Erythropoietin Ameliorates Experimental Autoimmune Myasthenia Gravis
Received: 30-Aug-2015 / Accepted Date: 30-Sep-2016 / Published Date: 05-Oct-2016
Abstract
Myasthenia gravis (MG) is an autoimmune disorder characterized by weakness and fatigability of skeletal muscles. Erythropoietin (EPO) is a cytokine required to maintain erythroid cells. Recombinant human EPO
(rHuEPO) was found to act also on immune cells. Beneficial effects of rHuEPO therapy have been demonstrated in several experimental autoimmune models.
We have tested the effect of rHuEPO on the course of experimental autoimmune MG (EAMG) when treatment was initiated either at the acute or at the chronic phase of the disease. A significant ameliorating effect on the course of EAMG was achieved when the treatment started at the acute phase of EAMG, which was accompanied by elevated numbers of Treg. In addition, rHuEPO reduced the levels of anti-acetylcholine receptor (AChR) antibodies.
Our findings suggest that rHuEPO interferes in the course and progression of EAMG and may thus open new clinical opportunities for its use for immunomodulation of MG.
Keywords: Erythropoietin; Myasthenia Gravis; Autoimmune disorders
5481Introduction
Myasthenia gravis (MG) is an autoimmune disease characterized by muscle weakness and fatigue, in which autoantibodies are generated against the nicotinic acetylcholine receptor (AChR) at the neuromuscular junction (NMJ) [1-3] and as a consequence, impair the transmission of signals from nerve to muscle. Experimental autoimmune MG (EAMG), induced by immunization of animals with Torpedo AChR, serves as a reliable model for the human disease and is suitable for investigating mechanism(s) underlying the clinical and immunopathological manifestations of myasthenia [4-6], as well as for examining new therapeutic strategies.
CD4+ T helper cells play a key role in MG and EAMG and assist autoreactive B cells to produce anti-AChR antibodies [7,8]. AChRspecific T cells and autoantibody-producing plasma cells have been found in hyperplastic MG thymus [9]. Upon antigenic stimulation naïve CD4+ T cells can proliferate and shift towards T helper type 1 (Th1), Th2, Th17 and regulatory T cell subsets. Th1 and Th17 cells are characterized by a production of pro-inflammatory cytokines; these subsets of Th cells participate in the development of autoimmune disorders. Th17 cells are a unique subset of CD4+ Th cells and may play an important pathogenic role in T cell mediated autoimmune diseases and tissue inflammation [10]. Another distinct T cell population, regulatory T cells (CD4+ CD25+ Foxp3+), termed Tregs, play a central role in maintaining peripheral tolerance and controlling the destructive self-reactive T cells found in autoimmune animal models of arthritis, multiple sclerosis, diabetes, and inflammatory bowel disease [11]. We have shown a defective function and number of Tregs that are associated with disease severity [12] and disequilibrium between T effector (Th1, Th2 and Th17) and Treg cells in EAMG [13]. One of the main goals in treatment of autoimmune diseases is to prevent damage to tissues by employing immunomodulatory therapeutic strategies that simultaneously target the pathogenic immune response while restoring immune tolerance.
The hormone erythropoietin (EPO), produced by the adult kidney, is an essential regulator of red blood cell production through EPO receptors (EPO-R) expressed on the surface of erythropoietic progenitor cells. It is widely applied to treat the anemia of various origins, mainly which associated with chronic kidney disease [14]. However, EPO-Rs were also found to be expressed in a variety of non-erythroid tissues [15,16]. EPO signaling was thus found to contribute to wound healing [17], angiogenesis [18] cardio- [19] and neuro-protective effects [20].
EPO was also found to have anti-inflammatory and immuno-modulatory effects [21], possibly via EPO-Rs expressed on cells of the immune system [22]. Such modulatory effects were exerted by EPO on dendritic cells [23] and on CD4+ T lymphocytes [24]. As a result, EPO treatment may be beneficial for a range of disorders including Alzheimer's disease, autoimmune diseases such as inflammatory bowel disease or autoimmune encephalomyelitis [21,25,26].
In the present study we questioned whether EPO could ameliorate the course of MG. In order to asses this issue we utilized an experimental model of myasthenia gravis EAMG in rats. Our results show that rHuEPO therapy exerts a protective effect on disease course in EAMG rats. The ameliorating effects were associated with elevated hematological parameters, reduced autoantibodies, increased Treg cell population and reduced pro-inflammatory cytokines. The observed mechanisms suggest a therapeutic potential of rHuEPO in suppressing EAMG.
