Cinnamon and Ginger Combined Extract Improved Diabetic Induced Developmental Defects of Inner Ear and Teeth of Rat Fetuses
Received: 17-Apr-2019 / Accepted Date: 29-Apr-2019 / Published Date: 06-May-2019
Abstract
Gestational diabetes is a public health problem associated with the development of birth defects, which encourage women to consume traditional medicine to improve the sharp rise of blood glucose level and maintain more safe gestation. The present work assess two main aspects, first the diabetic related retarding growth of inner ear and teeth formation and the second the role of one of the common traditional herbs during gestation, combined cinnamonginger extract searching for their safety and actual role in diabetic improvement. Twenty pregnant Wistar rats used and categorized in four groups: control, cinnamon-ginger extract, diabetic and diabetic and cinnamon-ginger mixture extract. Diabetes was induced at the 5th day of gestation using streptozotocin (60 mg/kg body weight citrate buffer pH 4.6 plus 100 mg nicotinamide/kg body weight, single dose). At 14th day of gestation, pregnant rats were euthanized by diethyl ether, sacrificed and their blood was collected and their femoral bone, islet of Langerhans and fetuses were separated. Decalcified femur, islets of Langerhans and head regions fetuses were fixed in 10% phosphate buffered formalin pH 7.4 and processed for histological investigation. 5 μm thick serial histological sections were cut and stained with hematoxylin eosin and immunohistochemically stained with caspase 3. Fetuses of diabetic mothers exhibited delayment growth of inner ear and mandibular tooth formation. The apparent increase of cell death in sensory cells lining the semicircular canal of the inner ear and dentine, enamel, stellate reticulum and dental papilla of teeth were detected post-caspase immunohistochemical staining. These were confirmed by alterations of glucose level, LDL and triglycerides and deterioration of maternal B cells and femoral epiphyseal cartilage. Daily oral administration of cinnamon-ginger watery extract (200 mg/kg body weight) from 5th day of gestation till 14th day improved the biochemical criteria of diabetes plus restoring almost the histological and immunohistochemical picture of both inner ear and tooth formation. These were explained by improvement of B-cells, maternal epiphysis and marked improvement of biochemical criteria of diabetes. The authors finally attributed the improvement of cinnamon-mixture extract to diabetic mother organs and fetal organs to the observed decrease of cell death, improvement of histological picture and amelioration of assessed biochemical markers. All of these may be some bioactive components of the plant extract which showed a potent antioxidant activity.
Keywords: Diabetes; Cinnamon; Ginger; Inner ear; Tooth development
Introduction
The Diabetes Mellitus (DM) is widely increased among population reached up to 382 million in 2014 [1]. International diabetes federation reported that 592 million (1/10 persons) developed DM by 2035 [2]. Type 2 diabetes impaired the function of B-cells leading to elevated blood sugar level and facilitated impairment of insulin secretion [3]. The 70% of 400 diabetic patients were found to develop dyslipidemia associated with cardiovascular disease and increased mortality [4,5]. Gestational diabetes reached an incidence of 16.9%, with average of ~21 million live births in 2013 [6]. Diabetes was found to retard bone growth and bone wound healing, damage of cartilage bone lining cells [7-9]. Hyperglycemia was found to inhibit osteoblast differentiation, impair deposition of osteoplastic calcium and bone growth [10,11]. In previous works, maternal diabetes has been interfered with birth growth defects in the experimental animals and interferes with organs growth defects [12-15]. Yet there is not available work concerned the influence of diabetes on the development defects of inner ear and tooth formation during in utero life [16,17].
At the same time, the wide use of the herbal medicine during pregnancy is of critical importance. Investigating 9,459 women from 23 countries revealed that 28.9% of them participating the use of herbal medicines in pregnancy such as Ginger, cranberry, valerian and raspberry [18]. Cinnamon and ginger showed anti-diabetic activity and exerted a potent effect against oxidative stress of diabetes [19-21].
The present study aimed to illustrate the aspects of intrauterine diabetes on the development of inner ear and mandibular teeth of 14th day old fetuses and the capacity of improvement cinnamon-ginger extract supplementation during gestation period.
Methodology
Applied dose of cinnamon and ginger watery extract
A known weight of dried cinnamon (Cinnamomum spp) and ginger (Zingiber officinale) were mixed thoroughly and freshly extracted with hot water, filtrate and allow to standing at room temperature. Each pregnant received 0.5 cc containing 200 mg of the extract materials per kg body weight. Each dose was daily orally administered from 6th day of gestation till 14th day of gestation.
Induction of diabetes
Experimental type 2 diabetes mellitus was induced in all the rats by a single intraperitoneal injection of streptozotocin (60 mg/kg) in citrate buffer (0.05 M) (pH 4.5) and 100 mg/kg ncotinamide [22]. Hyperglycemia was verified by measuring the blood glucose within 240-280 mg/dl were selected for the study.
Experimental animal work
Twenty fertile male and virgin female of albino rats (Rattus norvegicus) (at a ratio of 1 male to 3 females) weighing approximately 200 g body weight, were obtained from Hellwan Breeding Farm, Ministry of Health, Egypt and used for experimentation. They were housed in good ventilation on a 12 h light and dark cycle. Females were mated (1 male/3 females) overnight and zero dates of gestation were determined the next morning by observing the sperm in vaginal smear. The pregnant were arranged into four groups (n=4 per each), control, ginger watery extract, diabetes, diabetes and ginger extract. Experimental work was carried out from 6th day of gestation till 14th day of intrauterine growth. Free excess food and water was allowed ad Libitum. At the end of treatment, they were sacrificed by light diethyl ether anesthesia and dissected at 16th day prenatal. Blood was collected, allowed to coagulate and centrifuged. Serum was separated and kept in deep freezer -20ºC. Also, femur and pancreas of mother and head regions of their 14th day old fetuses were separated and fixed in 10% phosphate buffered formalin (pH 7.4) and processed for following investigations.
