Case Report Open Access
Glioblastoma of the Conus Medullaris Following Treatment of Hodgkin's Lymphoma: History of a Case and Literature Review
Pichon B1, Champiat S2,4, Aumont M1, Loussouarn D3, Frénel JS4, Mahé MA1 and Demoor-Goldschmidt C1,5-7*1Department of Radiotherapy, ICO René Gauducheau, Nantes, France
2Department of Medical Oncology, Gustave Roussy, Villejuif, France
3Department of Histopathology, CHU Nantes, Nantes, France
4Department of Medical Oncology, ICO René Gauducheau, Nantes, France
5Faculty of Medicine, University of Nantes, France
6Centre for Research in Epidemiology and Population Health (CESP), INSERM U1018, Université Paris-Sud, Orsay, Université Paris-Saclay, 94807 Villejuif, France
7Department of Clinical Research, Gustave Roussy, U1018 INSERM, B2M, 94805 Villejuif, France
- *Corresponding Author:
- Demoor-Goldschmidt C
Department of Radiotherapy
Institut de Cancérologie de l'Ouest- René Gauducheau
Bd J Monod, 44800, Nantes, St- Herblain, France
Tel: 0240679900
Fax: +33 (0) 240679722
E-mail: charlotte.demoor@ico.unicancer.fr
Received Date: March 09, 2017; Accepted Date: March 22, 2017; Published Date: March 27, 2017
Citation: Pichon B, Champiat S, Aumont M, Loussouarn D, Frénel JS, et al. (2017) Glioblastoma of the Conus Medullaris Following Treatment of Hodgkin's Lymphoma: History of a Case and Literature Review. OMICS J Radiol 6:257. doi: 10.4172/2167-7964.1000257
Copyright: © 2017 Pichon B, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Radiology
Abstract
Purpose: Reports of a glioblastoma arising in a previously irradiated field are rare in the literature, even more so in the conus medullaris of the spinal cord. Method: Case report and review of the literature reporting other radiation-induced intra-medullary glioblastomas. Results: We report a case of glioblastoma of the conus, which subsequently metastasized to the brain, arising in a 45 years old man, nine years after treatment for Hodgkin's lymphoma, which had included the administration of 41 Gy to the brain and spinal cord. Conclusion: Despite a well-conducted treatment by several lines of chemotherapy, the pronostic of radiationinduced glioblastoma is poor especially since these patients often cannot benefit from a re-irradiation due to the maximum dose supported by the spinal cord. Implications of cancer survivors: Associations are now established between therapeutic exposures and specific complications but considerable inter-individual variability is observed for a given therapeutic exposure. In this context, identification of genetic susceptibilities for specific treatment-associated late effect is a very promising future approach. Improvement in our knowledge on genetic susceptibilities may help define more personalized primary therapies that weigh treatment efficacy with the risk of late complications.
Keywords
Glioblastoma; Spinal cord; Hodgkin's lymphoma; Radiotherapy; Radiation-induced; Second cancer
Clinical Practice Points
Glioblastoma of the conus medullaris is a very rare grade 4 glial tumor that can appear in irradiated zone on the terminal medullary cone and may be in this case a serious complication after lymphoma.
Such secondary cancer and others emphasize the needs of improvement in our knowledge on genetic susceptibilities, which may help define more personalized primary therapies that weigh treatment efficacy with the risk of late complications.
Introduction
Glioblastomas, also known as grade IV astrocytomas, are extremely aggressive malignant tumors of the glial cells of the central nervous system. They arise more frequently in the brain than the spinal cord. Their estimated incidence in all sites is 1-5/100,000 in European and North American populations. The prognosis remains grave, whatever the initial site, and despite any surgical, chemotherapeutic or radiological treatment that may be tried.
Glioblastomas may be classified as primary or secondary. Among the secondary tumors of the central nervous system, a few cases of intracerebral glioblastomas in previously irradiated tissue have been reported [1-3], though they are considerably less frequent than secondary meningiomas [4,5]. A tumor must fulfil several criteria to be considered of radiation-induced etiology [2,6,7]: It must develop in a previous radiation field with an interval of several years between the tumors clinical presentation and the original radiotherapy; and be confirmed as histologically distinct from the tumor for which the radiotherapy was originally given.
The goal of this article is to emphasize with the case of a severe adverse effect after radiotherapy for Hodgkin’s lymphoma the needs of Improvement in our knowledge on genetic susceptibilities which may help define more personalized primary therapies that weigh treatment efficacy with the risk of late complications.