Materials and Methods
Animals
Female Lewis rats 6–7 week of age were obtained from the Animal Breeding Center of the Weizmann Institute of Science (Rehovot, Israel) and were maintained in the institute’s animal facilities. All experiments were performed according to the institutional guidelines for animal care.
Induction and clinical evaluation of EAMG
EAMG was induced in rats by injection of rats with Torpedo AChR, purified from the electric organ of Torpedo californica by affinity chromatography, as described [27]. Rats were immunized once in both hind footpads by s.c. injections of Torpedo AChR (40 μg/rat), emulsified in complete Freund's adjuvant (CFA) (Sigma-Aldrich, Rehovot, Israel) and supplemented with additional nonviable Mycobacterium tuberculosis H37RA 0.5 mg/rat (Difco Laboratories, Detroit, MI). Rats were monitored on alternate days for weight changes and the clinical score was recorded by double-blind evaluation and graded 0–4 as previously described [28]. All experimental groups consisted of 8 rats each, unless otherwise specified, and all experiments were repeated twice.
rHuEPO treatment regimens
Immediately after EAMG induction, rats were randomly divided into three groups (n=8 each): Group 1, Acute phase treatment: rHuEPO (Eprex 40,000 IU/ml, Janssen-Ortho Inc, Canada) treatment started one day after disease induction with 720 units injected i.p on alternate days for 2 weeks and then with 360 units on alternate days, until the end of the experiment. Group 2, Chronic phase treatment: rHuEPO treatment started 4 weeks after disease induction with 720 units injected i.p on alternate days for 2 weeks and then with 360 units on alternate days until the end of the experiment. Group 3, Control group: Rats were injected i.p with PBS on alternate days until the end of the experiment (see Figures 1C and 1D for a schematic outline of rHuEPO injections).
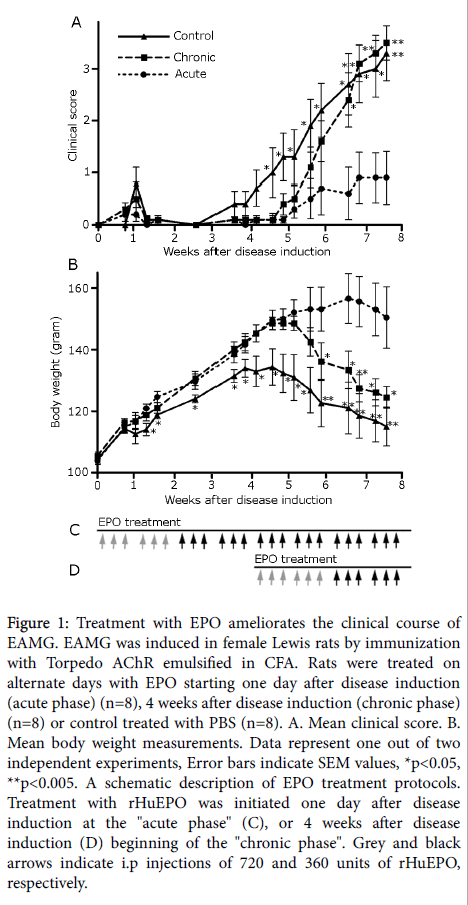
Figure 1: Treatment with EPO ameliorates the clinical course of EAMG. EAMG was induced in female Lewis rats by immunization with Torpedo AChR emulsified in CFA. Rats were treated on alternate days with EPO starting one day after disease induction (acute phase) (n=8), 4 weeks after disease induction (chronic phase) (n=8) or control treated with PBS (n=8). A. Mean clinical score. B. Mean body weight measurements. Data represent one out of two independent experiments, Error bars indicate SEM values, *p<0.05, **p<0.005. A schematic description of EPO treatment protocols. Treatment with rHuEPO was initiated one day after disease induction at the "acute phase" (C), or 4 weeks after disease induction (D) beginning of the "chronic phase". Grey and black arrows indicate i.p injections of 720 and 360 units of rHuEPO, respectively.
Anti-AChR antibodies measurement
Sera of treated rats were collected by retro-orbital bleeding 3 weeks following disease induction. Anti-rat AChR antibodies were determined by standard ELISA as follows:
96 well microtiter plates were coated with Rα1-205 [29] a recombinant fragment corresponding to the extracellular portion (amino acid residues 1-205) of the rat AChR α-subunit (10 mg/100 ml in Tris-Cl pH 8.0), and reacted with 100 μl of rat sera at a dilution of 1:1000 for total IgG. Rabbit anti-Rat alkaline phosphatase antibody (Jackson immunoresearch laboratories, Inc. West Grove, PA, 1:10,000 dilution in 1.5% BSA in PBS) was added and antibody levels were evaluated by measuring the optical density at 405 nm.