Biochemical investigations: Serum levels of Total Cholesterol (TC), Triglycerides (TG) and High Density Lipoproteins (HDL) were determined [23-25]. In case of Low Density Lipoproteins (LDL), it was calculated from the total concentrations of cholesterol (TC), HDLcholesterol and triglycerides according to Friedewald et al. [26]. The glucose regularly measured by blood glucometers one touch ultra (Life Scan Milipitas, CA, USA).
Histological investigation: Fourteen day old fetuses of the studied groups were separated and immediately fixed in 10% phosphate buffered formalin (pH 7.4), dehydrated in ascending grades of ethyl alcohol, cleared in toluene and mounted in molten paraplast at 58- 62ºC. Formalin fixed femur bone was subjected for decalcification by 5% EDTA. Serial five μm thick histological sections of maternal femur and islets and inner ear and mandible of the studied embryos were stained with hematoxylin and eosin and investigated under a bright field light microscope.
Immunohistochemistry for caspase 3
Deparifinized histological 5 μm thick sections of both inner ear and mandibular teeth of 14th day old fetuses were cut and mounted onto super frost plus glass slides (Fisher Thermo Scientific, Nepean, Ontario, Canada). The tissue sections were retained at normal room temperature and processed for antigen retrieval by digestion in 0.05% trypsin (pH 7.8) for 15 min at 37ºC and incubated with antibodies against anti-casp 3, dilution 1:100 (ThermoFisher Scientific, Fremont, CA, USA) for overnight at 4ºC. Then treated with a horseradish peroxidase streptavidin detection system (Dako), followed with DAB plus Chromagen to detect the immunoactivity by counterstaining with hematoxylin (Sigma). Sections incubated with 1% non-immune serum Phosphate Buffer Solution (PBS) solution served as negative controls. Specimens were observed with a Leica BM-5000 microscope (Leica Microsystems, Wetzlar, Germany) and photographed.
Results
Biochemistry of mothers
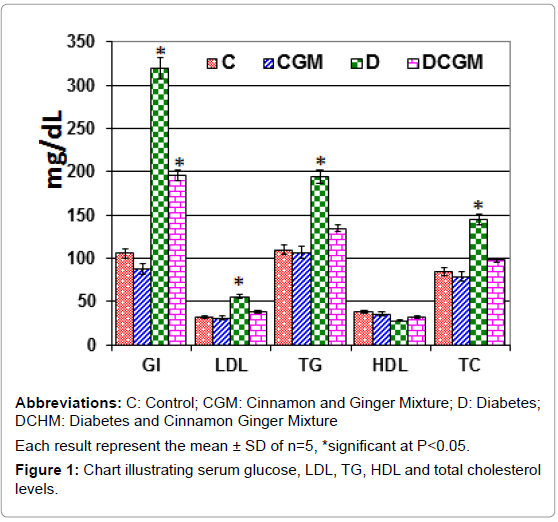
From Figure 1, induced diabetes significantly increased serum triglycerides, LDL, HDL, total cholesterol and glucose level of mothers at 14th day of gestation compared to the control. However, daily oral administrations of watery extract of combined cinnamon and ginger extract improved the biochemical criteria but were still not matched with the control levels.
Maternal organs
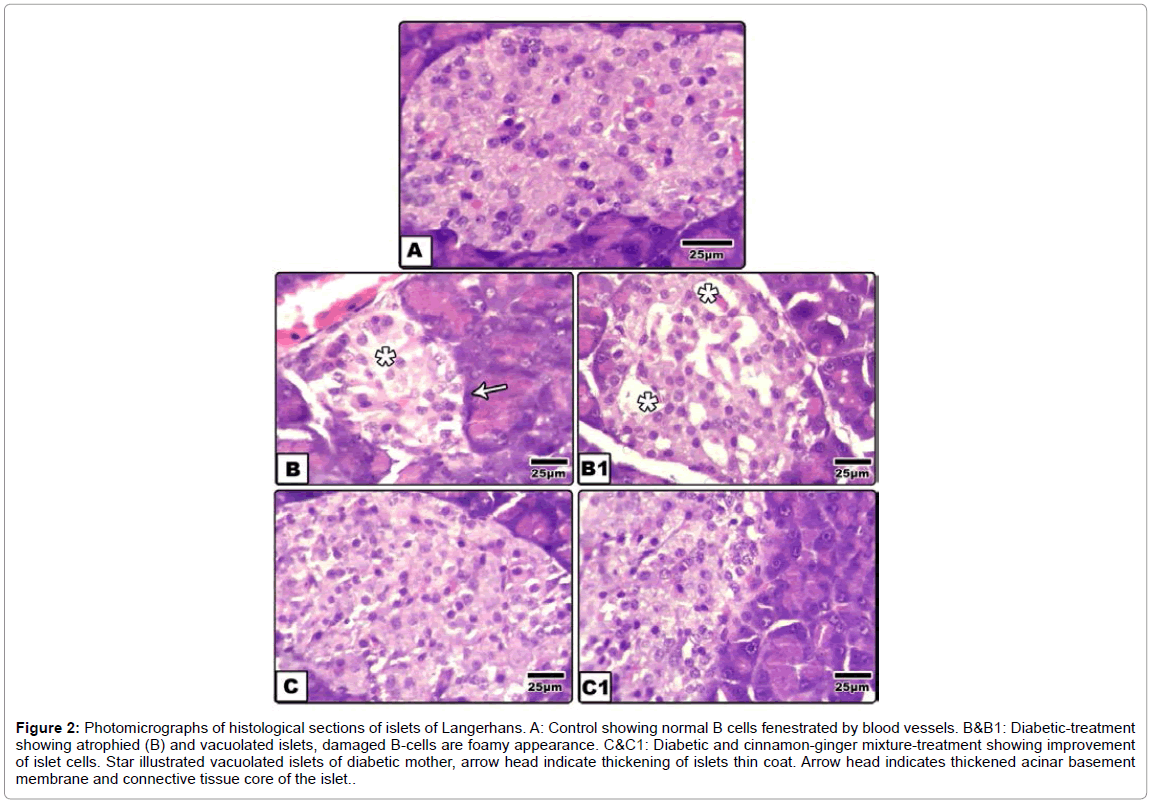
Islets of langerhans: Control islet illustrated the endocrine part of the pancreas. The islet take round-shaped structure and unsheathed by a thin collagenous fibrils and contained masses of polygonal epithelial cells, faintly stained compared to the exocrine region and infiltrated by a thin blood capillary. Different types of endocrine secreting cells, are detected such as α and β cells. Β cells occupied the deepest region and less distinguished from the other types by hematoxylin and eosin stain (Figure 2A).
