Development of an Experimental Model for Neurolisteriosis: Oral Infection with Listeria Monocytogenes Causes Brainstem Lesions and Impairment in Gerbils
Received: 30-Jul-2024 / Manuscript No. JNID-24-143775 / Editor assigned: 01-Aug-2024 / PreQC No. JNID-24-143775 (PQ) / Reviewed: 16-Aug-2024 / QC No. JNID-24-143775 / Revised: 21-Aug-2024 / Manuscript No. JNID-24-143775 (R) / Accepted Date: 23-Aug-2024 / Published Date: 23-Aug-2024 DOI: 10.4172/2314-7326.1000512
Abstract
Listeriosis, a foodborne disease caused by Listeria monocytogenes, is characterized by brain infection in elderly or immunosuppressed patients, maternal-fetal and neonatal infection. Although neurologic listeriosis is one of the most severe forms of the disease, animal models of L. monocytogenes-induced neurologic disease have not been thoroughly investigated. In this study, a model of oral infection in immunosuppressed gerbil was developed to verify the role of immunosuppression in L. monocytogenes infection affecting the brain. Female gerbils treated or not with dexamethasone were orally infected with 106, 108, 1010 CFU/animal of L. monocytogenes. Neurologic changes were evaluated up to 30 days post infection, and bacterial recovery determined in systemic organs as well as in the central nervous system. The group of immunocompromised gerbils infected with 1010 CFU/animal of L. monocytogenes had more severe clinical signs, and had higher bacterial loads in the brain. Bacteria were consistently isolated in the brainstem of infected immunosuppressed gerbils at 15 days post infection, which was associated with lower weight gain, higher neurologic deficit, and marked abscesses in the brainstem. Radiolabeled L. monocytogenes with technetium-99m disseminated at early time points of infection to the brain of immunocompetent and immunosuppressed gerbils. This animal model has great potential to contribute to the expansion of our knowledge on the pathogenesis of neurologic listeriosis.
keywords
Meriones unguiculatus; Neurologic listeriosis; Immunosuppression; Technetium-99m
Introduction
Listeria monocytogenes is a Gram-positive bacterium that causes food-borne infections in humans and animals, including ruminants and rodents [1]. Although it has a relatively low incidence, listeriosis represents an important risk to public health, due to the severity of sequelae, and high mortality rate in risk groups, such as pregnant women, newborns, elderly, or immunosuppressed patients [2]. Although the treatment of infection with antibiotic is effective, the mortality rate in children and immunocompromised adults is high, ranging from 24 to 52% [3]. Listeriosis may result in different clinical syndromes: febrile gastroenteritis, maternal- fetal and neonatal listeriosis, and bacteremia associated or not with central nervous system (CNS) infection [4]. The ability of L. monocytogenes to invade the CNS is associated with one of the most severe forms of disease. Neurologic changes due to L. monocytogenes infection are mainly associated with meningitis and [5]. Clinically it is characterized by fever, headache, sensorial changes, convulsions, and changes of the mental state [5]. Although less frequently, parenchymal abscesses, located primarily in the brainstem and to a lesser extent in the subcortical and thalamus areas, are associated with high rates of mortality and neurologic sequelae [6]. Signs of cerebellar infection such as ataxia and dysmetria, and in the spinal cord such as hemiparesis or hypoesthesia may occur [3,5].
Development of an animal model of neurologic listeriosis would be instrumental for a better understanding of the pathogenesis of L. monocytogenes in the CNS. However, the lack of appropriate animal models has been an obstacle for studying listeriosis in the CNS [7]. A suitable model for neurologic listeriosis would be characterized by infection and inflammation of the CNS after food-borne infection thus mimicking the natural course of the disease. The mouse is by far the most widely used animal model for studies of infectious diseases. However, mice do not become consistently infected when orally challenged with L. monocytogenes, making it difficult to study the pathogenic mechanisms of bacteria in CNS. The low binding affinity of bacterial proteins InlA and InlB with their respective murine cellular receptors (E-cadherin and c- MET), which are required for the invasion of enterocytes and other cells, represents a great limitation for the use of the mouse as a model of oral infection [1,4]. Thus, humanized E-cadherin of mouse or mouse-adapted InlA have been employed to increase this affinity allowing oral infection in this species, but these are not suitable models for CNS infection [8-10]. Therefore, alternative routes of experimental infection in mice are used, although they neglect the natural entrance of L. monocytogenes [6]. Previous studies have demonstrated murine models of listeriosis with brain lesion after administration of L. monocytogenes by intragastric, intravenous, intraperitoneal, intracerebral, or intraneural routes [5,6]. .Recently, Senay et al. 2020 [11] demonstrated neurological listeriosis in mice infected with L. monocytogenes strains isolated from cases of human or animal neurolisteriosis.
Gerbil is the only laboratory animal species that expresses specific cellular receptors for InlA and InlB that are similar to human, so it may be a physiologically relevant animal model for listeriosis [4,12]. However, there are only a few studies of infection with this species [13-17]. Importantly, there are no previous studies that have evaluated infection progression and development of neurologic and brain histopathological changes in gerbils experimentally infected with Listeria. Thus, the aim of this study was to develop a model of oral infection with L. monocytogenes in gerbils that is consistently associated with lesions in the CNS, and to verify the effect of corticosteroids (i.e. immunosuppression) on the susceptibility to infection.
Material and methods
Bacterial strain and growth conditions
- monocytogenes (ATTC 7644, serotype 1/2c) was grown in BHI broth (brain heart infusion, KASVI, Brazil) at 37°C under agitation (200 rpm) for 15 h. Subsequently, 400 μL of the subculture was inoculated into 20 mL of BHI broth at 37°C under agitation (200 rpm) for 4 h. Bacterial suspension was centrifuged at 3000 g for 10 min. The pellet was resuspended in 5 mL of PBS (phosphate buffered saline) and bacterial concentration was determined by spectrophotometry. The concentration of bacterial suspension was adjusted according to each inoculum. Inoculum concentrations were confirmed by serial dilution and plating on BHI agar.
Animal treatment and infection
Female 9-10 week-old gerbils (Meriones unguiculatus) were housed in cages at 22ºC under a 12-h light/dark cycle with food and water ad libitum at the Veterinary School of the Universidade Federal de Minas Gerais (UFMG). Animals were dewormed one week before initiating the experiments. All procedures were approved by Committee for Ethical Use of Experimental Animals (CEUA protocol 38/2015) of UFMG.
Considering the variation in body weight of 42 to 56 grams throughout the experiment, each gerbil was treated with 14-19 mg/kg dexamethazone every other day for 25 days until euthanasia. [18]. Immunocompetent gerbils were inoculated with PBS intraperitoneally on the same days. Infection was done orally with 100 μL of milk cream containing 1 × 106, 1 × 108, or 1 × 1010 colony forming units (CFU) of L. monocytogenes.
Different infective doses, 1 × 106, 1 × 108 and 1 × 1010, were compared based on morbidity, observing clinical and neurologic changes up to 30 days post infection (dpi). Gerbils with severe neurologic signs (ataxia, circling episodes or loss of righting reflex) were submitted to euthanasia. Gerbils without clinical sings were submitted to euthanasia at 30 dpi. Necropsy was performed and blood, cerebrum, cerebellum, brainstem, spleen, liver, and cecum were collected for bacteriology. The infecting dose with a greater number of neurologically affected animals (bacterial load in brain and neurologic signs) was then determined. Gerbils were grouped into four groups with 12 animals each: group I -uninfected immunocompetent (IMC mock); group II - uninfected immunosuppressed (IMS mock); group III - infected IMC; and group IV - infected IMS were infected orally with 1 x 1010 CFU/animal of L. monocytogenes. Clinical signs and histopathology of the CNS were observed in gerbils submitted to euthanasia at 10 and 15 dpi. Bacterial loads of various organs, neurologic deficit, body weight, and leukogram were assessed at 15 dpi.
Bacterial counting
To determine the bacterial load, organ samples were weighed and homogenized in 2 mL of PBS. Fragment of cecum was incubated for 1 h with 100 μg/mL gentamycin (Gibco, USA) to kill extracellular bacteria in the intestinal lumen before homogenization. Blood (100 μL) was obtained by intracardiac puncture after anesthetic overdose and seeded on BHI plates. Serial dilutions were plated on BHI agar (Kasvi, USA), and incubated at 37°C for 24 h for CFU counting.
Leukogram
Total blood (1 mL) was collected in EDTA by retro-orbital puncture, before treatment (-10 dpi) and at 10 days after treatment with dexamethasone (0 dpi) and by intracardiac puncture at 15 dpi. The total leukocyte count was performed by the hematologic counter CC 550 (CELM, Brazil), according to the manufacturer’s protocol. The differential leukocyte count was performed on Giemsa-stained blood smear, with a total of 100 cells counted.