Hb, HCT and RBC detection
The hematological parameters –Hb, HCT and RBC, were measured on automated hematological analyzer by A.M.L (American Medical Laboratories, Israel LTD).
T regulatory cell characterization by flow cytometry
Rat splenocytes were depleted of red blood cells using ACK lysis buffer (Sigma) and resuspended in PBS. Surface markers on splenocytes (3 × 106 cells) were analyzed by binding to anti-CD4-APC (Biolegend, San Diego, CA), and anti-CD25-PE (eBioscience) mAbs. To evaluate intracellular Foxp3 expression, lymphocytes were stained with anti-CD4 and anti-CD25 mAbs, fixed and permeabilized according to the manufacturer’s instructions (Foxp3 detection kit, BD Biosciences). Lymphocyte staining with anti-Foxp3-PerCP-Cy5.5 (eBioscience) was determined by flow cytometry and data acquisition was performed with FlowJo software (TreeStar Inc., Ashland, OR, USA).
RNA isolation and quantitative real-time PCR
Total RNA was extracted from rat splenocytes at the end of the experiment (8 weeks following immunization with AChR). Extraction was performed using the high pure RNA Isolation Kit (Roche, Mannheim, Germany) according to the manufacturer's instructions. The concentration of total RNA was measured by NanoDrop ND-1000 Spectrophotometer (Thermo Scientific, Wilmington, DE). Complementary DNA was prepared and quantitative real-time reverse transcription (PCR) was performed using the LightCycler system (Roche) according to the manufacturer’s instructions. Primer sequences (forward and reverse, respectively) were: Rat IL-18 5′- ACAAACCCTCCCCACTAAC - 3′, 5′- ACGTGTTCCAGGACACAACA-3′, TNF-α 5′- CACGTCGTAGCAAACC-3′, 5′-GGTGAGGAGCACATAGT-3′, TGF- β 5′-CAAGGGCTACCATGCCAACT-3′, 5′- CCGGGTTGTGTTGGTTGTAGA-3′ and β-actin 5′- TACTGCCCTGGCTCCTAGCA-3′, 5′- GTAGTGTCCGTAAGTCCT-3′. The levels of β-actin were used to normalize gene expression levels.
Statistical analysis
Differences in mean values between experimental groups were assessed with the Student’s t test. P values lower than 0.05 were considered significant.
Results
EPO treatment ameliorates EAMG in rats
We evaluated the potential of rHuEPO to ameliorate the severity of EAMG in rats. EPO was administered, starting either one day after EAMG induction (acute phase), or starting 4 weeks after primary immunization (chronic phase of disease), as schematically depicted in Figures 1C and 1D). A control, non-treated group, consisted of rats in which EAMG has been induced, as in other groups, and were administered with PBS, instead of with rHuEPO.
Control rats, receiving only PBS following immunization, developed a typical two-phase EAMG, with an early short phase of mild muscular weakness around 8 days post disease induction (p.i.) and a later phase of progressive muscular weakness from day 25 p.i., which has been aggravated during the observation period of 8 weeks. A delay in disease onset was observed in both acute and chronic phase treatment groups (Figure 1A).
A marked reduction in the severity of EAMG, as measured by mean clinical scores, was observed in the acute phase rHuEPO-treated rats. Thus, for example, the disease severity of rats in the acute phase-treated group, at day 46 p.i. (mean clinical score=0.6 ± 0.5) was significantly lower from that of the control, non-treated rats (mean clinical score=2.70 ± 0.6, p ≤ 0.009). However, only a minor ameliorating effect was observed in the chronic phase rHuEPO-treated group (Figure 1A).
Loss of body weight can be an additional indicator for disease severity. The results of body weight measurements (Figure 1B) corroborated the results obtained by mean clinical score determinations. There has been a marked improving effect on body weight values in the acute phase rHuEPO-treated rats, when compared with body weight values in the control, PBS-treated rats.
As shown in Figure 1B, in control rats, body weight peaked at day 25 p.i. and then decreased progressively, as myasthenic disease progressed in both the control and chronic phase rHuEPO-treated groups. No decrease in body weight was determined from day 25 up to day 55 p.i. in acute phase rHuEPO-treated rats. For instance, on day 41 p.i., the body weight in the acute phase rHuEPO-treated rats was significantly higher (mean body weight=153.1 ± 7.2 gr) as compared with the control PBS-treated rats (mean body weight=122.6 ± 7.2 gr, p ≤ 0.005). In agreement with the mean clinical score data, there has been only a minor effect on body weight values in the chronic phase rHuEPO-treated rats.