Figure 2:Photomicrographs of histological sections of islets of Langerhans. A: Control showing normal B cells fenestrated by blood vessels. B&B1: Diabetic-treatment showing atrophied (B) and vacuolated islets, damaged B-cells are foamy appearance. C&C1: Diabetic and cinnamon-ginger mixture-treatment showing improvement of islet cells. Star illustrated vacuolated islets of diabetic mother, arrow head indicate thickening of islets thin coat. Arrow head indicates thickened acinar basement membrane and connective tissue core of the islet.
In diabetic group, the islet cells lacked normal integrity and many of them are degenerated and appeared foamy-like structure. Vacuoles of varying sizes were infiltrated extra-cellularly. The outline coat of the islets become either loosely separated from exocrine pancreatic acinar cells or thickened. Cinnamon-ginger extract-treatment to diabetic mother showed restored almost the normal characteristic structure compared with both diabetic–treatment and control (Figures 2B-2C1).
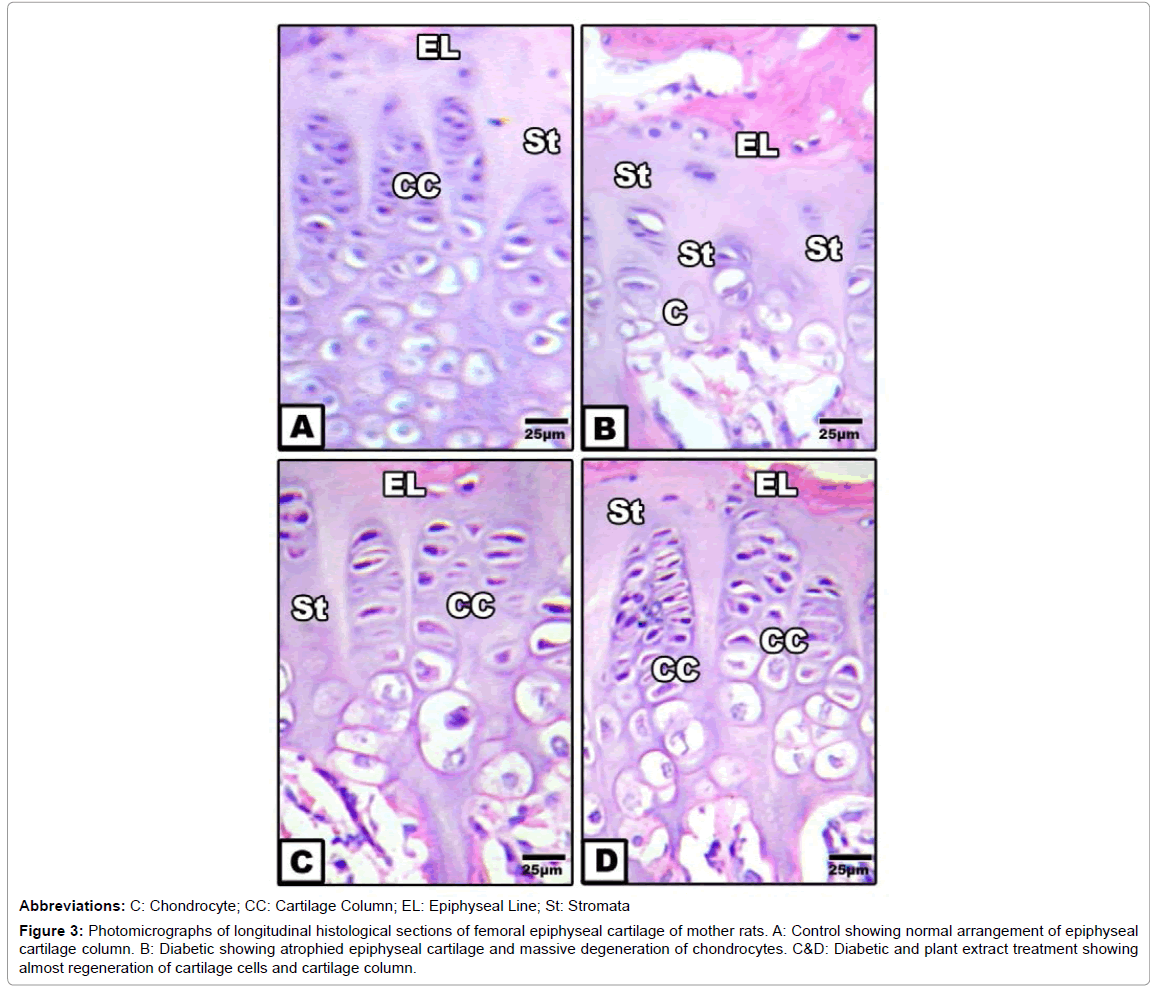
Femoral epiphyseal cartilage: Control epiphyseal cartilage showed regular arrangement of cartilage column with vesicular differentiation of chondrocytes within their lacunae. The matrix is comparatively reduced. Cinnamon and ginger extract exhibited similar findings as in control. On the other hand, diabetic-treatment possessed comparative atrophy of the epiphyseal cartilage associated with marked loss of chondrocytes. Few numbers of chondrocytes were distributed in haphazard fashion in widened hyaline stromal. Epiphyseal line is irregularly outlined. In diabetic mother daily orally administered cinnamon-ginger extract exhibited amelioration characterized by regeneration of cartilage cells which organized in newly formed cartilage column, but less comparatively decreased from the control (Figures 3A-3D).