Case Report
A medullary glioblastoma was diagnosed in a 45-year-old Caucasian male. Relevant medical history included a melanoma in situ on the skin of the back treated by excision, and a stage IIISB nodular sclerosing Hodgkin's lymphoma (Ann Arbor classification), both presenting at the age of 35. No relevant family history of malignancy was noticed.
History of the Hodgkin's lymphoma
Six months after his melanoma had been treated, the patient complained of enlarged glands in the region of the right clavicle, associated with sweats. A node of the left transverse cervical chain enlarged to 3 cm, was entirely filling the left supraclavicular fossa. Open biopsy and frozen section of the lesion suggested a nodular sclerosing Hodgkin's Lymphoma (HL). The full work-up confirmed the HL, stage IIIB of Ann Arbor classification with bilateral subclavicular, mediastinal involvement, and splenomegaly. The patient responded only partially to the current protocol of chemotherapy ABVD (Adriamycin-bleomycin-vinblastine-dacarbazine), so a course of vindesin-doxorubicin-carmustine-etoposide-methylprednisolone then followed. The chemotherapy was supplemented by radiotherapy over ten weeks. X-ray photons (15 MV) from a linear accelerator were administered in two courses of treatment: to a supra-diaphragmatic mantle field encompassing the mediastinum, and the subclavicular and axillary nodes on each side, receiving 40 Gy in 20 fractions administered daily over four weeks; and a sub-diaphragmatic block field that included the aortic, coeliac and splenic nodes, receiving 30 Gy in 15 fractions over six weeks. The maximum dose received by the spinal cord was 41 Gy. The treatment was reasonably well tolerated, apart from some episodes of nausea and epigastric pain. This treatment resulted in complete remission.
History of the Glioblastoma
Nine years after radiotherapy, the patient presented with progressive difficulty in walking and urinary urgency. On examination, the patient was found to be ataxic with paraparesis. There was a motor deficit of the dorsiflexors of the feet, absent patellar reflexes, and loss of sensation to both touch and point discrimination anteriorly on both the thigh and leg bilaterally.
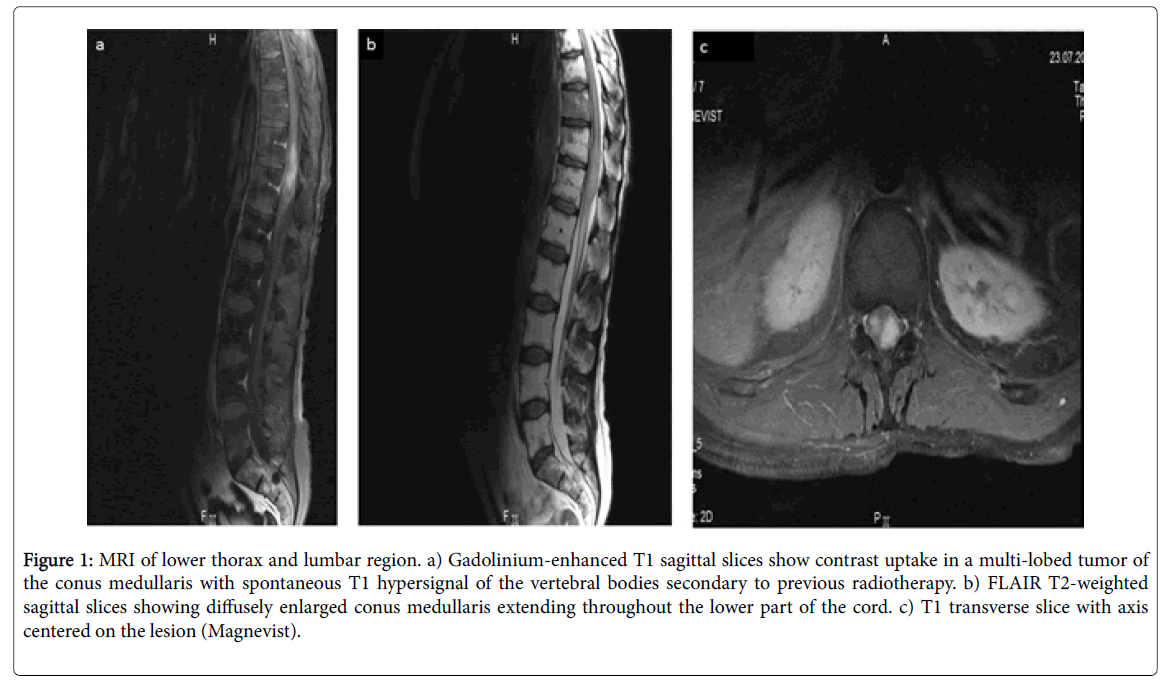
Lumbar puncture and analysis of the CSF revealed high protein (1.85 g/L), significant lymphocytosis with no B-lymphocytes, normal LDH, and normal immuno-electrophoresis; an lumbar and sacral MRI showed an inflammatory hypersignal with increased cord volume as high as T8/9 with a 5 cm area of multiple homogenous contrast uptake in the cord and some nerve roots (Figure 1).