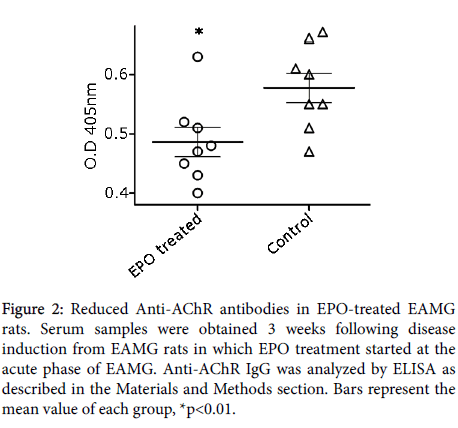
To further determine the ability of rHuEPO treatment to alter the level of anti-AChR antibodies, we determined the levels of these antibodies by ELISA. AChR-specific antibodies in serum samples obtained 3 weeks following disease induction from EAMG rats receiving rHuEPO treatment at the acute phase. This assay demonstrated a significant decrease in the levels of total anti-AChR antibodies in the rHuEPO-treated group (0.49 ± 0.025 p ≤ 0.01) as compared to control-treated rats (0.58 ± 0.025) (Figure 2).
Figure 2: Reduced Anti-AChR antibodies in EPO-treated EAMG rats. Serum samples were obtained 3 weeks following disease induction from EAMG rats in which EPO treatment started at the acute phase of EAMG. Anti-AChR IgG was analyzed by ELISA as described in the Materials and Methods section. Bars represent the mean value of each group, *p<0.01.
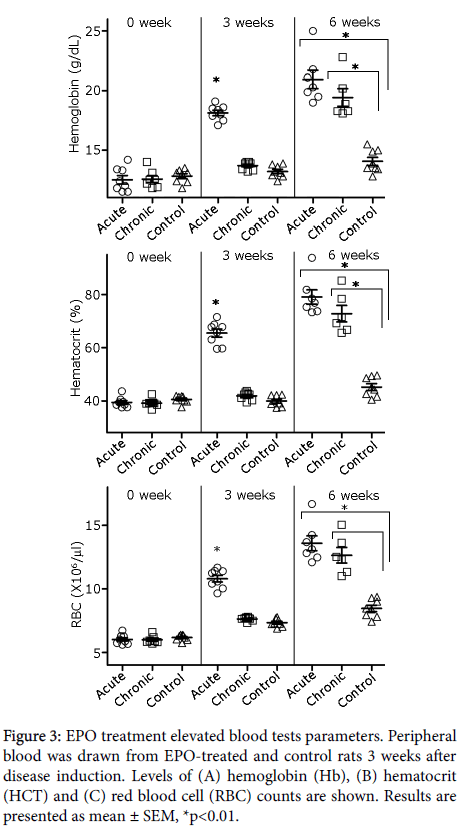
Effect of rHuEPO on hemoglobin, hematocrit and red blood cell count
The effect of rHuEPO treatment on hemoglobin (Hb), hematocrit (HCT) and red blood cell (RBC) counts in the whole blood of the experimental rats was analyzed. Figure 3, top panel, shows a significant, expected, rise in Hb levels in acute phase rHuEPO-treated rats (18.1 ± 0.2; p<0.001), 3 weeks following from disease induction, as compared to Hb levels measured in chronic phase rHuEPO-treated (13.7 ± 0.1) and control-treated rats (13.2 ± 0.2). Six weeks after disease induction both rHuEPO treatment regimens led to significant increase in Hb levels (20.9 ± 0.8, p<0.01 and 19.4 ± 0.7; p<0.01, respectively), as compared to the control-treated rats (14.1 ± 0.3). HCT levels (Figure 3, middle panel) essentially mirrored the rise in Hb: they were significantly elevated in acute phase rHuEPO-treated rats (65.5 ± 1.5; p<0.001) 3 weeks following disease induction, as compared to chronic phase rHuEPO-treated (41.9 ± 0.5) and control rats (39.9 ± 0.2). Six weeks following disease induction, both rHuEPO regimens led to a significant increase in HCT (79 ± 2.7, p<0.01 and 72.8 ± 3.1, respectively), as compared to the control rats (45.2 ± 1.3). Similarly (Figure 3, bottom panel), following 3 weeks from disease induction, RBC counts were significantly elevated in the acute phase of rHuEPO-treated rats (10.8 ± 0.3, p<0.001), as compared to chronic phase rHuEPO-treated (7.6 ± 0.1) and control rats (7.3 ± 0.1). Six weeks following disease induction, both rHuEPO regimens led to increased RBC counts (13.6 ± 0.6, p<0.001 and 12.6 ± 0.6, p<0.001, respectively), as compared to the control rats (8.5 ± 0.3).