Figure 3:Photomicrographs of longitudinal histological sections of femoral epiphyseal cartilage of mother rats. A: Control showing normal arrangement of epiphyseal cartilage column. B: Diabetic showing atrophied epiphyseal cartilage and massive degeneration of chondrocytes. C&D: Diabetic and plant extract treatment showing almost regeneration of cartilage cells and cartilage column.
Intra-uterine growth of teeth and inner ear of 14th day old fetuses
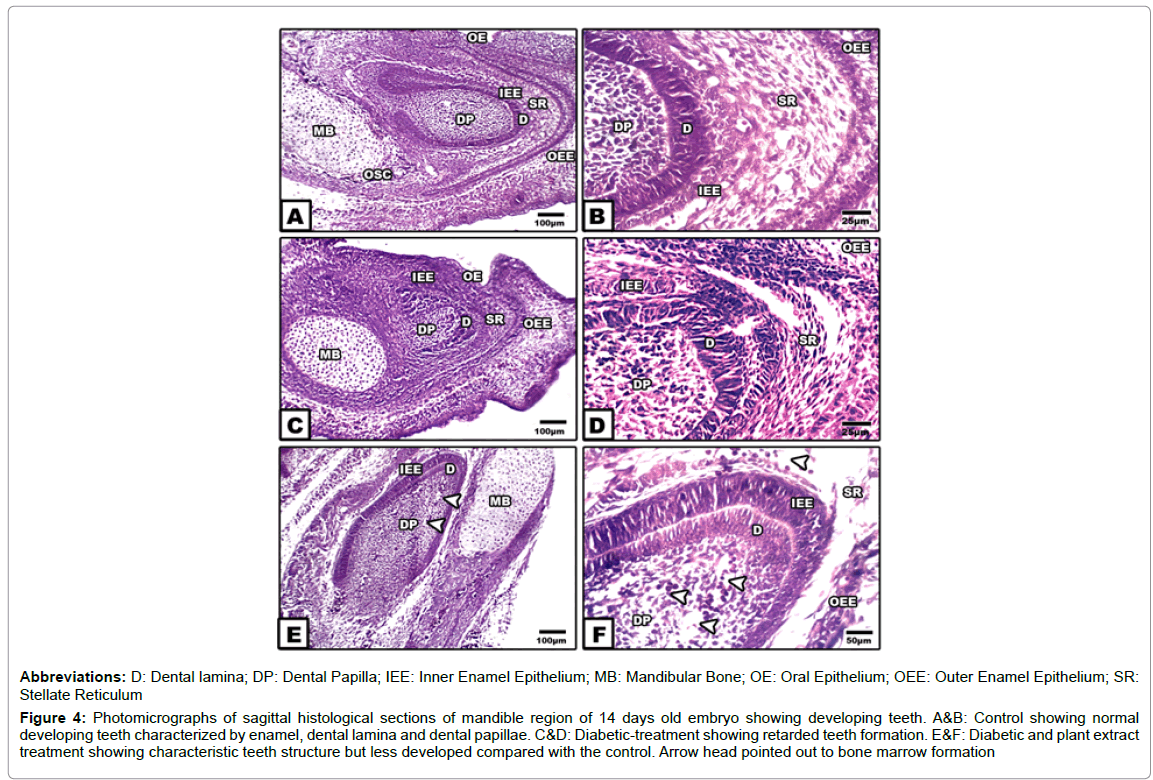
Teeth development: The primordial teeth structure is early differentiated from the neural crest mesenchyma cells and developed into the early primordial contact with the mandibular formation. In control and cinnamon-ginger extract treatment, odontogenesis make their first formation by a thickened band of cells acquiring the dental papilla which is differentiated into odontoblasts and constructing the primordial teeth structure. Early dentin formation enhanced the overlying inner enamel epithelial cells to develop into ameloblasts and forming the future the enamel matrix. At the peripheral region of the dentin, the enamel epithelium acquired a cuboidal-shaped structure and differentiates into inner and outer one. In between both layers of enamel epithelium, the stellate reticulum are differentiated and are composed of stellate mesenchyma cells. At the cervical region the root developed via conjugation between the dentine and the outer and inner enamel epithelium forming the cervical loop, the hertwig’s epithelial root sheath. The dental papilla in between the dentine is composed of ectomesenchymal cells infiltrated by blood capillaries (Figures 4A and 4B).
In fetuses of diabetic mother, there was a detected growth defects in teeth formation, beside ill-differentiated enamel layers, dentine and stellate reticulum. The dentine is less dense and infiltrated by necrotic zones. Hypoplasia and abnormal configuration of the inner enamel epithelium were observed. Eosinophilia and necrotic patches were observed on their building epithelium. The stellate reticulum become swollen and infiltrated by widened vacuoles. The dentine was comparative less thickened and exhibited damaging of some odentoblast cells. The dental pulp epithelium showed a widespread of eosinophilic epithelium manifested cell death (Figures 4C and 4D).
Figure 4:Photomicrographs of sagittal histological sections of mandible region of 14 days old embryo showing developing teeth. A&B: Control showing normal developing teeth characterized by enamel, dental lamina and dental papillae. C&D: Diabetic-treatment showing retarded teeth formation. E&F: Diabetic and plant extract treatment showing characteristic teeth structure but less developed compared with the control. Arrow head pointed out to bone marrow formation.
In fetuses of diabetic mothers and supplemented cinnamon and ginger extract, there was a considerable improvement of the tooth structure, almost similar to the control. The different tooth layers are markedly distinguished and normal (Figures 4E and 4F).