Histology and immunohistochemistry
To evaluate microscopic changes in the CNS, the brain was fixed in 10% buffered formalin solution and coronal sections were made in specific regions (cerebral cortex, hippocampus, cerebellum, and brainstem) with the aid of a wooden matrix. Samples of the brain were embedded in paraffin, sectioned in a microtome (4 μm-thick sections), and stained with hematoxylin and eosin (HE). Histopathological changes were described. Inflammatory and degenerative changes were scored according to the intensity: 0 = absent, 1 = mild, 2 = moderate or 3 = severe, for each region of the brain: cerebrum (cortex, hippocampus, and thalamus), cerebellum and brainstem (midbrain, pons, and medulla). The lesions were categorized as microabscess and meningeal or perivascular inflammatory (meningoencephalitis) and scores of each category established for each of the brain regions.
Immunohistochemistry anti-L. monocytogenes reactions were performed according to Oliveira et al. 2016 [19]. Sections were deparaffinized, hydrated and incubated twice in 10% hydrogen peroxide solution for 5 min to block endogenous peroxidase. Sections were then incubated with 2.5% skim milk in PBS at 37ºC for 45 min. An anti-L. monocytogenes serum (Listeria O antiserum poly. Serotypes 1 and 4, DIFCO-BD, Detroit, USA) was used as primary antibody with a 1:100 dilutions for 45 min at 37ºC. Sections were washed three times in PBS for 5 min, and then incubated with a secondary biotinylated antibody (LSAB, Dako, USA) at 37ºC for 20 min, washed three times in PBS for 5 min, and incubated with the LSAB reagent (LSAB, Dako, USA) for 20 min. Sections were washed three times in PBS for 5 min, and then developed with AEC (Aminoethylcarbazole Substrate Chromogen System, Dako, USA) for 10 min, and washed with distilled water. Sections were counterstained with Mayer’s hematoxylin for 30 sec, washed and mounted with Faramount (Dako, USA). Localization of immunostained bacteria was associated with histopathological lesions.
Microglia (Iba1+), astrocytes (GFAP+) and T lymphocytes (CD3+) were detected also by immunohistochemistry. Sections were dewaxed, rehydrated and followed to antigenic retrieval in Target Retrieval Solution, Citrate pH 6 (Agilent, USA) either in water bath (Iba1 and GFAP) at 96°C for 25 minutes or by pressurized heat (CD3) at 125ºC for 2 minutes (Pascal Pressure Cooker; Dako Cytomation, DK). Endogenous peroxidase was blocked by three bathes of 10% hydrogen peroxide solution in methanol for 5 min each. Non-specific proteins were blocked with either 6% skim milk in PBS for 60 min (Iba1 and GFAP) or using the Novolink Polymer Detection System (CD3) (Leica Biosystems, UK) as recommended by the manufacturer. Sections were incubated for 16 hours at 4°C with the primary antibodies anti-Iba1 (Antibody Iba1, Rabbit, Wako Chemicals, USA / Dilution: 1:1000), anti-CD3 (Clone CD3-12, USA / Dilution: 1:100) and anti-GFAP (Glial Fibrillary Acidic Protein – clone 2E1, Santa Cruz Biotechnology, USA / Dilution: 1:500). Secondary HRP-conjugated antibodies for Iba-1 and GFAP (EnVisionTM FLEX, High pH, Agilent, USA) and CD3 (Novolink Polymer Detection System, Leica Biosystems, UK) were used according to the manufacturers’ instructions. Staining was performed with 3'-Diaminobenzidine (DAB) and counterstaining was carried out using Harris hematoxylin. Slides were observed under light microscopy and digital images from scaned slides were captured by Panomoric Viewer (3DHISTECH, Hungria).
Evaluation of blood-brain barrier permeability
To evaluate permeability of blood-brain barrier (BBB), the Evans Blue dye was used as an albumin extravasation marker, as previously described by Huang et al. [20]. Gerbils from four experimental groups were anesthetized with xylazine and ketamine at 15 dpi. Subsequently, a 2% Evans Blue solution was injected intraperitoneally (5 mg/kg of body weight). After 24 h, gerbils were euthanized and perfused with physiological saline (5 mL per animal). The brain was removed and separated into two hemispheres. Each brain hemisphere was homogenized in 1 mL of PBS and then centrifuged at 4000 g for 30 min at 4°C. The supernatant was collected in aliquots, and 0.7 mL of 100% trichloroacetic acid was added to 0.7 mL of supernatant. The mixture was incubated at 4°C for 18 h and then centrifuged 4000 g for 30 min at 4°C. The dye extracted from the tissue was quantified in 630 nm by microplate reader MR-96A (MINDRAY, China), and concentrations were determined based on a standard curve. The result was normalized and expressed as mg/mL Evans Blue per brain.
SHIRPA test
In order to evaluate neurologic and behavioral changes due to L. monocytogenes infection, gerbils were submitted to the SHIRPA test. The SHIRPA test (Smith Kline Beecham Pharmaceuticals; Harwell MRC Mouse Genome Center and Mammalian Genetics Unit; Imperial College School of Medicine at St Mary's Royal London Hospital; St Bartholomew's Royal London School of Medicine; Phenotype Assessment) was performed on 24 gerbils from all four experimental groups at -10, 0, and 15 dpi [Table 1]. Lists the parameters evaluated in each category evaluated in SHIRPA test [21].
| Functional Domains | Parameters |
|---|---|
| Reflex and sensory function | Visual positioning, pinna reflex, corneal reflex, pinched back and postural reflex |
| Neuropsychiatric status | Spontaneous activity, transfer excitation, escape to the touch, positional passivity, bite, fear, irritability, aggression, and vocalization |
| Motor behavior | Body position, tremor, pelvic elevation, ambulation, tail elevation, trunk bending, negative geotaxis, and locomotor activity |
| Autonomous function | Breath rate, defecation, urination, eyelid closing, piloerection, skin color, heart rate, lacrimation, and salivation |
| Muscle tone and strength. | Strength upon grasping, body tone, limb tone, and abdominal tone. |
Table 1: Classification of functional categories according to the parameters evaluated in SHIRPA test .
Total RNA extraction and quantitative RT-PCR
The brainstem of gerbils infected with L. monocytogenes at 15 dpi and non-infected controls were used to evaluate transcription of cytokines including the proinflammatory interferon gamma (IFNγ) and the anti-inflammatory interleukin 10 (IL-10). Extraction of total RNA was done using Trizol Plus (Invitrogen, USA) according to the manufacturer's instructions. Total RNA was purified using the DNAse kit (Invitrogen, USA) and quantified by spectrophotometry. cDNA was synthesized using 500 ng of total RNA using Taqman Reverse Transcription Reagents (Applied Biosystems, USA). Reverse transcription was performed by sequential incubations at 65ºC for 5 min and 4ºC for 2 min, followed by extension at 37ºC for 30 min and inactivation at 95ºC for 5 min. Real- time PCR was performed using 2 μL of cDNA, 1 μL of each pair of primers (10 μM), 8.5 μL of RNAses free water and 12.5 μL of SYBR Green PCR master mix (Applied Biosystems, USA) in the thermal cycler Step One plus (APPLIED Biosystems, USA) according to the manufacturer's instructions. Data were analyzed using the comparative threshold cycle (CT) method [22]. GAPDH and β-actina were tested as normalizer genes and GAPDH was more stable, then the CT values were normalized based on the expression of GAPDH. Primers designed and used in this study are listed in [Table 2].
| Gene | Primers | product size |
|---|---|---|
| GAPDH | 5’GGCCATCAATGACCCCTTCA3’ 5’CCGTTCTCAGCCTTGACTGT3’ |
102 bp |
| β-actina | 5’CCACCATGTACCCAGGCATT3’ 5’ACTCCTGCTTGCTGATCCAC3’ |
177 bp |
| IFN-γ | 5’AGGAAGCGGAAAAGGAGTCG3’ 5’AGTGCTGGCAGGATTGTTCT3’ |
72 bp |
| IL-10 | 5’ACTTGGGTTGCCAAGCCTTA3’ 5’TTGATTTCTGGGCCGTGGTT3’ |
88 bp |
Table 2: Primers used for quantitative RT-PCR.