Figure 1: MRI of lower thorax and lumbar region. a) Gadolinium-enhanced T1 sagittal slices show contrast uptake in a multi-lobed tumor of the conus medullaris with spontaneous T1 hypersignal of the vertebral bodies secondary to previous radiotherapy. b) FLAIR T2-weighted sagittal slices showing diffusely enlarged conus medullaris extending throughout the lower part of the cord. c) T1 transverse slice with axis centered on the lesion (Magnevist).
Positron-emission tomography with 18-fluorodeoxyglucose (18- FDG) showed cord involvement at T11-T12 with bony involvement of T10.
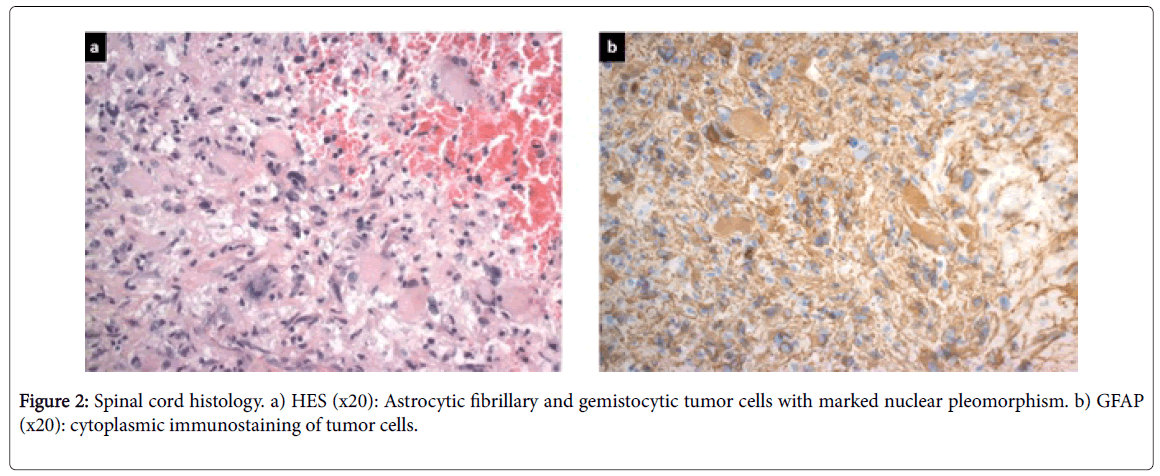
A decompressive and diagnostic surgical procedure was performed to exclude a relapse of the lymphoma. The neurological symptoms showed no improvement on high dose corticosteroids. Pathology reported a high grade astrocytoma of the spinal glial cells of glioblastoma type. Immuno-markers showed a heterogenous expression of GFAP, absent CD20 B-lymphocytes; some CD3 marking of reactive T-lymphocytes, p53 expression in 15% of cell nuclei, and no expression of EGF-receptors or to a modified anti-IDH1 antibody. The proportion of proliferating cells expressing MIB-1 was 15% (Figure 2a and 2b).
As the patient had already received 41 Gy to his spinal cord, the maximum dose being 45 Gy, further radiotherapy was contraindicated. Despite several courses of treatment including first line chemotherapy with monthly temozolomide alone, second line treatment with irinotecan and bevacizumab, third line with fotemustine, and finally a trial of carboplatine and etoposide (VP16), the tumor progressed, both locally and metastasizing to the brain, causing the patient's death 22 months after diagnosis.