Effect of rHuEPO on inflammatory response
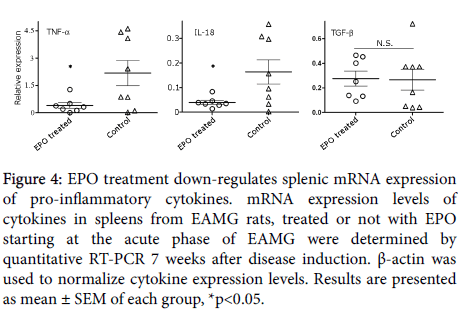
At the end of the experiment, the effect of rHuEPO treatment (initiated at the acute phase of disease) on pro- and anti- inflammatory cytokines’ mRNA expression levels, was analyzed. As shown in Figure 4. EAMG rats, treated with rHuEPO, displayed a significant reduction in splenic TNF-α and IL-18 mRNA transcripts (0.46 ± 0.15, p<0.02 and 0.04 ± 0.01, p<0.02, respectively), as compared to control rats (2.18 ± 0.7 and 0.16 ± 0.05, respectively). In contrast, no significant changes were determined for the expression levels of TGF-β in rHuEPO-treated, as compared to control rats.
Figure 4: EPO treatment down-regulates splenic mRNA expression of pro-inflammatory cytokines. mRNA expression levels of cytokines in spleens from EAMG rats, treated or not with EPO starting at the acute phase of EAMG were determined by quantitative RT-PCR 7 weeks after disease induction. β-actin was used to normalize cytokine expression levels. Results are presented as mean ± SEM of each group, *p<0.05.
Induction of T regulatory cells by rHuEPO treatment
We then asked whether rHuEPO treatment in EAMG rats was associated with production of regulatory T-cells. Flow cytometry analyses were performed on splenocytes from acute phase rHuEPOtreated group and controls. As depicted in Figure 5, a significant upregulation in the percentage of CD4+CD25+Foxp3+ Treg subset of cells was apparent in the spleens of rHuEPO-treated EAMG rats (6 ± 0.32, p<0.004), as compared to their control counterparts (4.8 ± 0.1).
Figure 5: Up-regulation of CD4+CD25+Foxp3+ T-cell frequency in spleens of EPO- treated EAMG rats. Spleens were obtained from EAMG rats 7 weeks following disease induction in which treatment with EPO started at the acute phase of EAMG. Numbers represent the percentage of CD4+ T cells co-expressing CD25 and Foxp3. Results are presented as the mean ± SEM of each group, *p<0.005.
Discussion
In this study we have shown that treatment with EPO in an experimental rat model of myasthenia could suppress the disease. EPO treatments were administered after disease induction, initiated one day after disease induction (acute phase), or 4 weeks after disease induction (chronic phase). The beneficial effects of EPO treatment were reflected by improved clinical score, attenuated body weight loss and elevated blood parameters (Hb, HCT and RBC counts). These were accompanied by decreased antibodies to AChR in the sera of the EPO-treated rats. Down-regulated mRNA expression levels of pro-inflammatory cytokines and up-regulation of Treg subset in the EPO-treated rats may shed light on the underlying mechanisms.
EPO was administered in two regimens as indicated above, yet the therapeutic potential of EPO was significantly greater when treatment was initiated at the acute phase (starting at day one post disease induction). However, different doses and route of administrations may still be required to optimize EPO suppression of EAMG.
Treatment of EAMG with rHuEPO augmented the baseline levels of the blood parameters (Hb, HCT and RBC), when rats were treated at the acute phase. This effect was reflected as well, when measured after the chronic phase treatment (Figure 3). It was shown that rHuEPO treatment in equivalent doses and schedule of administration, in rats that were kept in hypoxic conditions, could modify the main haematological parameters tested [30]. Moreover, pretreatment with EPO exerts anti-inflammatory effects in hepatic ischemia/reperfusion-injured rats via decrease in TNF-α, IL-1β, IL-6 and TLR2 [31].