Radiolabeling and biodistribution of listeria monocytogenes with 99mTc
A more sensitive method was employed to quantify the systemic dissemination of L. monocytogenes at early stages of infection. Radiolabeling of L. monocytogenes with technetium-99m (99mTc) was based on Diniz et al. [23] with modifications. Briefly, 1 mL of a suspension containing 1010 CFU/animal of L. monocytogenes was added to 500 μL of the solution stannous chloride reducing agent (SnCl2.2H2O/3 mg/mL/HCl 0.25N), and incubated at 37°C for 15 min. After incubation, 500 μL of sodium pertechnetate (Na99mTcO4, IPEN, Brazil) with 74 MBq (2 mCi) activity were added, incubated at 37ºC for 45 min, and centrifuged 4000 g for 20 min. Further, 50 μL of the supernatant were collected and the rest was discarded. To the precipitate 2 mL of PBS were added, homogenized and 50 μL was collected. The supernatant and precipitate radioactivity was determinate by a gamma counter (Wizard, Finland). This procedure was repeated by three times. The percent of 99mTc incorporated into the bacterial cells (precipitate) was determined using the following equation and the results were express in cpm (counts per minute): % radiolabelled = μCi (precipitate) / μCi (precipitate + supernatant) x 100
The 99mTc-L. monocytogenes stability was investigated by in vitro studies in order to determine if the 99mTc atoms remained bound to bacterium. Within this purpose, the radiolabeling was performed as described above. The 99mTc-L. monocytogenes suspension was incubated at 37 °C for 24 h and centrifuged at 4000 g for 20 min. Aliquots (50 μL) of supernatant and resuspended precipitate were collected to radioactivity determination. Bacterial viability of 99mTc-L. monocytogenes was determined by plating in BHI agar and compared to unlabeled bacteria.
To verify the biodistribuition of 99mTc-L. monocytogenes, IMS and IMC gerbils were orally infected with 100 μL of milk cream containing 1010 CFU 99mTc-L. monocytogenes. For scintigraphic images at 3 hpi, gerbils (n=7) were anesthetized with a solution of ketamine and xylazine and placed in dorsal recumbency under the gamma camera (Mediso, Hungary) equipped with a low-energy high-resolution collimator. Images were acquired with a 256×256×16 matrix size, with 20% energy window set at 140 keV, for a period of 10 min. For the ex vivo biodistribution studies at 3 and 24 hpi, gerbils (n=7) were euthanized with an overdose of anesthesia, organs such as stomach, intestine, liver, spleen, and brain were collected, weighed and the radioactivity was then measured (Wizard, Finland). An aliquot of 99mTc-L. monocytogenes containing the same ingested dose was counted simultaneously in a separate tube, which was defined as 100% radioactivity. The results were expressed as the percentage of the ingested dose per gram of tissue (% ID/g), according to the following equation: % ID/g = [cpm/g (tissue) / standard dose] × 100 where: cpm = counts per minute
Statistical analysis
Statistical analysis was performed using the statistical software Prisma 4.0 (GraphPad Software, USA). Bacteriology results of the experimental model were evaluated using the Student’s t test. For the analysis of the parameters of the SHIRPA test functional categories, the area under the curve (AUC) was calculated, and then the Tukey test was used. Parametric data were submitted to analysis of variance (ANOVA) followed by the Tukey test, whereas non parametric data were submitted to the Kruskal-Wallis test. The chi-square test was used to analyze the morbidity rate and frequency of CNS inflammatory change. The level of significance was set at p <0.05.
Results
Listeria monocytogenes-infected immunosuppressed gerbils developed neurologic signs
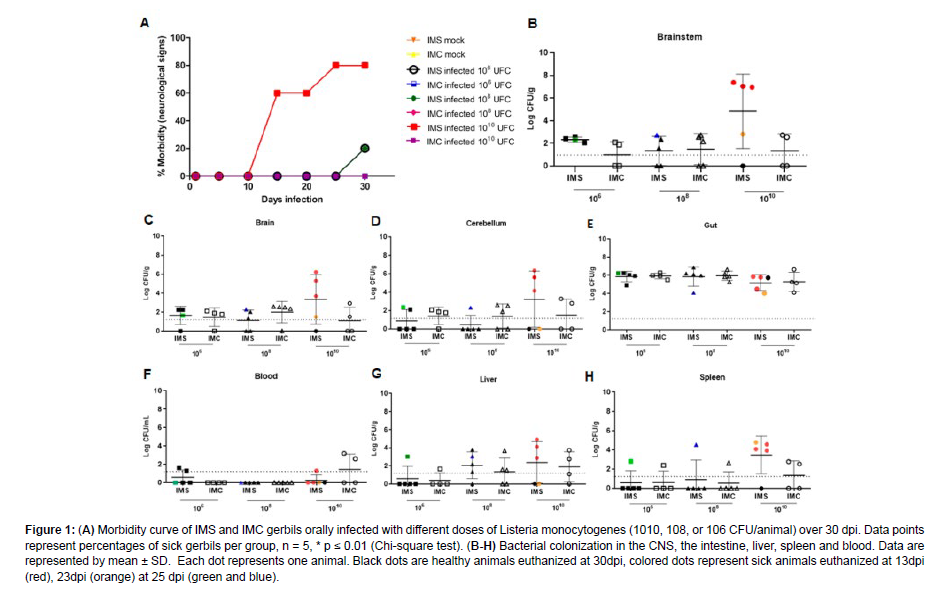
In order to verify whether immunosuppressed gerbils are a suitable experimental model for the study of neurologic listeriosis, and to determine the best infective dose for this model, immunosuppression was induced by corticosteroids, and manifestation of neurologic signs were evaluated in gerbils orally infected with L. monocytogenes. Morbidity rate in each group was determined up to 30 dpi [Figure 1]. Neurologic signs (video supplementary data) (ataxia and stereotypic behaviours) were observed in 80% (4/5) of IMS gerbils infected with 1010 CFU with three gerbils affected at 13 dpi and one at 23 dpi. IMS gerbils infected with 108 or 106 CFU had a lower percentage of sick animals with only one affected gerbil in each group at 25 dpi. The other groups did not develop neurologic changes.
Figure 1: (A) Morbidity curve of IMS and IMC gerbils orally infected with different doses of Listeria monocytogenes (1010, 108, or 106 CFU/animal) over 30 dpi. Data points represent percentages of sick gerbils per group, n = 5, * p ≤ 0.01 (Chi-square test). (B-H) Bacterial colonization in the CNS, the intestine, liver, spleen and blood. Data are represented by mean ± SD. Each dot represents one animal. Black dots are healthy animals euthanized at 30dpi, colored dots represent sick animals euthanized at 13dpi (red), 23dpi (orange) at 25 dpi (green and blue).
In order to evaluate bacterial colonization in the CNS and systemic organs after oral infection by different inocula (1 x 106, 1 x 108, or 1 x 1010 CFU/animal) of L. monocytogenes in IMS and IMC gerbils, bacterial loads were determined in blood, spleen, liver, cecum, brainstem, cerebrum, and cerebellum at the time of euthanasia in gerbils with neurologic signs (13 dpi, 23 dpi or 25dpi) or at 30 dpi for gerbils that did not become sick. A higher colonization was observed in the intestine, even 30 dpi, demonstrating that L. monocytogenes has tropism to this organ, as expected. In Figure 1, bacterial loads were high in the brainstem of IMS gerbils infected with 1010 CFU that showed neurologic signs. No significant difference was observed in bacterial loads recovered from the cerebrum, cerebellum, cecum, spleen, liver, and blood between infected IMS and IMC gerbils [Figure 1]. Almost all gerbils euthanized at 30 dpi did not recover or recovered very little CFU of L. monocytogenes in the tissues evaluated. These results demonstrate that gerbils developed brain infection after oral challenge with 1010 CFU of L. monocytogenes, suggesting a tropism of L. monocytogenes to the brainstem.
Systemic infection with listeria monocytogenes was favored by immunosuppression in gerbils
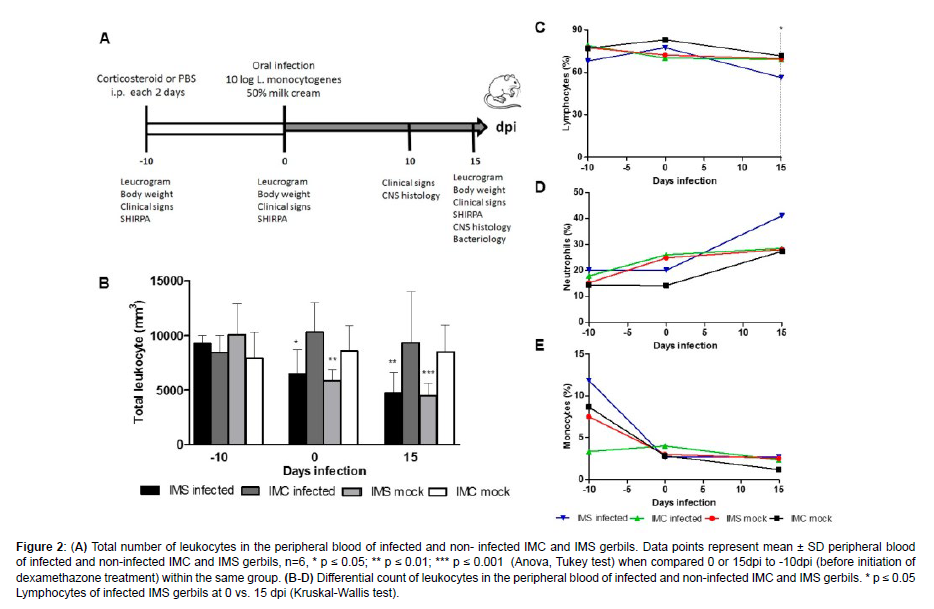
Considering that in the first experiment neurologic changes were observed in IMS gerbils infected with 1010 CFU of L. monocytogenes starting at 13 dpi, we established the investigation of bacterial colonization in the liver, spleen, intestine, blood, cerebrum, cerebellum, and brainstem at 15 dpi, and histopathological examination of the brain at 10 and 15 dpi [Figure 2].