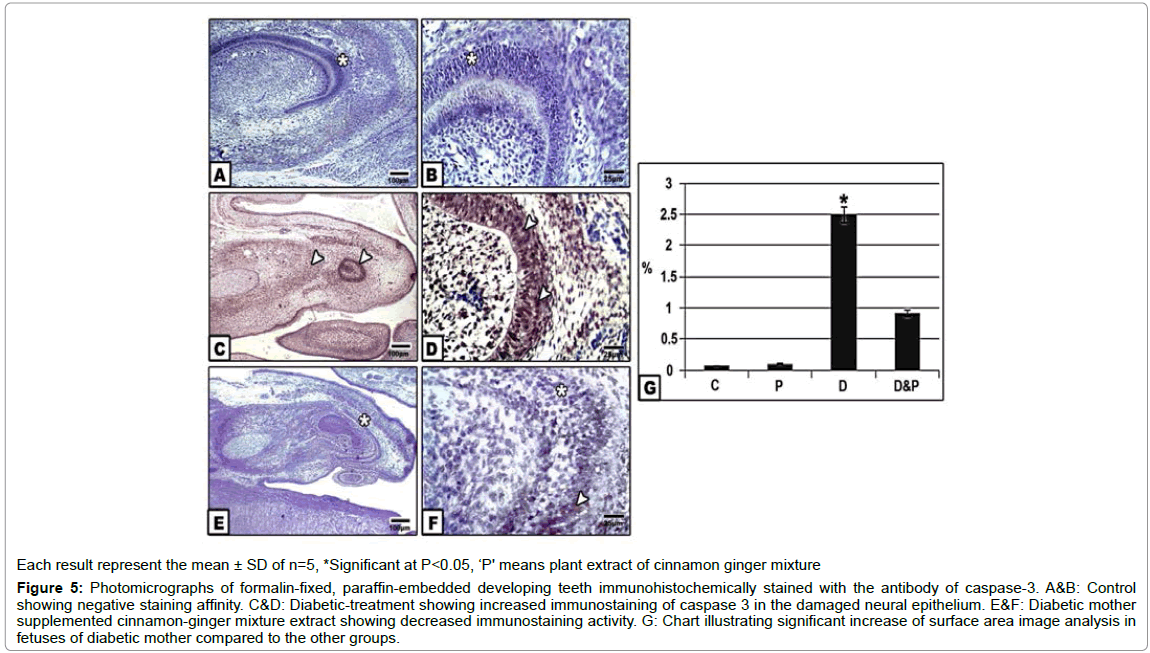
Following immunohistochemically stained with caspase 3 revealed the increased dark-brown staining affinity in dental papillae, dentine , inner enamel epithelium and stellate reticular cells of fetuses of diabetic group compared to the control and other groups. Image analysis of increased immunostaining active area with caspase 3 was detected in those of diabetic mothers in comparison with the other groups (Figures 5A-5G).
Figure 5:Photomicrographs of formalin-fixed, paraffin-embedded developing teeth immunohistochemically stained with the antibody of caspase-3. A&B: Control showing negative staining affinity. C&D: Diabetic-treatment showing increased immunostaining of caspase 3 in the damaged neural epithelium. E&F: Diabetic mother supplemented cinnamon-ginger mixture extract showing decreased immunostaining activity. G: Chart illustrating significant increase of surface area image analysis in fetuses of diabetic mother compared to the other groups.
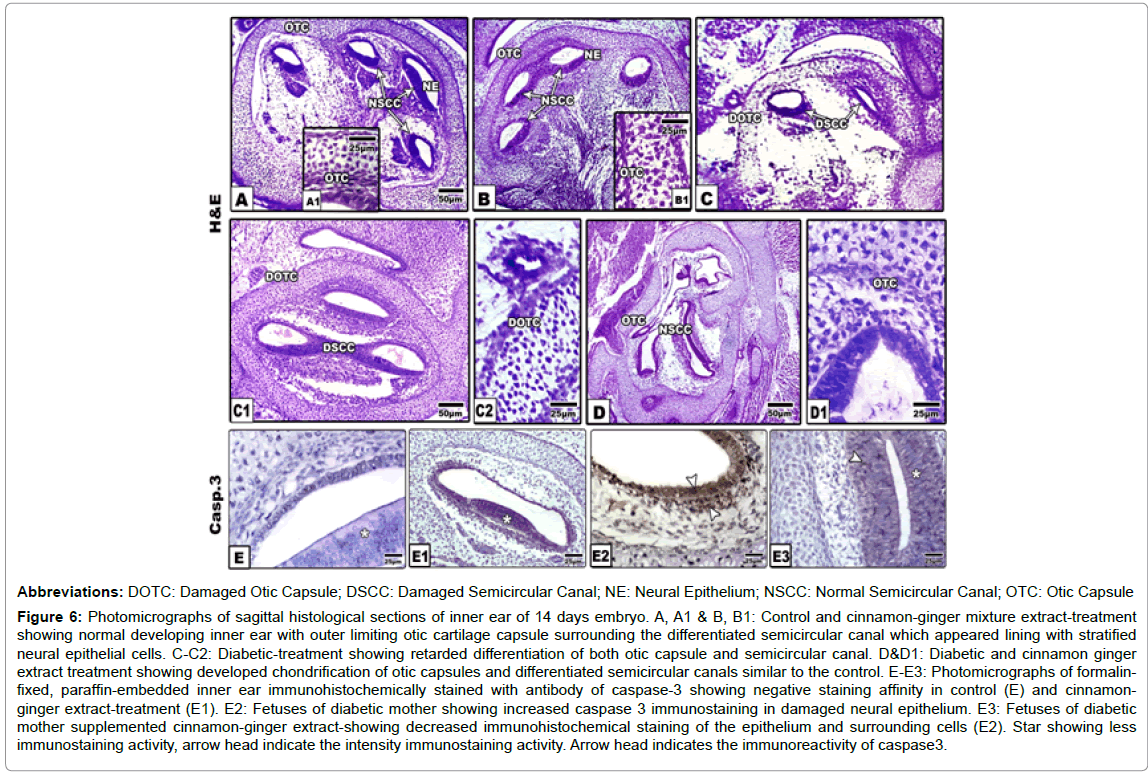
Inner ear development: The inner ear develops from pre-placodal region ectodermal zone around the peripheral region of the neural tube close to the rhomboencephalon. In both control and cinnamonginger extract-treatment, the inner ear is composed of well-developed otic capsule located inside the temporal region, on each side of the base region of the skull. The otic capsule exhibited a high degree of chondrification. It is composed of avascular hyaline cartilaginous cells embedded in hyaline matrix. The otic capsule surround by the perichondrial sheath composed of loose connective tissue and chondroblast progenitor cells. The otic capsule surrounded three welldefined horizontal ellipsoid-shaped structures, the semicircular canals. Each one is characterized by the presence of cristae in the ampullary area (crista ampullaris). The inner side of each of the semicircular canal is lined with a pseudo-stratified epithelium enclosed in between the hair-cells and resting on basal lamina (Figures 6A-6B1).