Discussion
Secondary malignancy following curative treatment for a Hodgkin's lymphoma is well-recognized in the literature [8,9]. We have found nine cases of spinal cord glioblastomas which were possibly secondary to radiotherapy [3,7,10-16], of which the radiotherapy had been indicated for lymphatic disease in five [7,10,11,13,14]. All patients were male, aged less than 50 years old [7,10,11,13,14] with a presentation interval following radiotherapy of three to nine years (Table 1).
| Case | Author | Reference | Year | Number of cases | Sex | Initial disease | Age at first radiotherapy (years) | Radiation dose (Gy) | Maximum dose to spinal cord (Gy) | Radiation field | Interval between presentations (years) | Glial tumor | Tumor site | Follow-up duration | Secondary tumor treatments |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Clifton et al. | 11 | 1980 | 1 | Male | Hodgkin's lymphoma | 21 | 50/30 | 49.69 | Mantle+cervical+para-aortic | 6 | Glioblastoma | Cervico-thoracic C4-T2 | 10 weeks | Laminectomy |
| 2 | Bazan et al. | 10 | 1990 | 1 | Male | Hodgkin's lymphoma | 19 | 40 | Not reported | Mantle, Waldeyer's ring, para-aortic, splenic | 7 | Astrocytoma Grade II-III | Cervical C6 | 6 months | Laminectomy, chemotherapy |
| 3 | Riffaud et al. | 14 | 2005 | 1 | Male | Hodgkin's lymphoma | 30 | 40 | Not reported | Mediastinal | 9 | Anaplastic glioma | Cervico-thoracic C6-T2 | 11 months | Laminectomy, etoposide, cisplatine, fotemustine+radiotherapy (54 Gy) |
| 4 | Ng et al. | 13 | 2007 | 1 | Male | Hodgkin's lymphoma | 26 | 30.6 | Not reported | Residual lymphadenopathy in the anterior mediastinum | 3 | Glioblastoma | Thoracic | A few weeks | Radiotherapy (50 Gy)+temozolomide |
| 5 | Kikkawa et al. | 7 | 2013 | 1 | Male | Lymphoblastic T-cell lymphoma | 21 | 30 | 37.8 | Residual lymphadenopathy in the anterior mediastinum | 9 | Glioblastoma | Cervico-thoracic C6-T6 with cranio-spinal dissemination | 9 months | Cranio-spinal radiotherapy (30 Gy)+peri-ventricular boost (30 Gy)+cervico-thoracic boost (22.5 Gy)+simultaneous temozolomide plus maintenance |
| 6 | Pichon et al. | Present case | 2014 | 1 | Male | Hodgkin's lymphoma | 35 | 40/30 | Not calculated and reported in the file | Mantle plus lumbar-aortic and splenic block | 9 | Glioblastoma | Terminal cone extending rostrally to T8/T9 then disseminating craniospinally | 24 months | Laminectomy, temozolomide, irinotecan+bevacizumab, fotemustine, carboplatine+etoposide |
Table 1: Malignant glial tumors of the spinal cord following radiotherapy for lymphoma.
This well-recognized risk of radiotherapy represents a rare, but extremely serious, complication of potentially curative treatments for lymphoma, occurring in a patient who has potentially been cured of his disease. The patient's individual predisposition to the development of neoplastic disease caused by a defect in cellular immunity may also play its part. The iatrogenic potential of lymphoma treatments derives from both their direct effects on healthy cells, as well as their immunosuppressive effects more generally. Both may be relevant in the development of the second cancer. Combining chemotherapy and radiotherapy increases this risk, with a significant correlation between the irradiated volume and the risk of a second cancer [9,17,18], which has led to reductions in field size and dose (20-30 Gy) in Hodgkin's lymphoma being recommended [19]. Large treatment fields are now less and less often prescribed. In this case, it is possible there was a genetic predisposition, given the strong family history of malignancy in addition to the patient's own, although there was no evidence for recognized predisposition to malignancy such as the Li-Fraumeni syndrome.
In any case, a family history for genetic predisposition should be sought, and referral to the onco-genetic service may be advisable to inform judgment in the consideration of the risks and benefits of any therapeutic course proposed. Given the range of possible therapeutic choices, such a decision would generally be reached in consultation with a multidisciplinary team. Radiotherapy should be avoided in malignancy associated with p53 mutations or the Li-Fraumeni syndrome.
Radiation-induced glioblastoma of the spinal cord remains a rare condition with nine cases reported in the world literature [3,7,10-16] and the threshold dose to the spinal cord likely to cause a secondary tumor, has not, to our knowledge, been established. In the six cases reported of glial tumors arising in the irradiated field following treatment for Hodgkin's lymphoma, significant doses were received by the spinal cord, of between 37.8 and 49.69 Gy (Table 1). The radiationinduced cerebral glioblastomas were more likely to occur in the physical penumbra at the margins of the treatment field than at the center, where the maximum dose had been received [3,20].