Figure 2: (A) Total number of leukocytes in the peripheral blood of infected and non- infected IMC and IMS gerbils. Data points represent mean ± SD peripheral blood of infected and non-infected IMC and IMS gerbils, n=6, * p ≤ 0.05; ** p ≤ 0.01; *** p ≤ 0.001 (Anova, Tukey test) when compared 0 or 15dpi to -10dpi (before initiation of dexamethazone treatment) within the same group. (B-D) Differential count of leukocytes in the peripheral blood of infected and non-infected IMC and IMS gerbils. * p ≤ 0.05 Lymphocytes of infected IMS gerbils at 0 vs. 15 dpi (Kruskal-Wallis test).
Initially, dexamethasone-induced immunosuppression was confirmed by peripheral blood leukocyte counting. A significant decrease in total leukocytes was observed in uninfected and infected IMS gerbils when compared 0 or 15 to -10 dpi [Figure 2]. No difference was observed in the total leukocytes count of gerbils not treated with corticosteroids (uninfected and infected IMC) In female gerbils, lymphocytes, neutrophils, and monocytes correspond to approximately 59%, 11%, and 2% of blood leukocytes, respectively [24]. In the leukocyte differential counting, a significant decrease in the percentage of lymphocytes after infection with L. monocytogenes was observed in infected IMS gerbils at 15 dpi [Figure 2]. These results demonstrated that immunosuppression in gerbils infected with L. monocytogenes is associated with lymphopenia.
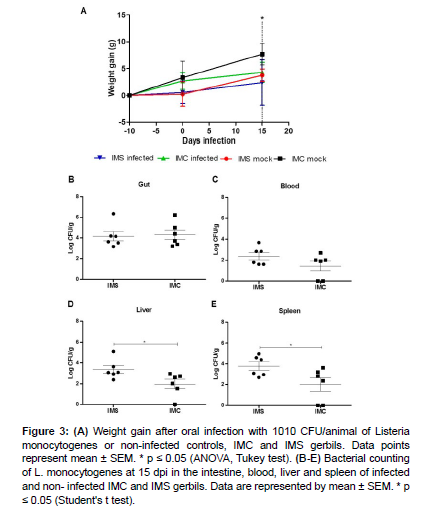
Reduction in weight gain in rodents may reflect lower food intake due to anorexia, so the weight gain variation of the animals in each group was measured over the course of the experiment [Figure 3]. Infected IMS gerbils had a lower weight gain compared to controls (uninfected IMC) at 15 dpi, when there was a significant increase in bacterial loads in the liver and spleen of infected IMS gerbils when compared to infected IMC gerbils [Figure 3]. These results suggest that immunosuppression impairs the control of L. monocytogenes colonization in these systemic sites of infection.
Listeria monocytogenes consistently colonize the brainstem in gerbils
Figure 3: (A) Weight gain after oral infection with 1010 CFU/animal of Listeria monocytogenes or non-infected controls, IMC and IMS gerbils. Data points represent mean ± SEM. * p ≤ 0.05 (ANOVA, Tukey test). (B-E) Bacterial counting of L. monocytogenes at 15 dpi in the intestine, blood, liver and spleen of infected and non- infected IMC and IMS gerbils. Data are represented by mean ± SEM. * p ≤ 0.05 (Student's t test).
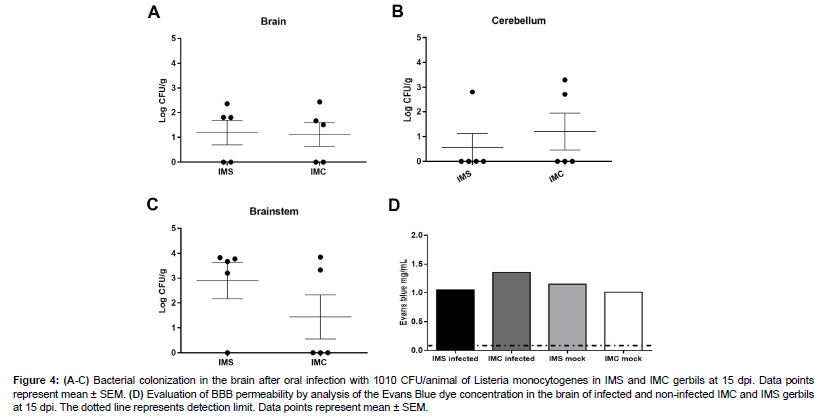
An initial experiment demonstrated that gerbils with evident clinical signs had a marked infection in the brainstem [Figure 1]. Therefore, we verified whether the brainstem colonization was more consistent in immunosupressed gerbils at 15 dpi by comparing bacterial loads in different regions of the brain, namely brainstem, cerebellum, and cerebrum, in IMS and IMC gerbils infected with L. monocytogenes.
Bacteria were recovered from brain and cerebellum was similar in both groups [Figure 4]. Bacteria were recovered from brainstem in 4/5 (80%) IMS gerbils infected with L. monocytogenes, while bacteria were recovered in only 2/5 (40%) gerbils in IMC [Figure 4]. The bacterial loads in the brainstem of IMS gerbils were around 3 logs while brainstem IMC gerbils or brain and cerebellum of both groups recovered around 1 log [Figure 4]. These results confirm that L. monocytogenes consistently colonized the brainstem of IMS gerbils.
Figure 4: (A-C) Bacterial colonization in the brain after oral infection with 1010 CFU/animal of Listeria monocytogenes in IMS and IMC gerbils at 15 dpi. Data points represent mean ± SEM. (D) Evaluation of BBB permeability by analysis of the Evans Blue dye concentration in the brain of infected and non-infected IMC and IMS gerbils at 15 dpi. The dotted line represents detection limit. Data points represent mean ± SEM.
Changes in BBB permeability may favor both the spread of bacteria to the CNS and severity of clinical signs associated with brain acute inflammation [25,26]. We verified whether corticosteroid therapy or L. monocytogenes infection altered BBB permeability by quantifying Evans Blue dye extravasation to brain parenchyma at 15 dpi. There was no significant increase of the dye concentration in the brain parenchyma of the uninfected IMS, infected IMS or infected IMC compared to control group. Thus, there is no evidence of changes in BBB permeability are associated to infection at 15 dpi [Figure 4].
Oral infection with listeria monocytogenes elicited inflammation in the brainstem of gerbils
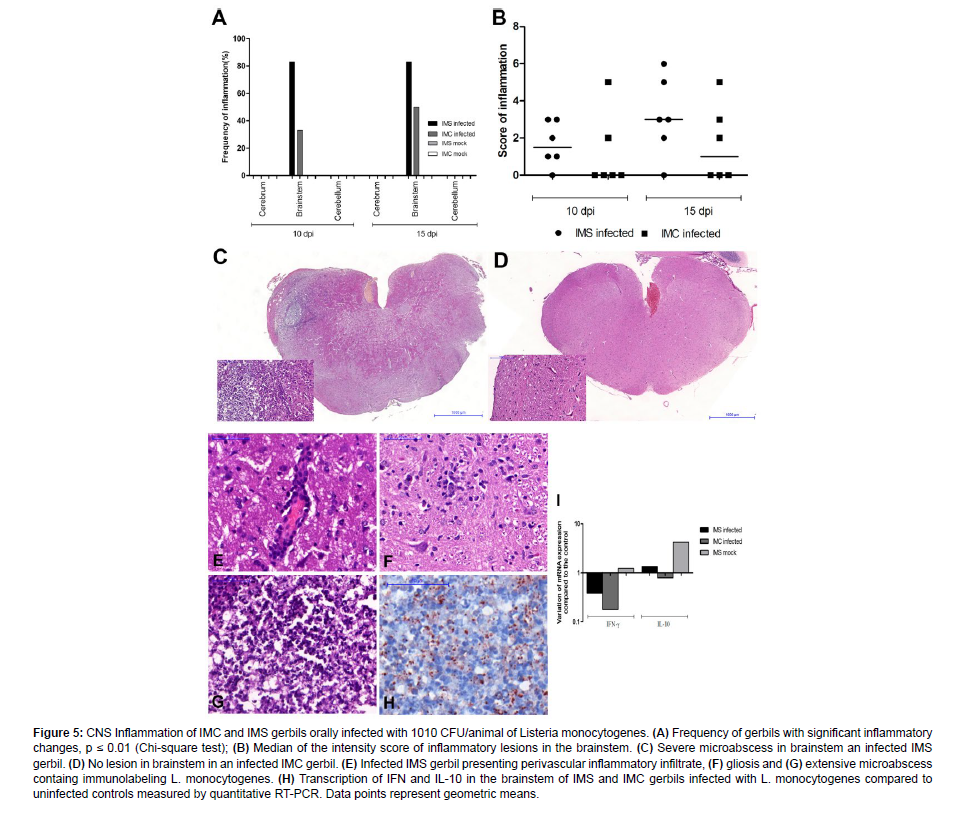
To characterize CNS inflammatory lesions associated with oral infection with L. monocytogenes in gerbils, sections of the cerebral cortex, thalamus, hippocampus, brainstem, and cerebellum were histologically evaluated at 10 and 15 dpi, and the distribution of the bacterium in the CNS was determined by immunohistochemistry at 15 dpi. Lesions in the brainstem (microabscess, perivascular cuffs, and meningitis) were observed unilaterally and predominantly close to the spinal tract of the trigeminal nerve. These lesions were predominant in the group of infected IMS gerbils compared to infected IMC at 10 and 15 dpi [Figure 5]. No significant histopathological changes were observed in the cerebral cortex and cerebellum.