Figure 6:Photomicrographs of sagittal histological sections of inner ear of 14 days embryo. A, A1 & B, B1: Control and cinnamon-ginger mixture extract-treatment showing normal developing inner ear with outer limiting otic cartilage capsule surrounding the differentiated semicircular canal which appeared lining with stratified neural epithelial cells. C-C2: Diabetic-treatment showing retarded differentiation of both otic capsule and semicircular canal. D&D1: Diabetic and cinnamon ginger extract treatment showing developed chondrification of otic capsules and differentiated semicircular canals similar to the control. E-E3: Photomicrographs of formalinfixed, paraffin-embedded inner ear immunohistochemically stained with antibody of caspase-3 showing negative staining affinity in control (E) and cinnamonginger extract-treatment (E1). E2: Fetuses of diabetic mother showing increased caspase 3 immunostaining in damaged neural epithelium. E3: Fetuses of diabetic mother supplemented cinnamon-ginger extract-showing decreased immunohistochemical staining of the epithelium and surrounding cells (E2). Star showing less immunostaining activity, arrow head indicate the intensity immunostaining activity. Arrow head indicates the immunoreactivity of caspase3.
In fetuses of diabetic group, there was a detected gross defects of the inner ear explained by comparatively atrophy, delayed chondrification of otic capsule and swollen semicircular canal with ill-defined inner lining layer. Experimental diabetic group supplemented combined cinnamon and ginger extract exhibited improvement of the inner ear organization structure, characterized by a chondrified otic capsule and well defined semicircular canals (Figures 6C-6D1).
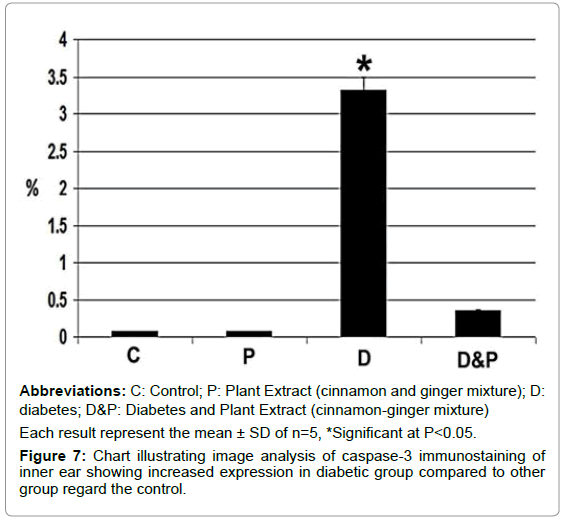
Concerning immunohistochemical staining with cysteine-aspartic acid protease 3 (caspase 3), there was a detected overexpression of apoptic marker in the neural epithelial cells lining the semicircular canal of diabetic group in comparison with control and those of diabetic mother supplemented cinnamon-ginger mixture extract. The immunohistochemical reaction was comparatively decreased in the neural lining cells and their surroundings (Figures E2 and E3).
Image analysis revealed increased regional immunohistochemical staining activity of caspase 3 in inner ear of fetus of diabetic mother compared to other groups (Figure 7).
Discussion and Conclusion
The inner ear is a complex sensory organ responsible for balance and sound detection. It is responsible for the senses of hearing, balance and detection of acceleration. During the fetal period, the otic capsule follows an outward-inward developmental pattern with a progressive thickening of the highly porous external cortex while the inner cortical bone remains thinner. The present findings revealed that 14th day old fetuses of diabetic mother rats exhibited delayement and atrophy of the otic capsule of the inner ear associated with disorganized the semicircular canal and their lining epithelium compared to the control. Increased apoptic neuronal cells lining the semicircular canal were explained by overexpression of caspase 3.
The present findings agree with the work of many authors although all the work done on adult organs. Diabetes induced auditory hair cell loss, hearing threshold shifts and synaptopathies in human and mice [27-29]. Tsuda et al. reported reduction of the capillary lumens in the cochlea stria vascularis associated with wall of blood capillaries and decreased blood flow to the inner ear [30]. Also, it is reported that diabetes was associated reduction of type I and type II vestibular hair cell, in the saccules expressed as arteriole thickening leading to damage of the vestibular sensory epithelia [31].
Taking in consideration that the semicircular canal is lined with the sensory neuronal cells. The mechanism of hyperglycemia associated cell damage was explained by many authors. Dewanjee et al., reported that hyperglycemia, induced oxidative stress in neurons, resulted from activated polyol pathway, hexosamine pathway, PKCs signaling, AGEs pathway and PARP pathway [32].
Also, from the present study, fetuses of diabetic mother exhibited defects in histogenesis of teeth formation of 14 day-old fetuses characterized by swollen stellate reticulum infiltrated by widened vacuoles, damaged odentoblast cells and delayed formation and deterioration of may patches of enamel body.
The present findings agree with the work of Silva-Sousa et al., in neonates of diabetic mother rats [33]. The authors reported structural defects of the enamel characterized by elliptical nuclei of the ameloblasts associated with increase of the interstitial area of the stellate reticulum. It is known that tooth formation is an excellent model of epithelial and mesenchymal tissues interactions illustrating morphogenesis and cytodifferentiations.