The EPO response in hematopoietic cells relies not only on the EPO concentration but also on EPO-R expression on the relevant target cells. EPO-R is expressed on CD4+ T lymphocytes and its levels may be modulated during lymphocyte activation [22,32].
We observed that the EPO therapeutic effects were mediated by Treg up-regulation in the spleen of the treated rats. However, it remains to be clarified whether modulation of the Treg also occurs in other immune compartments and in the disease target organ, the muscle itself. In addition, future studies are needed to determine the proportions of T cells, exerting an anti- and pro- inflammatory effects in those tissues.
It was shown that EPO might possess several immunomodulatory properties, in chronic renal failure patients treatment with rHuEPO normalizes activation parameters of CD4+ T lymphocytes and their proliferative capacity, which could explain its immune-modulatory effects [24]. Positive effects of EPO treatment on the suppression of other autoimmune diseases have been shown. EPO treatment in experimental autoimmune neuritis inhibited Th17 and Th1 cells (secreting IFN-γ and TNF-α), whereas it promoted the increase of CD4+Foxp3+ regulatory T cells and Th2 cells. In macrophages, EPO reduced cytotoxicity and facilitated their protective effects [33].
Systemic administration of rHuEPO in a rodent model of experimental autoimmune encephalomyelitis (EAE) improved the neurological functional recovery and markedly reduced the pathogenic cytokines TNF-α and IL-6 [34-36]. In addition, in this model EPO treatment modulated the Treg/Th17 balance in both the periphery and the inflamed spinal cord by promoting a large expansion in Treg and inhibiting Th17 cells [37,38]. The immunomodulatory role of EPO was shown also in a collagen–induced arthritis model [37,38] and in chemically induced colitis in mice [39].
Of note, ARA290, is a non-erythropoietic 11 amino acids analog of EPO which possesses the anti-inflammatory and neuro-protective effects of EPO, without stimulating hematopoiesis. This peptide has been reported to alter T cell function to suppress inflammation and to ameliorate EAE [40] and protect from experimental autoimmune neuritis [41].
Sarcoidosis is an immune-mediated, inflammatory orphan disease of unclear etiology that can affect many organs associated with small-fiber neuropathy (SFN) that leads to autonomic dysfunction [42]. In a clinical trial, ARA 290 was shown to improve symptoms in patients with sarcoidosis-associated small nerve fiber loss and to increase corneal nerve fiber density [43]. The efficacy of this peptide could be further tested in other autoimmune models to assess its potential to shift the unbalanced immune response.
In parallel to several other neuro-immonmodulatory diseases in which EPO has been shown to play a protective role, we have attempted in the present study to test the protective potential of EPO on myasthenia gravis, an autoimmune neuromuscular disorder.
Conclusion
We report here on the potential of EPO to attenuate EAMG in vivo. The protective effect of EPO is mainly mediated by inhibited cellular immune response, including up-regulation of Tregs and down regulation of inflammatory cytokines expression. Our results suggest that EPO could ameliorate EAMG by regulating the immune response, thus providing new insights into therapeutic approaches for myasthenia gravis and other autoimmune diseases.
Acknowledgements
We thank and respect the deceased co-author, Prof. Miriam C. Souroujon, for her seminal contribution to this study.
References
- Engel AG, Lindstrom JM, Lambert EH, Lennon VA (1977) Ultrastructural localization of the acetylcholine receptor in myasthenia gravis and in its experimental autoimmune model. Neurology 27: 307-315.
- Engel AG, Tsujihata M, Lindstrom JM, Lennon VA (1976) The motor end plate in myasthenia gravis and in experimental autoimmune myasthenia gravis. A quantitative ultrastructural study. Ann N Y Acad Sci 274: 60-79.
- Lindstrom JM, Seybold ME, Lennon VA, Whittingham S, Duane DD (1976) Antibody to acetylcholine receptor in myasthenia gravis. Prevalence, clinical correlates, and diagnostic value. Neurology 26: 1054-1059.
- Christadoss P, Poussin M, Deng C (2000) Animal models of myasthenia gravis. Clin Immunol 94: 75-87.
- Fuchs S, Aricha R, Reuveni D, Souroujon MC (2014) Experimental Autoimmune Myasthenia Gravis (EAMG): from immunochemical characterization to therapeutic approaches. J Autoimmun 54: 51-59.
- Souroujon MC, Brenner T, Fuchs S (2010) Development of novel therapies for MG: Studies in animal models. Autoimmunity 43: 446-460.