At 10 dpi, the lesions were characterized by unilateral mild multifocal microabcesses with neutrophils and glial cells, mild to moderate multifocal perivascular inflammatory infiltrate and mild multifocal meningitis. With disease progression at 15 dpi, we observed that the lesions were predominantly unilateral (only one gerbil had a bilateral lesion), and they were characterized by multifocal microabcesses that were mild to severe or focally extensive, and surrounded by moderate multifocal meningitis, mild to severe multifocal perivascular cuff, and mild neuronal degeneration. Multiple immunostained L. monocytogenes, predominantly within neutrophils in the microabcess and also in adjacent glial cells were observed associated with microscopic lesions.
Expression of inflammatory mediators contributes to the development of inflammatory changes as well as infection control by inducing protective innate immune response [27]. IFN-γ is an important pro-inflammatory cytokine to control the systemic infection by L. monocytogenes [28] as well as bacterial spread to the CNS [29,30]. IL-10 is an immunoregulatory cytokine that may favor susceptibility to infection by L. monocytogenes [31] although studies in mice indicate that increased resistance to infection is dependent on IL-10 [32]. Thus, we evaluated the expression of IFN-γ and IL-10 transcripts in the brain stem of IMS and IMC gerbils infected or not with L. monocytogenes at 15 dpi by quantitative RT-PCR. Expression of IFN-γ and IL-10 transcripts compared to the control group (uninfected IMC) did not significantly alter at 15 dpi in the different experimental groups. However, it was observed that treatment with dexamethasone induced an increased transcription of IL-10 in uninfected gerbils [Figure 5].
Figure 5: CNS Inflammation of IMC and IMS gerbils orally infected with 1010 CFU/animal of Listeria monocytogenes. (A) Frequency of gerbils with significant inflammatory changes, p ≤ 0.01 (Chi-square test); (B) Median of the intensity score of inflammatory lesions in the brainstem. (C) Severe microabscess in brainstem an infected IMS gerbil. (D) No lesion in brainstem in an infected IMC gerbil. (E) Infected IMS gerbil presenting perivascular inflammatory infiltrate, (F) gliosis and (G) extensive microabscess containg immunolabeling L. monocytogenes. (H) Transcription of IFN and IL-10 in the brainstem of IMS and IMC gerbils infected with L. monocytogenes compared to uninfected controls measured by quantitative RT-PCR. Data points represent geometric means.
Microscopically, there were microabcesses in the brain stem consisting predominantly of neutrophils [Figure 5] and L. monocytogenes immunolabeled by IHQ [Figure 5 and Figure 6]. We showed also by immunohistochemistry that the microabcess has activated microglia on the periphery of the lesion and on the adjacent neuropil [IBA1 + cell - Figure 6], astrocytes activated on the adjacent neuropil [GFAP+ cell – Figure 6] and a few CD3+ lymphocytes infiltrated within and in the periphery of the lesion [Figure 6]. These findings demonstrate that in addition to the neutrophils present in the microabscess, glial cells are involved in the inflammatory process induced in neurolisteriosis.
Figure 6: Cell types in brainstem lesisons of IMC and IMS gerbils orally infected with 1010 CFU/animal of Listeria monocytogenes. (A) Immunostained L. monocytogenes (LSAB) in red inside neutrophils and glial cells in the microabscess of infected IMS gerbil. Increase of immunostained (B) microglias (IBA+), (C) astrocytes (GFAP+), and (D) T cells (CD3+) associated to the microabscess of infected IMS gerbil.
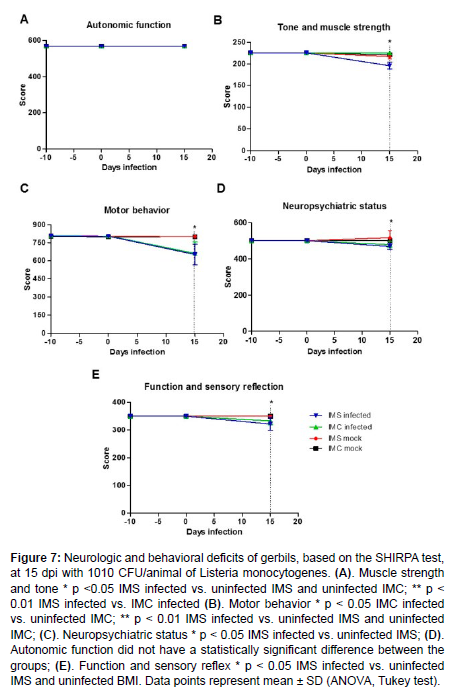
Among gerbils evaluated by histopathology only one IMS infected gerbil at 15 dpi developed neurologic signs. This neurologically affected gerbil had the most extensive brain abscess among all experimentally infected gerbils. The SHIRPA test was employed in this study to evaluate behavioral and neurologic changes in gerbils infected with 1010 CFU/animal of L. monocytogenes at -10, 0, and 15 dpi [Figure 7]. There was no difference between the groups in the parameters related to the autonomic function. However, a significant reduction in the functional categories, namely muscle tone and strength, motor behaviour, neuropsychiatric state and function, and sensory reflex, was observed in infected IMS gerbils when compared to the other groups. These data demonstrated that infection with L. monocytogenes impaired neurologic functions of gerbils, which was aggravated by immunosuppression.
Figure 7: Neurologic and behavioral deficits of gerbils, based on the SHIRPA test, at 15 dpi with 1010 CFU/animal of Listeria monocytogenes. (A). Muscle strength and tone * p <0.05 IMS infected vs. uninfected IMS and uninfected IMC; ** p < 0.01 IMS infected vs. IMC infected (B). Motor behavior * p < 0.05 IMC infected vs. uninfected IMC; ** p < 0.01 IMS infected vs. uninfected IMS and uninfected IMC; (C). Neuropsychiatric status * p < 0.05 IMS infected vs. uninfected IMS; (D). Autonomic function did not have a statistically significant difference between the groups; (E). Function and sensory reflex * p < 0.05 IMS infected vs. uninfected IMS and uninfected BMI. Data points represent mean ± SD (ANOVA, Tukey test).
Biodistribution of radiolabeled listeria monocytogenes at early stages of infection
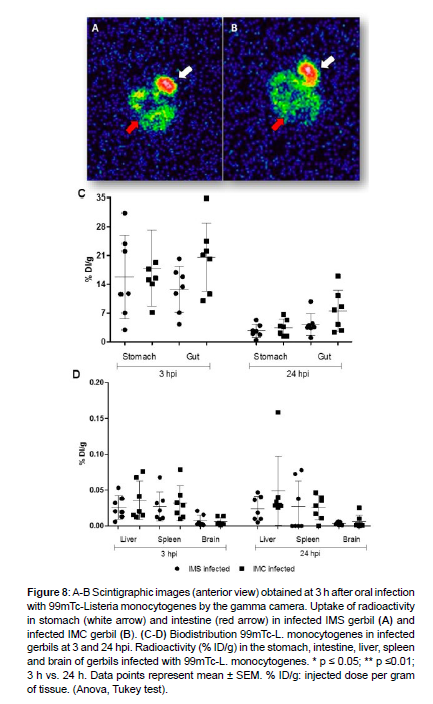
Previous studies employed labeling of bacteria with radioactive isotopes [23,33,34], but was not L. monocytogenes. Diniz et al. [23] labeled Escherichia coli with technetium-99m (99mTc) to investigate bacterial translocation in an experimental model of obstructive jaundice. 99mTc is the most widely used radiotracer in nuclear medicine because of its availability and ideal physical characteristics. For the first time, Labeling of L. monocytogenes with the 99mTc radioisotope in the presence of the reducing agent SnCl2.2H2O allowed an effective binding of the isotope to the bacteria. The labeling of L. monocytogenes with 99mTc was of the order of 86.2%, indicating an approximate content of 13.8% of radiochemical impurities; which is considered acceptable [35]. The binding of 99mTc atoms with the bacteria remained stable and viable for at least 24 h (85.7%).