Hyperglycemia and gestational diabetes have been associated with increased birth defects and congenital malformations and in turn reflected on dental enamel growth leading to enamel hypoplasia in the deciduous teeth of children of diabetic mothers and neonate of diabetic rats [33-37]. Tooth eruption and mineralization were delayed in offspring of hyperglycemic women and animal models [38].
On the other hand, fetuses of diabetic mother supplemented cinnamon and ginger extract resulted in amelioration of the chondrification of otic capsules and differentiation of the semicircular canal and its lining sensory cells. At the same time restored the almost original structure of tooth formation with approximately architecture picture of the control. Cinnamon and ginger extract improve the B cells of islet Langerhans which intern restore the almost original glucose level and improve the increased average of LDL, triglycerides and total cholesterol. The anti-diabetic potential of cinnamon was attributed to its rich in procyanidins and catechins and antioxidant activities which protect pancreatic beta cells from damage and reduce oxidative stress/ inflammation and scavenge free radicals [39,41].
Also, Ginger contained bioactive components such as vallinoids, viz. gingerol and paradol, shogaols, zingerone and galanals A and B which improve diabetes in patients and experimental animals as well as possesses a potent scavenging of free radicals in diabetic rats [21,42-46].
The author finally concluded that cinnamon and ginger extract is promising as anti-diabetic and improve the growth of developing body organs such as inner ear and tooth formation.
References
- Wild S, Roglic G, Green A, Sicree R, King H (2004) Globalprevalenceof diabetes: Estimates for theyear2000andprojectionsfor2030. Diabetes Care 27: 1047-1053.
- Aguiree F, Brown A, Cho NH, Dahlquist G, Dodd S, et al. (2013)Diabetes atlas.
- Al-GoblanAS,Al-Alfi MA,Khan MZ (2014) Mechanism linking diabetes mellitus and obesity. Diabetes Metab Syndr Obes7: 587-591.
- Saed L,Deihim Z,Naghshbandi MK,Rajabnia M,Naleini SN (2019) Cardiovascular events in patients with over 10 years history of type 2 diabetes mellitus. Diabetes Metab Syndr 13: 68-72.
- Rivellese AA, Riccardi G, Vaccaro O (2010) Cardiovascular risk in women with diabetes.Nutr Metab Cardiovasc Dis20: 474-480.
- GuariguataL,Linnenkamp U,Beagley J,Whiting DR,Cho NH (2014) Global estimates of the prevalence of hyperglycaemia in pregnancy. Diabetes Res Clin Pract103: 176-185.
- Lu H, Kraut D, Gerstenfeld LC, Graves DT (2003) Diabetes interferes with the bone formation by affecting the expression of transcription factors that regulate osteoblast differentiation. Endocrinology 144: 346-352.
- He H, Liu R, Desta T, Leone C, Gerstenfeld LC, et al. (2004) Diabetes causes decreased osteoclastogenesis, reduced bone formation, and enhanced apoptosis of osteoblastic cells in bacteria stimulated bone loss. Endocrinology 145: 447-452.
- Kayal RA, Tsatsas D, Bauer MA, Allen B, Al-Sebaei MO, et al. (2007) Diminished bone formation during diabetic fracture healing is related to the premature resorption of cartilage associated with increased osteoclast activity. J Bone Min Res 22: 560-568.
- Santana RB, Xu L, Babakhanlou C, Amar S, Graves DT, et al. (2003) A role for advanced glycation end products in diminished bone healing in type 1 Diabetes. Diabetes 52: 1502-1510.
- Atar M, Atar-Zwillenberg DR, Verry P, Spornitz UM (2004) Defective enamel ultrastructure in diabetic rodents.Int J Paediatr Dent14: 301-307.
- El-Sayyad HIH, El-Sherbiny MA, Sobh MA, Ibrahim M (2010) Protective effects of Morus alba leaves extract against congenital and skeletal malformations of diabetic and hypercholesterolemic mother albino rat's offspring. Egypt J Exp Biol 6: 365-374.
- El-Sayyad HIH, El-Shershaby EM, Saleh, DF (2013) Effects of experimental diabetes and gliclazide on pregnant albino rats and their pups. JIMR 1: 22-29.
- El-Sayyad HIH , Al-Haggar MMS, El-Ghawet HA, Bakr IHM (2014) Effect of maternal diabetes and hypercholesterolemia on fetal liver of albino Wistar rats. Nutrition 30: 326-336.
- El-Sayyad HIH, El-Ghawet HA, Al-Haggar MS, Bakr IH (2015) Impairment of bone growth of wistar rat fetuses of diabetic and hypercholesterolemic mothers. Egyptian J Basic and Appl Sci 2: 1-12.
- El-Sayyad HIH, El-Sherbiny EM, Sobh MA, Abou-El-Naga AM, Ibrahim MAN, et al. (2011) Protective effects of Morus alba leaves extract on ocular functions of pups from diabetic and hypercholesterolemic mother rats. Int J Biol Sci 7: 715-728.
- El-Sayyad HI, Al-Haggar MS, El-Ghawet HA, Bakr IH (2012) Cardiomyopathy and angiogenesis defects of wistar rat fetuses of diabetic and hypercholesterolemic mothers. Nutrition 28: e33-43.
- Kennedy DA,Lupattelli A,Koren G,Nordeng H (2013) Herbal medicine use inpregnancy: Results of a multinational study. BMC Complement Altern Med13: 355.
- Azimi P,Ghiasvand R,Feizi A,Hariri M,Abbasi B (2014) Effects of cinnamon, cardamom, saffron, andginger consumption on markers of glycemic control, lipid profile, oxidative stress and inflammation in type 2diabetes patients. Rev Diabet Stud11: 258-266.
- Kaur G,Invally M,Khan MK,Jadhav P (2018) A nutraceutical combination ofCinnamomumcassia and Nigella sativa for Type 1diabetes mellitus. J Ayurveda Integr Med9: 27-37.
- Ahmadi R,Pishghadam S,Mollaamine FZ, Monfared MR (2013) Comparing the effects ofgingerand glibenclamide on dihydroxybenzoic metabolites produced in stz-induced diabetic rats. Int J Endocrinol Metab11: e10266.