- Conti-Fine BM, Milani M, Wang W (2008) CD4+ T cells and cytokines in the pathogenesis of acquired myasthenia gravis. Ann N Y Acad Sci 1132: 193-209.
- Elson CJ, Barker RN (2000) Helper T cells in antibody-mediated, organ-specific autoimmunity. Curr Opin Immunol 12: 664-669.
- Cavalcante P, Le Panse R, Berrih-Aknin S, Maggi L, Antozzi C, et al. (2011) The thymus in myasthenia gravis: Site of "innate autoimmunity"? Muscle Nerve 44: 467-484.
- Fouser LA, Wright JF, Dunussi-Joannopoulos K, Collins M (2008) Th17 cytokines and their emerging roles in inflammation and autoimmunity. Immunol Rev 226: 87-102.
- Sakaguchi S (2000) Regulatory T cells: key controllers of immunologic self-tolerance. Cell 101: 455-458.
- Gertel-Lapter S, Mizrachi K, Berrih-Aknin S, Fuchs S, Souroujon MC (2013) Impairment of regulatory T cells in myasthenia gravis: studies in an experimental model. Autoimmun Rev 12: 894-903.
- Mu L, Sun B, Kong Q, Wang J, Wang G, et al. (2009) Disequilibrium of T helper type 1, 2 and 17 cells and regulatory T cells during the development of experimental autoimmune myasthenia gravis. Immunology 128: e826-836.
- Glaspy JA (2009) Erythropoietin in cancer patients. Annu Rev Med 60: 181-192.
- Cravedi P, Manrique J, Hanlon KE, Reid-Adam J, Brody J, et al. (2014) Immunosuppressive effects of erythropoietin on human alloreactive T cells. J Am Soc Nephrol 25: 2003-2015.
- Fisher JW (2003) Erythropoietin: physiology and pharmacology update. Exp Biol Med (Maywood) 228: 1-14.
- Erbayraktar Z, Erbayraktar S, Yilmaz O, Cerami A, Coleman T, et al. (2009) Nonerythropoietic tissue protective compounds are highly effective facilitators of wound healing. Mol Med 15: 235-241.
- Chong ZZ, Kang JQ, Maiese K (2002) Angiogenesis and plasticity: role of erythropoietin in vascular systems. J Hematother Stem Cell Res 11: 863-871.
- Oba T, Yasukawa H, Nagata T, Kyogoku S, Minami T, et al. (2015) Renal Nerve-Mediated Erythropoietin Release Confers Cardioprotection During Remote Ischemic Preconditioning. Circu J 79: 1557-1567.
- Alural B, Duran GA, Tufekci KU, Allmer J, Onkal Z, et al. (2014) EPO Mediates Neurotrophic, Neuroprotective, Anti-Oxidant, and Anti-Apoptotic Effects via Downregulation of miR-451 and miR-885-5p in SH-SY5Y Neuron-Like Cells. Front Immunol 5: 475.
- Maiese K, Chong ZZ, Li F, Shang YC (2008) Erythropoietin: elucidating new cellular targets that broaden therapeutic strategies. Prog Neurobiol 85: 194-213.
- Lisowska KA, Debska-Slizien A, Bryl E, Rutkowski B, Witkowski JM (2010) Erythropoietin receptor is expressed on human peripheral blood T and B lymphocytes and monocytes and is modulated by recombinant human erythropoietin treatment. Artificial Organs 34: 654-662.
- Lifshitz L, Prutchi-Sagiv S, Avneon M, Gassmann M, Mittelman M, et al. (2009) Non-erythroid activities of erythropoietin: Functional effects on murine dendritic cells. Mol Immunol 46: 713-721.
- Lisowska KA, Debska-Slizien A, Radzka M, Witkowski JM, Rutkowski B, et al. (2010) Recombinant human erythropoietin treatment of chronic renal failure patients normalizes altered phenotype and proliferation of CD4-positive T lymphocytes. Artifi Orga 34: 77-84.
- Chateauvieux S, Grigorakaki C, Morceau F, Dicato M, Diederich M (2011) Erythropoietin, erythropoiesis and beyond. Biochem Pharmacol 82: 1291-1303.
- Nairz M, Sonnweber T, Schroll A, Theurl I, Weiss G (2012) The pleiotropic effects of erythropoietin in infection and inflammation. Microbes Infect 14: 238-246.
- Aharonov A, Tarrab-Hazdai R, Silman I, Fuchs S (1977) Immunochemical studies on acetylcholine receptor from Torpedo californica. Immunochemistry 14: 129-137.