To demonstrate the Biodistribution of 99mTc-L. Monocytogenes during the first hours post infection (hpi), radiolabeled bacteria was inoculated into gerbils via the oral route. Scintigraphic images indicated similar uptake when infected IMS were compared to infected IMC gerbils [Figure 8]. 99mTc-L. monocytogenes was detected in stomach and intestine at 3 hpi, demonstrating that L. monocytogenes localized in the digestive tract at the early stages after oral infection. There were not enough uptakes for the acquisition of the images in these same organs at 24 hpi, due to elimination of the bacteria through the feces. In addition, an ex vivo Biodistribution study was performed to determine the radioactivity in stomach, intestine, liver, spleen, and brain. The results demonstrated that there is a greater 99mTc-L. Monocytogenes uptake in stomach and intestine at 3 hpi, with marked decreased at 24 hpi, which was compatible to the scintigraphic images [Figure 8]. Furthermore, the radiolabeled bacteria had greater uptake in the liver and spleen, and lower uptake in brain, with no significant differences between groups at 3 and 24 hpi [Figure 8]. These results demonstrated that the radiolabeling of L. monocytogenes with 99mTc provided a suitable model for the study of bacterial spread to the CNS after oral infection, and that immunosuppression did not influence systemic bacterial spread at early stages of infection.
Figure 8: A-B Scintigraphic images (anterior view) obtained at 3 h after oral infection with 99mTc-Listeria monocytogenes by the gamma camera. Uptake of radioactivity in stomach (white arrow) and intestine (red arrow) in infected IMS gerbil (A) and infected IMC gerbil (B). (C-D) Biodistribution 99mTc-L. monocytogenes in infected gerbils at 3 and 24 hpi. Radioactivity (% ID/g) in the stomach, intestine, liver, spleen and brain of gerbils infected with 99mTc-L. monocytogenes. * p ≤ 0.05; ** p ≤0.01; 3 h vs. 24 h. Data points represent mean ± SEM. % ID/g: injected dose per gram of tissue. (Anova, Tukey test).
Discussion
This study demonstrated for the first time that orally L. monocytogenes-infected gerbils developed neurologic listeriosis characterized by bacterial colonization of CNS with tropism to the brainstem and rhomboencefalitis, and dexamethazone induced-immusuppressed resulting in evident neurologic deficits Although gerbils are laboratory animals naturally susceptible to listeriosis [36], there are just a few studies evaluating the CNS infection by L. monocytogenes in this species [13,14,17].
Additionally, this study was the first to demonstrate that bacterial spread to the CNS occurred just a few hours after oral infection with L. monocytogenes. Previous studies have demonstrated that intracranial [37] or intravenous [38] inoculations of L. monocytogenes result in infection of the CNS at 24 h after inoculation in mice. However, the earliest time point previously reported of bacteria detection in the CNS is 5 dpi after oral infection [39]. Previous studies were successful when using 99mTc-E. coli for evaluation of bacterial translocation of the intestinal mucosa to other systemic organs [40,41]. Therefore, we verified that 99mTc-L monocytogenes efficiently disseminated to the CNS at early time points following oral infection. Additionally, immunosuppression did not contribute to a higher dissemination of the bacterium to the CNS up to 24 hpi. Thus, impairment of immune system may influences in situ after spread bacteria colonization of systemic organs and brainstem.
In this study we demonstrated that high bacterial numbers (8-10 CFU log) of L. monocytogenes colonized the intestine and then disseminate and colonize the liver, spleen, and CNS in gerbils orally infected, which is in agreement with previous studies [14,17]. Some authors suggest that the high microvascular density of the brainstem contributes to the hematogenic infection of L. monocytogenes in this site (Oevermann et al., 2010, Berche, 1995; Altamira et al., 1999) [3,42,43]. However, an intra-axonal route through the cranial nerves should also be considered as a pathway for L. monocytogenes infection of the CNS [3,44-46]. Findings of the present study support the hypothesis that L. monocytogenes spreads to the brainstem through the trigeminal nerve. This is based on the finding of microabscesses predominantly near the trigeminal nerve tract in the brainstem. Ascending infection along the trigeminal nerve is thought to be important for Listeria pathogenesis in the brainstem [29], although other cranial nerves, including the abducent, glossopharyngeal, vagus, and accessory, may also allow the entry of L. monocytogenes [47]. L. monocytogenes can invade the oral mucosa and reach the nerve by small abrasions in ruminants, which usually develop rhomboencephalitis in cases of listeriosis [3]. In a murine model, L. monocytogenes induces romboencephalitis via the retrograde neuronal route when it was unilaterally inoculated into the facial muscle or directly into peripheral parts of the facial nerve [11,45,48]. The mechanism of CNS invasion by the neuronal pathway in humans is poorly supported because most food products related to human listeriosis such as salads, meat, milk and cheese are not abrasive, and therefore do not favor transposition of bacteria to deeper tissues [3,48]. However, it has been proposed that gingivitis or dental caries may contribute to neuronal Listeria invasion in humans [49]. Interestingly, reports of Listeria-induced brainstem encephalitis have been associated with dental surgery in human patients, which supports this hypothesis [47,50,51]. Furthermore, infected patients who develop signs of unilateral cranial nerve paralysis due to neuritis subsequently developed abscesses in the brainstem [52,53]. It is worth noting that rhomboencephalitis is a less common form, corresponding to 1 to 10% of the cases of human neurologic listeriosis, but it is the most serious form when compared to meningoencephalitis that accounts for 90% of the cases [54-56].
This oral infection model of gerbil rhomboencephalitis was characterized by microabscesses containing neutrophils, necrosis and bacteria, as well as gliosis and perivascular mononuclear inflammatory infiltrate. These changes are similar to lesions described in gerbil infected with L. monocytogenes through the middle ears [13], and to lesions commonly observed in cases of neurolisteriosis in ruminants [3]. By immunohistochemistry, we had shown that glial cells such as microglia and astrocytes are enlarged in infected brainstem, and lymphocytes are also present in the area adjacent to the lesion, as previously demonstrated in naturally infected sheep [57]. Microglia is considered a target cell for Listeria in the CNS and also important in controlling the spread of the pathogen and in the presence of cerebral homeostasis [58-60] Wang et al. [60] demonstrate in vitro and in vivo that microglia can release extracellular traps with the production of reactive cytosolic oxygen species to contribute in the control of Listeria infection in CNS. Astrocytes participate in repairing damaged parts of the brain. Astrocytes are activated in the brain of sheep naturally infected with L. monocytogenes and more GFAP expression is associated with greater severity of the injury [61]
Our study demonstrated that neurologic signs more evident in imunossupressed gerbils (i.e., motor incoordination, unilateral ataxia, paresis, and seizures) and anorexia may be associated with rhomboencephalitis,but several gerbils with mild inflammation in brainstem at 15 dpi did not develop any evident clinical sign. Therefore, prolonged immunossupresion (for more than 25 days) in this gerbil model of infection may provide better experimental conditions to consistently assess clinical and neurologic changes.
Interestingly, immunodeficient gerbils were more susceptible to systemic infection with brainstem infection more consistent. Roulo et al 2014 [14] demonstrated that during gestation, considered immunssupreed status, also increased recovered of L. monocytogenes in brain of pregnant gerbils, but did not show if bacteria has tropism for any CNS region. The most important host defense against listeriosis is cell-mediated immunity. Therefore, individuals with immune dysfunction are particularly susceptible to L. monocytogenes infection [62]. Jaguezeski et al. 2020 [17] showed that leucocitosis occur in non-imunossupresed gerbil with subclinical listeriosis. In this study, infected immunosuppressed gerbils had leukopenia with lymphopenia, and increased susceptibility to infection with L. monocytogenesin in systemic sites. Immunosuppressive therapy reduces cell-mediated immunity to prevent rejection of organ transplantation, but also increases the risk of infection to bacteria in the CNS, especially L. monocytogenes [63]. Corticosteroid therapy may result in a decrease in the phagocytic functions of neutrophils and macrophages, as well as in the deactivation of monocytes, promoting greater susceptibility to infection [64].