Distant metastasis arising from spinal cord glioblastomas has already been reported [7,21-27], with a poor prognosis of around six months [27]. Taking best known practice in cerebral glioblastoma as a guide, the optimum treatment would seem to be complete macroscopic surgical resection followed by a combination of radiotherapy and chemotherapy [28]. However, this course is rarely feasible given the previous radiotherapy, the unresectable nature of the tumor, and the patient's poor general condition. Further administration of radiotherapy is usually compromised by the dose already received by the spinal cord, at risk for radiation myelitis, though three centers have nevertheless attempted additional radiotherapy [7,13,14]. Where surgery seems contraindicated, stereotactic radiotherapy may be contemplated, with the goal of effective decompression of the cord and pain relief. Palliative chemotherapy often seems the only therapeutic option. We were unable to find any report of a patient with a radiationinduced spinal cord glioblastoma surviving longer than 24 months. This grave prognosis seems impervious to all therapeutic manoeuvres attempted to date (Table 1).
Progress remains to be made on the evaluation of a patient's radio-sensitivity with the future goal of tailoring radiation doses to the patient's genetic characteristics. Identification of genetic susceptibilities for specific treatment-associated late effect is a very promising future approach. Improvement in our knowledge on genetic susceptibilities may help define more personalized primary therapies that weigh treatment efficacy with the risk of late complications. For this patient, another treatment may have been chosen if physicians knew he had a specific radio-sensitivity. Tumors arising in irradiated fields must be histologically distinguished from a late relapse of the original malignancy. A radiation-induced spinal cord glioblastoma following Hodgkin's lymphoma remains an exceptionally rare occurrence. To date, because of the very small number of cases, no standard therapy can be rationally suggested. The prognosis remains grim with-or without cerebro-spinal metastasis, and no treatment has been shown to be effective.
Compliance with Ethical Standards
Informed consent
Written informed consent was obtained from the patient's wife for publication of this report and the accompanying images.
Source(s) of funding-Disclosure of potential conflicts of interest
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Declaration of interested conflicts
The authors declare no interested conflicts with this article.
References
- Fukui K, Inamura T, Nakamizo A, Ikezaki K, Inoha S, et al. (2001) A case showing effective radiotherapy for a radiation-induced glioblastoma. No Shinkei Geka Neuro Sur 29: 673-677.
- Liwnicz BH, Berger TS, Liwnicz RG, Aron BS (1985) Radiation-associated gliomas: a report of four cases and analysis of postradiation tumors of the central nervous system. Neurosurgery 17: 436-445.
- Marus G, Levin CV, Rutherfoord GS (1986) Malignant glioma following radiotherapy for unrelated primary tumors. Cancer 58: 886-894.
- Watts C (1976) Meningioma following irradiation. Cancer 38: 1939-1940.
- Ron E, Modan B, Boice JD, Alfandary E, Stovall M, et al. (1988) Tumors of the brain and nervous system after radiotherapy in childhood. New Eng J Med 319: 1033-1039.
- Cahan WG, Woodard HQ, Higinbotham NL, Stewart FW, Coley BL (1998) Sarcoma arising in irradiated bone-Report of eleven cases.
- Kikkawa Y, Suzuki SO, Nakamizo A, Tsuchimochi R, Murakami N, et al. (2013) Radiation-induced spinal cord glioblastoma with cerebrospinal fluid dissemination subsequent to treatment of lymphoblastic lymphoma. Surg Neurol Int 4: 27.
- Arseneau JC, Sponzo RW, Levin DL, Schnipper LE, Bonner H, et al. (1972) Nonlymphomatous malignant tumors complicating Hodgkin's disease: Possible association with intensive therapy. N Eng J Med 287: 1119-1122.
- Canellos G, Arseneau J, Devita V, Whang-Peng J, Johnson RC (1975) Second malignancies complicating Hodgkin's disease in remission. The Lancet 305: 947-949.
- Bazan C, New PZ, Kagan-Hallet KS (1990) MRI of radiation induced spinal cord glioma. Neuroradiology 32: 331-333.
- Clifton MD, Amromin GD, Perry MC, Abadir R, Watts C, et al. (1980) Spinal cord glioma following irradiation for Hodgkin's disease. Cancer 45: 2051-2055.