- Ghasemi A,Khalifi S,Jedi S (2014) Streptozotocin-nicotinamide-induced rat model of type 2 diabetes. Acta Physiol Hung 101: 408-420.
- Deeg R, Ziegenhorn J (1983) Kinetic enzymic method for automated determination of total cholesterol in serum.Clin Chem 29: 1798-1802.
- Fossati P, Prencipe L (1982) Serum triglycerides determined colorimetrically with an enzyme that proceduces hydrogen peroxide.Clin Chem28: 2077-2080.
- Grove TH (1979) Effect of reagent PH on determination of the high-density lipoprotein cholesterol by precipitation with sodium phototungestate-magnesium.Clin Chem 25: 560-564.
- Friedewald WT, Levy RI, Fredrickson DS (1972) Estimation of low density lipoprotein cholesterol in plasma without use preparative ultracentri-fuge.Clin Chem18: 499-502.
- Jáuregui-Renaud K,Aranda-Moreno C,Herrera-Rangel A (2017) Utricular hypofunction in patients with type 2diabetes mellitus. Acta Otorhinolaryngol Ital37: 430-435.
- Mohammad JM,Robabeh S,Shahin K,Saeed T,Maryam A (2018) Auditory function and motor proficiency in type 1 diabetic children: A case-control study. Int J Pediatr Otorhinolaryngol109: 7-12.
- Han WK,Kim EH,Shin SA,Shin DS,Kim BJ,et al. (2018) Susceptibility of diabetic mice to noise trauma. Biomed Res Int2018: 7601232.
- Tsuda J,Sugahara K,Hori T,Kanagawa E,Takaki E,et al. (2016) A study of hearing function and histopathologic changes in the cochlea of the type 2diabetesmodel Tsumura Suzuki obesediabetesmouse. Acta Otolaryngol136: 1097-1106.
- Kocdor P,Kaya S,Erdil M,Cureoglu S,Paparella MM,et al. (2016) Vascular and neuroepithelial histopathology of the saccule in humans withdiabetes mellitus. Otol Neurotol37: 553-557.
- DewanjeeS,Das S,Das AK,Bhattacharjee N,Dihingia A,et al. (2018) Molecular mechanism of diabetic neuropathy and its pharmacotherapeutic targets. Eur J Pharmacol833: 472-523.
- Silva-Sousa YTC, Peres LC, Foss MC (2003) Are there structural alterations in the enamel organ of offspring of rats with alloxan-induced diabetes mellitus? Braz Dent J 14: 162-167.
- Pedersen LM, Tygstrup I, Pedersen J (1964) Congenital malformations in newborn infants of diabetic women. Correlation with maternal diabetic vascular complications. Lancet 1: 1124-1126.
- Wei D, Loeken MR (2014) Increased DNA methyltransferase 3b (Dnmt3b)-mediated CpG island methylation stimulated by oxidative stress inhibits expression of a gene required for neural tube and neural crest development in diabetic pregnancy. Diabetes 63: 3512-3522.
- Norén JG (1984) Microscopic study of enamel defects in deciduous teeth of infants of diabetic mother. Acta Odontol Scand 42: 153-156.
- Pascon T, Barbosa AMP, Cordeiro RCL, Bussaneli DG, Prudencio CB, et al. (2019) Prenatal exposure to gestational diabetes mellitus increases developmental defects in the enamel of offspring. PLoS ONE 14: e0211771.
- Lal S, Cheng B, Kaplan S, Softness B, Greenberg E, et al. (2008) Accelerated tooth eruption in children with diabetes mellitus. Pediatrics 121: e1139-e1143.
- Nonaka G-I, Morimoto S, Nishioka I (1983) Tannins and related compounds. Part 13. Isolation and structures of trimeric, tetrameric and pentameric proanthicyanidins from cinnamon.J Chem Soc Perkin Trans. Pp: 2139-2145.
- Peng X, Cheng KW, Ma J, Chen B,Ho CT,et al. (2004) Cinnamon bark proanthocyanidins as reactive carbonyl scavengers to prevent the formation of advanced glycation endproducts.J Agric Food Chem52: 65-70.
- Li H,Yao Y,Li L (2017) Coumarins as potential antidiabetic agents. J Pharm Pharmacol69: 1253-1264.
- Kaul PN, Joshi BS (2001) Alternative medicine: Herbal drugs and their critical appraisal-part II.Prog Drug Res57: 1-75.
- Shukla Y, Singh M (2007) Cancer preventive properties of ginger: A brief review.Food Chem Toxicol 45: 683-690.
- Suk S, Kwon GT, Lee E, Jang WJ, Yang H, et al. (2017) Gingerenone A, a polyphenol present in ginger, suppresses obesity and adiposetissue inflammationin high-fat diet-fed mice. Mol Nutr Food Res 61: 10.
- IlkhanizadehB, Shirpoor A, Ansari MHK, Nemati S, Rasmi Y (2016) Protective effects of ginger (Zingiber officinale) extract against diabetes-induced heart abnormality in rats. Diabetes Metab J 40: 46-53.
- Heras NDL,Valero-Muñoz M,MartÃn-Fernández B,Ballesteros S,López-Farré A,et al. (2017) Molecular factors involved in the hypolipidemic and insulin-sensitizing effects of aginger(Zingiber officinale Roscoe) extract in rats fed a high-fat diet. Appl Physiol Nutr Metab42: 209-215.
Citation: El-Ghawet HA, El-Sayyad HIH, Ghanem NFF, Mkhemar SSG (2019) Cinnamon and Ginger Combined Extract Improved Diabetic Induced Developmental Defects of Inner Ear and Teeth of Rat Fetuses. J Preg Child Health 6:411.
Copyright: © 2019 El-Ghawet HA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Share This Article
Recommended Journals
Open Access Journals
Article Usage
- Total views: 2427
- [From(publication date): 0-2019 - Jan 30, 2025]
- Breakdown by view type
- HTML page views: 1793
- PDF downloads: 634