Suppression of the immune system impairs may the control and elimination of bacteria, including in the CNS, favouring inflammatory lesions and neurologic changes. However, in this study, immunosuppressed or immunocompetent gerbils developed abscesses associated with consistent colonization of brainstem after oral infection with L. monocytogenes. Senay et al. 2020 [11] describe a model of rhomboencephalitis in mice infected with human brainstem isolate. There is no information about neurotriopism of the strain ATCC 7644 used in this study, originally isolated from human. Although the neurotropism of the may be important for the manifestation of experimental neurolisteirosis [11], host factors may also contribute to the manifestation of the disease in animal model [14]. In this study, we showed that although immunosuppression favors systemic infection, it did not seem to contribute significantly to the manifestation of rhomboencephalitis in gerbils, as observed in humans [65,66] and the murine model [11]. However, the neurological signs observed in were evidenced only in the immunosuppressed infected animals. How dexamethazone treatment influenced the clinical manifestation is not known. It is possible that immunosuppression may partially contributes to colonization and inflammation of the brainstem and, consequently, to behavioral changes.
Listeriosis in immunocompromised individuals may result in severe infections leading to meningitis and meningoencephalitis, with lethality rates reaching 25 to 30% [67,68]. History of corticotherapy is considered a risk factor for Listeria-associated meningitis in human pacients [69]. Neurologic listeriosis occurs in 5 to 10% of individuals undergoing organ transplant. Patients with AIDS are susceptible to infection with L. monocytogenes in the CNS due to the significant decrease in CD4+ cells at late stages of the disease [63]. On the other hand, rhombencephalitis, as seen in this study, is found in healthy people. Although less frequent, listerial encephalitis of the brainstem has a high mortality, and severe sequelae are frequently reported in survivors. [65,66]. Thus, with a better understanding of the clinical manifestation and development of brainstem damage, it is important to try to reduce mortality or minimize its sequelae.
In this study, it was demonstrated that infected immunocompromised gerbils had higher systemic bacterial colonization and morbidity associated to behavioral and neurologic changes, which were more intense than those observed in immunocompetent gerbils. Thus, here we described a suitable model for the study of neurologic listeriosis, including factors that may influence susceptibility in immunosuppressed individuals.
Acknowledgements
This work had financial support of FAPEMIG APQ-00764-16. VNC, SOAF, RLS, and TAP are supported by CNPq. We thank Dr. Geovanni Dantas Cassali, Laboratory of Comparative Pathology, Department of General Pathology, ICB-UFMG, for support immunohistochemistry analyses. All Animal procedures were approved by Committee for Ethical Use of Experimental Animals (CEUA protocol 38/2015) of UFMG, Brazil.
References
- Hoelzer K, Pouillot R, Dennis S (2012) Listeria monocytogenes growth dynamics on produce: a review of the available data for predictive modeling. Foodborne Pathog Dis 9: 661-673.
- Doganay M (2003) Listeriosis clinical presentation. FEMS Immunol Med Microbiol 35: 173-175.
- Oevermann A, Zurbriggen A, Vandevelde M (2010) Rhombencephalitis Caused by Listeria monocytogenes in Humans and Ruminants: A Zoonosis on the Rise? Interdiscip Perspect Infect Dis 632513.
- D'Orazio SE (2014) Animal models for oral transmission of Listeria monocytogenes. Front Cell Infect Microbiol 4: 15.
- Drevets DA, Bronze MS (2008) Listeria monocytogenes: epidemiology, human disease, and mechanisms of brain invasion. FEMS Immunol Med Microbiol 53: 151-165.
- Lecuit M (2007) Human listeriosis and animal models. Microbes Infect 9: 1216-1225.
- Hayashi T, Nagai S, Fujii H, Baba Y, Ikeda E, et al. (2009) Critical roles of NK and CD8+ T cells in central nervous system listeriosis. J Immunol 182: 6360-6368.
- Lecuit M, Vandormael-Pournin S, Lefort J, Huerre M, Gounon P, et al. (2001) transgenic model for listeriosis: role of internalin in crossing the intestinal barrier. Science 292: 1722-1725.
- Bou Ghanem EN, Jones GS, Myers-Morales T, Patil PD, Hidayatullah AN, et al.(2012) InlA promotes dissemination of Listeria monocytogenes to the mesenteric lymph nodes during food borne infection of mice. PLoS Pathog 8: e1003015.
- Bergmann S, Beard PM, Pasche B, Lienenklaus S, Weiss S, et al. (2013) Influence of internalin A murinisation on host resistance to orally acquired listeriosis in mice. BMC Microbiol 13: 90.
- Senay TE, Ferrell JL, Garrett FG, Albrecht TM, Cho J, et al. (2020) Neurotropic lineage III strains of Listeria monocytogenes disseminate to the brain without reaching high titer in the blood. mSphere 5: e00871-20.
- Disson O, Lecuit M (2013) Invitro and invivo models to study human listeriosis: mind the gap. Microbes Infect 15: 971-980.
- Blanot S, Joly MM, Vilde F, Jaubert F, Clement O, et al. (1997) A gerbil model for rhombencephalitis due to Listeria monocytogenes. Microb Pathog 23: 39-48.
- Roulo RM, Fishburn JD, Amosu M, Etchison AR, Smith MA, et al. (2014) Dose response of Listeria monocytogenes invasion, fetal morbidity, and fetal mortality after oral challenge in pregnant and nonpregnant Mongolian gerbils. Infect Immun 82: 4834-4841.
- Harris J, Paul O, Park SH, White SJ, Budachetri K, et al. (2019) Oxygen deprivation influences the survival of Listeria monocytogenes in gerbils. Transl Anim Sci 3: 102-112.
- Jaguezeski AM, Souza CF, Perin G, Reis JH, Gomes TMA, et al. (2019) Effect of free and nano-encapsulated curcumin on treatment and energetic metabolism of gerbils infected by Listeria monocytogenes. Microb Pathog 134: 103564.
- Jaguezeski AM, da Silva AS, Gomes TMA, Bottari NB, Lopes TF, et al. (2019) Experimental listeriosis: A study of purinergic and cholinergic inflammatory pathway. Vet Microbiol 241: 108528.
- Feng X, Wei C, Adam RD, Li Z, Lu S et al. (2010) Phylogenetic status of Pneumocystis from corticosteroid-treated gerbils. Sci China Life Sci 53: 1239-1246.
- Oliveira TS, Bull V, Furtini R, Drummond RSM, Costa EA, et al. (2016) Neurological diseases of cattle diagnosed by histopathology in Minas Gerais. Braz J Vet Pathol 9: 62-69.
- Huang B, Krafft PR, Ma Q, Rolland WB, Caner B, et al. (2012) Fibroblast growth factors preserve blood-brain barrier integrity through RhoA inhibition after intracerebral hemorrhage in mice. Neurobiol Dis 46: 204-214.
- Rogers DC, Peters J, Martin JE, Ball S, Nicholson SJ, et al. (2001) SHIRPA, a protocol for behavioral assessment: validation for longitudinal study of neurological dysfunction in mice. Neurosci Lett 306: 89-92.
- Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25: 402-408.
- Diniz SO, Resende BM, Nunan EA, Simal CJ, Cardoso VN et al. (1999) 99mTechnetium labelled Escherichia coli. Appl Radiat Isot 51: 33-36.
- Lindstrom NM, Moore DM, Zimmerman K, Smith SA (2015) Hematologic Assessment in Pet Rats, Mice, Hamsters, and Gerbils: Blood Sample Collection and Blood Cell Identification. Clin Lab Med 35: 629-640.
- Wang S, Peng L, Gai Z, Zhang L, Jong A, et al. (2016) Pathogenic Triad in Bacterial Meningitis: Pathogen Invasion, NF-κB Activation, and Leukocyte Transmigration that Occur at the Blood-Brain Barrier. Front Microbiol 7: 148.
- Yau B, Mitchell AJ, Too LK, Ball HJ, Hunt NH et al. (2016) Interferon-γ-Induced Nitric Oxide Synthase-2 Contributes to Blood/Brain Barrier Dysfunction and Acute Mortality in Experimental Streptococcus pneumoniae Meningitis. J Interferon Cytokine Res 36: 86-99.
- Sankowski R, Mader S, Valdés-Ferrer SI (2015) Systemic inflammation and the brain: novel roles of genetic, molecular, and environmental cues as drivers of neurodegeneration. Front Cell Neurosci 9: 28.
- Dussurget O, Bierne H, Cossart P (2014) The bacterial pathogen Listeria monocytogenes and the interferon family: type I, type II and type III interferons. Front Cell Infect Microbiol 4: 50.
- Jin Y, Dons L, Kristensson K, Rottenberg ME (2001) Neural route of cerebral Listeria monocytogenes murine infection: role of immune response mechanisms in controlling bacterial neuroinvasion. Infect Immun 69: 1093-1100.
