the Environment Protection Act, (EPA) 1986 covers broad array of rules related to biosafety of GMO. Statutory bodies have been shown in (figure 2). RDAC is advisory while RCGM, IBSC, and GEAC has role of approval of R-DNA products while under state government SBCC and DLC works as monitoring role. RDAC was setup in 1990, and mainly concern with biotech policy framework regarding r-DNA work. Review committee on genetic manipulation (RCGM) has members from various scientific community which has to conduct meeting time to time in every months. Genetic engineering appraisal (approval) committee GEAC, is the Apex committee to permit/authorize the use of GMO and products thereof for large scale field trials and commercial applications. State biotechnology coordination committee (SBCC) is related with monitoring the activity of GMO and in case of violation has role in punitive action in every state. District level committees (DLC), has role in post monitoring of biosafety related issue. Commercial release is also monitored on the basis of invasiveness, gene persistence, gene flow in unrelated organism, adaptation for resistance property. The check is performed for gene contamination via pollen grains or via other mode in ecosystem and for imported items phytosanitary certification is essential along with quarantine procedures [6]. |
| Concluding Remarks |
| It’s hard to deny that GMOs has positive impact but need is to strengthen regulatory framework so that there should not be any delay in commercialization after research. Many bill still pending with government such as BRAI Bill 2013. Gene of Bt Cry1Ab/1Ac shows that they are not persistent into soil more than two months. Similarly trials for golden rice found to be safe and there was no contamination reported with other rice [8]. Therefore need to strengthen the regulatory framework at various levels. |
References
|
Figures at a glance
 |
 |
| Figure 1 | Figure 2 |
Post your comment
Relevant Topics
- Anaerobic Biodegradation
- Biodegradable Balloons
- Biodegradable Confetti
- Biodegradable Diapers
- Biodegradable Plastics
- Biodegradable Sunscreen
- Biodegradation
- Bioremediation Bacteria
- Bioremediation Oil Spills
- Bioremediation Plants
- Bioremediation Products
- Ex Situ Bioremediation
- Heavy Metal Bioremediation
- In Situ Bioremediation
- Mycoremediation
- Non Biodegradable
- Phytoremediation
- Sewage Water Treatment
- Soil Bioremediation
- Types of Upwelling
- Waste Degredation
- Xenobiotics
Recommended Journals
Article Tools
Article Usage
- Total views: 20501
- [From(publication date):
September-2015 - Apr 04, 2025] - Breakdown by view type
- HTML page views : 15520
- PDF downloads : 4981