- Lennon VA, Lambert EH, Leiby KR, Okarma TB, Talib S (1991) Recombinant human acetylcholine receptor alpha-subunit induces chronic experimental autoimmune myasthenia gravis. J Immunol 146: 2245-2248.
- Maiti PK, Feferman T, Im SH, Souroujon MC, Fuchs S (2004) Immunosuppression of rat myasthenia gravis by oral administration of a syngeneic acetylcholine receptor fragment. J Neuroimmunol 152: 112-120.
- Sanchis-Gomar F, Martinez-Bello VE, Domenech E, Nascimento AL, Pallardo FV, et al. (2009) Effect of intermittent hypoxia on hematological parameters after recombinant human erythropoietin administration. Eur J Appl Physiol 107: 429-436.
- Liu QS, Cheng ZW, Xiong JG, Cheng S, He XF, et al. (2015) Erythropoietin pretreatment exerts anti-inflammatory effects in hepatic ischemia/reperfusion-injured rats via suppression of the TLR2/NF-B pathway. Transplant Proc 47: 283-289.
- Lisowska KA, Bryl E, Witkowski JM (2011) Erythropoietin receptor is detectable on peripheral blood lymphocytes and its expression increases in activated T lymphocytes. Haematologica 96: 12-13.
- Luo B, Jiang M, Yang X, Zhang Z, Xiong J, et al. (2013) Erythropoietin is a hypoxia inducible factor-induced protective molecule in experimental autoimmune neuritis. Biochimica et Biophysica Acta 1832: 1260-1270.
- Agnello D, Bigini P, Villa P, Mennini T, Cerami A, et al. (2002) Erythropoietin exerts an anti-inflammatory effect on the CNS in a model of experimental autoimmune encephalomyelitis. Brain research 952: 128-134.
- Savino C, Pedotti R, Baggi F, Ubiali F, Gallo B, et al. (2006) Delayed administration of erythropoietin and its non-erythropoietic derivatives ameliorates chronic murine autoimmune encephalomyelitis. J Neuroimmunol 172: 27-37.
- Zhang J, Li Y, Cui Y, Chen J, Lu M, et al. (2005) Erythropoietin treatment improves neurological functional recovery in EAE mice. Brain Res 1034: 34-39.
- Chen SJ, Wang YL, Lo WT, Wu CC, Hsieh CW, et al. (2010) Erythropoietin enhances endogenous haem oxygenase-1 and represses immune responses to ameliorate experimental autoimmune encephalomyelitis. Clin Exp Immunol 162: 210-223.
- Yuan R, Maeda Y, Li W, Lu W, Cook S, et al. (2008) Erythropoietin: a potent inducer of peripheral immuno/inflammatory modulation in autoimmune EAE. PLoS One 3: e1924.
- Nairz M, Schroll A, Moschen AR, Sonnweber T, Theurl M, et al. (2011) Erythropoietin contrastingly affects bacterial infection and experimental colitis by inhibiting nuclear factor-κB-inducible immune pathways. Immunity 34: 61-74.
- Chen H, Luo B, Yang X, Xiong J, Liu Z, et al. (2014) Therapeutic effects of nonerythropoietic erythropoietin analog ARA290 in experimental autoimmune encephalomyelitis rat. J Neuroimmunol 268: 64-70.
- Liu Y, Luo B, Han F, Li X, Xiong J, et al. (2014) Erythropoietin-derived nonerythropoietic peptide ameliorates experimental autoimmune neuritis by inflammation suppression and tissue protection. PloS One 9: 90942.
- Heij L, Dahan A, Hoitsma E (2012) Sarcoidosis and pain caused by small-fiber neuropathy. Pain Res Treat 2012: 256024.
- Dahan A, Dunne A, Swartjes M, Proto PL, Heij L, et al. (2013) ARA 290 improves symptoms in patients with sarcoidosis-associated small nerve fiber loss and increases corneal nerve fiber density. Mol Med 19: 334-345.
Citation: Reuveni D, Gertel-Lapter S, Aricha R, Mittleman M, Fuchs S, et al. (2016) Erythropoietin Ameliorates Experimental Autoimmune Myasthenia Gravis. J Clin Exp Neuroimmunol 1:108.
Copyright: ©2016 Reuveni D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Share This Article
Recommended Journals
Open Access Journals
Article Usage
- Total views: 11922
- [From(publication date): 10-2016 - Apr 02, 2025]
- Breakdown by view type
- HTML page views: 10977
- PDF downloads: 945