- Drevets DA, Dillon MJ, Schawang JE, Stoner JA, Leenen PJ et al. (2010) IFN-gamma triggers CCR2-independent monocyte entry into the brain during systemic infection by virulent Listeria monocytogenes. Brain Behav Immun 24: 919-929.
- Kelly JP, Bancroft GJ (1996) Administration of interleukin-10 abolishes innate resistance to Listeria monocytogenes. Eur J Immunol 26: 356-364.
- Foulds KE, Rotte MJ, Seder RA (2006) IL-10 is required for optimal CD8 T cell memory following Listeria monocytogenes infection. J Immunol 177: 2565-2574.
- Ardehali R, Mohammad SF (1993) 111Indium labeling of microorganisms to facilitate the investigation of bacterial adhesion. J Biomed Mater Res 27: 269-275.
- Eaves-Pyles T, Wong HR, Alexander JW (2000) Sodium arsenite induces the stress response in the gut and decreases bacterial translocation in a burned mouse model with gut-derived sepsis. Shock 13: 314-319.
- Sampson CB (1996) Complications and difficulties in radiolabelling blood cells: a review. Nucl Med Commun 17: 648-658.
- Disson O, Nikitas G, Grayo S, Dussurget O, Cossart P, et al. (2009) Modeling human listeriosis in natural and genetically engineered animals. Nat Protoc 4: 799-810.
- Schlüter D, Buck C, Reiter S, Meyer T, Hof H, et al. (1999) Immune reactions to Listeria monocytogenes in the brain. Immunobiology 201: 188-195.
- Hardy JW, Levashova Z, Schmidt TL, Contag CH, Blankenberg FG et al. (2015) [99mTc]Annexin V-128 SPECT Monitoring of Splenic and Disseminated Listeriosis in Mice: a Model of Imaging Sepsis. Mol Imaging Biol 17: 345-354.
- Bou Ghanem EN, Myers-Morales T, Jones GS, D'Orazio SE (2013) Oral transmission of Listeria monocytogenes in mice via ingestion of contaminated food. J Vis Exp 75: e50381.
- Costa KA, Soares AD, Wanner SP, Santos Rd, Fernandes SO et al. (2014) L-arginine supplementation prevents increases in intestinal permeability and bacterial translocation in male Swiss mice subjected to physical exercise under environmental heat stress. J Nutr 144: 218-223.
- Soares AD, Costa KA, Wanner SP, Santos RG, Fernandes SO, et al. (2014) Dietary glutamine prevents the loss of intestinal barrier function and attenuates the increase in core body temperature induced by acute heat exposure. Br J Nutr 112: 1601-1610.
- Berche P (1995) Bacteremia is required for invasion of the murine central nervous system by Listeria monocytogenes. Microb Pathog 18: 323-336.
- Altimira J, Prats N, López S, Domingo M, Briones V, et al. (1999) Repeated oral dosing with Listeria monocytogenes in mice as a model of central nervous system listeriosis in man. J Comp Pathol 121: 117-125.
- Dons L, Weclewicz K, Jin Y, Bindseil E, Olsen JE, et al.(1999) Rat dorsal root ganglia neurons as a model for Listeria monocytogenes infections in culture. Med Microbiol Immunol 188: 15-21.
- Antal EA, Løberg EM, Bracht P, Melby KK, Maehlen J et al. (2001) Evidence for intraaxonal spread of Listeria monocytogenes from the periphery to the central nervous system. Brain Pathol 11: 432-438.
- Henke D, Rupp S, Gaschen V, Stoffel MH, Frey J, et al. (2015) Listeria monocytogenes spreads within the brain by actin-based intra-axonal migration. Infect Immun 83: 2409-2419.
- Castro A, Hernández OH, Uribe CS, Guerra A, Urueña P et al. (2013) Encefalitis del tallo cerebral y mielitis por Listeria monocytogenes [Brainstem encephalitis and myelitis due to Listeria monocytogenes: a case report and literature review]. Biomedica 33: 343-349.
- Disson O, Lecuit M (2012) Targeting of the central nervous system by Listeria monocytogenes. Virulence 3: 213-221.
- Roche SM, Gracieux P, Milohanic E, Albert I, Virlogeux-Payant I, et al. (2005) Investigation of specific substitutions in virulence genes characterizing phenotypic groups of low-virulence field strains of Listeria monocytogenes. Appl Environ Microbiol 71: 6039-6048.
- Drevets DA, Leenen PJ, Greenfield RA (2004) Invasion of the central nervous system by intracellular bacteria. Clin Microbiol Rev 17: 323-3247.
- Kennard C, Howard AJ, Scholtz C, Swash M (1979) Infection of the brainstem by Listeria monocytogenes. J Neurol Neurosurg Psychiatry 42: 931-933.
- Medina-Flores R, Germanwala A, Molina JT, Meltzer CC, Wiley CA et al. (2004) October 2003: a 59-year-old woman with sudden onset of diplopia. Listerial rhombencephalitis. Brain Pathol 14: 225-226.
- Armstrong RW, Fung PC (1993) Brainstem encephalitis rhombencephalitis due to Listeria monocytogenes: case report and review. Clin Infect Dis 16: 689-702.
- Uldry PA, Kuntzer T, Bogousslavsky J, Regli F, Miklossy J, et al. (1993) Early symptoms and outcome of Listeria monocytogenes rhombencephalitis: 14 adult cases. J Neurol 240: 235-242.
- McLauchlin J (1990) Human listeriosis in Britain, 1967-85, a summary of 722 cases. 2. Listeriosis in non-pregnant individuals, a changing pattern of infection and seasonal incidence. Epidemiol Infect 104: 191-201.
- Bazooyar B (2015) Rhombencephalitis by Listeria Monocytogenes in Two Diabetic Patients. Arch Iran Med 18: 613-615.
- Krueger N, Low C, Donachie W (1995) Phenotypic characterization of the cells of the inflammatory response in ovine encephalitic listeriosis. J Comp Pathol 113: 263-275.
- Virna S, Deckert M, Lütjen S, Soltek S, Foulds KE et al. (2006) TNF is important for pathogen control and limits brain damage in murine cerebral listeriosis. J Immunol 177: 3972-3982.
- Frande-Cabanes E, Fernandez-Prieto L, Calderon-Gonzalez R, Rodríguez-Del Río E, Yañez-Diaz S, et al. (2014) Dissociation of innate immune responses in microglia infected with Listeria monocytogenes. Glia 62: 233-246.
- Wang C, Wang Y, Shi X, Tang X, Cheng W, et al. (2019) The TRAPs From Microglial Vesicles Protect Against Listeria Infection in the CNS. Front Cell Neurosci 13: 199.
- Özyildiz Z, Dinçel GD, Terzi OS, Özsoy SY, Kul O, et al. ( 2018) Immunohistochemical investigation of the damage to and repair of myelin, and astrocyte activity in small ruminants resulting from with natural meningoencephalitic listeriosis Ankara Üniv Vet Fak Derg 65: 283-290.
- Hernandez-Milian A, Payeras-Cifre A (2014) What is new in listeriosis? Biomed Res Int 358051.
- Zunt JR (2002) Central nervous system infection during immunosuppression. Neurol Clin 20: 1-22.
- Roquilly A, Lejus C, Asehnoune K (2012) Agressions cérébrales, immunité et infections [Brain injury, immunity and infections]. Ann Fr Anesth Reanim 31: 97-100.
- Karlsson WK, Harboe ZB, Roed C, Monrad JB, Lindelof M, et al (2017) Early trigeminal nerve involvement in Listeria monocytogenes rhombencephalitis: case series and systematic review. J Neurol 264: 1875-1884.
- Wei P, Bao R, Fan Y (2020) Brainstem Encephalitis Caused by Listeria monocytogenes. Pathogens 9: 715.
- Hamon M, Bierne H, Cossart P (2006) Listeria monocytogenes: a multifaceted model. Nat Rev Microbiol 4: 423-434.
- Ramaswamy V, Cresence VM, Rejitha JS, Lekshmi MU, Dharsana KS, et al (2007) Listeria--review of epidemiology and pathogenesis. J Microbiol Immunol Infect 40: 4-13.
- Lim S, Chung DR, Kim YS, Sohn KM, Kang SJ, et al. (2017) Predictive risk factors for Listeria monocytogenes meningitis compared to pneumococcal meningitis: a multicenter case-control study. Infection 45: 67-74.
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Citation: Gomes AG, et al. (2024) Development of an Experimental Model for Neurolisteriosis: Oral Infection with Listeria Monocytogenes Causes Brainstem Lesions and Impairment in Gerbils. J Neuroinfect Dis 15: 512. DOI: 10.4172/2314-7326.1000512
Copyright: © 2024 Gomes AG, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 1333
- [From(publication date): 0-2024 - Apr 04, 2025]
- Breakdown by view type
- HTML page views: 1104
- PDF downloads: 229