- Grabb PA, Kelly DR, Fulmer BB, Palmer C (1996) Radiation-induced glioma of the spinal cord. Pediatric Neurosurgery 25: 214-219.
- Ng C, Fairhall J, Rathmalgoda C, Stening W, Smee R (2007) Spinal cord glioblastoma multiforme induced by radiation after treatment for Hodgkin disease: Case report. J Neurosurgery: Spine 6: 364-367.
- Riffaud L, Bernard M, Lesimple T, Morandi X (2006) Radiation-induced spinal cord glioma subsequent to treatment of Hodgkin’s disease: case report and review. J Neuro-oncol 76: 207-211.
- Steinbok P (1980) Spinal cord glioma after multiple fluoroscopies during artificial pneumothorax treatment of pulmonary tuberculosis: case report. J Neurosurg 52: 838-841.
- Ahn SJ, Kim IO (2012) Spinal cord glioblastoma induced by radiation therapy of nasopharyngeal rhabdomyosarcoma with MRI findings: case report. Kor J Rad 13: 652-657.
- Boivin JF, Hutchison GB, Lyden M, Godbold J, Chorosh J, et al. (1984) Second primary cancers following treatment of Hodgkin's disease. J Nat Cancer Inst 72: 233-241.
- Ng AK, Bernardo MP, Weller E, Backstrand K, Silver B, et al. (2002) Second malignancy after Hodgkin disease treated with radiation therapy with or without chemotherapy: long-term risks and risk factors. Blood 100: 1989-1996.
- Diehl V, Thomas RK, Re D (2004) Part II: Hodgkin's lymphoma-diagnosis and treatment. The Lancet Oncol 5: 19-26.
- Hely M, Fryer J, Selby G (1985) Intramedullary spinal cord glioma with intracranial seeding. J Neurol Neurosurg Psych 48: 302-309.
- Elsamaloty H, Zenooz NA, Mossa-Basha M (2006) Glioblastoma multiforme (GBM) of the conus medullaris with brain and brain stem metastases. Eur J Rad Extra 58: 59-62.
- Kendrick DF, Bonnin JM, Garcia JH (1987) Metastases of a spinal glioblastoma multiforme into an intracranial arachnoid cyst. Neurosurgery 20: 780-783.
- Medhkour A, Chan M (2005) Extremely rare glioblastoma multiforme of the conus medullaris with holocord and brain stem metastases, leading to cranial nerve deficit and respiratory failure: a case report and review of the literature. Surg Neurol 63: 576-582.
- Mayer RR, Warmouth GM, Troxell M, Adesina AM, Kass JS (2012) Glioblastoma multiforme of the conus medullaris in a 28-year-old female: A case report and review of the literature. Clin Neurol Neurosurg 114: 275-277.
- Mori K, Imai S, Shimizu J, Taga T, Ishida M, Matsusue Y (2012) Spinal glioblastoma multiforme of the conus medullaris with holocordal and intracranial spread in a child: a case report and review of the literature. The Spine Journal 12: e1-e6.
- Takara E, Ide M, Yamamoto M, Imanaga H, Jimbo M, et al. (1985) Case of intracranial and spinal dissemination of primary spinal glioma. No Shinkei Geka Neurol Surg 13: 301-305.
- Cohen AR, Wisoff JH, Allen JC, Epstein F (1989) Malignant astrocytomas of the spinal cord. J Neurosurg 70: 50-54.
- Stupp R, Mason WP, Van Den Bent MJ, Weller M, Fisher B, et al. (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Eng J Med 352: 987-996.
Relevant Topics
- Abdominal Radiology
- AI in Radiology
- Breast Imaging
- Cardiovascular Radiology
- Chest Radiology
- Clinical Radiology
- CT Imaging
- Diagnostic Radiology
- Emergency Radiology
- Fluoroscopy Radiology
- General Radiology
- Genitourinary Radiology
- Interventional Radiology Techniques
- Mammography
- Minimal Invasive surgery
- Musculoskeletal Radiology
- Neuroradiology
- Neuroradiology Advances
- Oral and Maxillofacial Radiology
- Radiography
- Radiology Imaging
- Surgical Radiology
- Tele Radiology
- Therapeutic Radiology
Recommended Journals
Article Tools
Article Usage
- Total views: 3556
- [From(publication date):
April-2017 - Jul 12, 2025] - Breakdown by view type
- HTML page views : 2706
- PDF downloads : 850