Anti-Hu Antibody- and Anti-Zic4 Antibody-Positive Paraneoplastic Neurological Syndrome Presenting with Cerebellar Degeneration and Cranial Neuropathy: A Case Report
Received: 06-Oct-2022 / Accepted Date: 20-Oct-2022 / Published Date: 27-Oct-2021
Abstract
We present an illustrative case of a 62-year-old woman who developed progressive worsening of neurological symptoms with the simultaneous involvement of both peripheral and central nervous system. Our patient presented high titers of Anti-Hu and anti-Zic4 antibodies in her serum. We review the literature and found that multiple cranial palsies and concurrent cerebellar degeneration are uncommon in anti-Hu paraneoplastic syndromes. The patient was treated with intravenous immunoglobulin and methylprednisolone without significant improvement of symptoms.
Keywords: Cranial neuropathy, Cerebellar degeneration, Neurologic immune related adverse events, anti-Hu antibody, Paraneoplastic syndrome, case report
Abbreviations
PNS: Paraneoplastic Neurological Syndromes; SCLC: Small Cell Lung Cancer; LEMS: Lamber-Eaton Myasthenic Syndrome; LE: Limbic Encephalitis; SPS: Stiff-Person Syndrome; OM: Opsoclonus-Myoclonus; CGP: Chronic Gastrointestinal Pseudo- Obstruction; SSN: Sensory Neuronopathy; EM: Encephalomyelitis; CNS: Central Nervous System; CSF: Cerebrospinal Fluid; FDG-PET: Fluorodeoxyglucose Positron Emission Tomography; PCD: Paraneoplastic Cerebellar Degeneration
Introduction
Paraneoplastic Neurological Syndromes (PNS) consists of a group of immune-mediated neurological disorders that commonly occur with malignancies such as Small Cell Lung Cancer (SCLC), breast cancer, lymphoma and thymoma and are not caused by the tumor itself, metastasis, infection, ischemia or metabolic disruptions [1]. PNS are rare, affecting less than 1/100.000 patients with cancer. The most common PNS are Lamber-Eaton Myasthenic Syndrome (LEMS), subacute cerebellar ataxia, Limbic Encephalitis (LE), Opsoclonus- Myoclonus (OM), retinopathies, Stiff-Person Syndrome (SPS), Chronic Gastrointestinal Pseudo-obstruction (CGP), Sensory Neuronopathy (SSN), encephalomyelitis and dermatomyositis [2]. In most cases, the neurological feature precedes manifestation of associated cancers and patients are primarily referred to the neurologist to identify the causes [3]. What was interesting about this case was the presentation: the patient had initial peripheral nervous system (cranial neuropathy – abducens, facial and vagus nerve) involvement followed by clinical features from the Central Nervous System (CNS). Primary CT of thorax did not reveal any important pathological signs which made the diagnosis challenging.
Case Report
A 62-year-old female patient who presented with nausea, fatigue, hoarseness, and dysphagia was referred to the internal neurology department for a consultation (DAY 1). She reported the onset of the symptoms over the previous two months, with a progressive inability to eat and drink. The main initial complaints also included diplopia, ptosis, and gait ataxia. The patient had a personal history of smoking (20 pack years), had undergone a total thyroidectomy 6 years before and a family history of diabetes, cardiovascular diseases, thyroidopathy and hypertension. She reported being healthy prior to current condition and denied any alcohol or drug abuse. On examination, the patient was oriented, cachectic with facial weakness. Pupils were of equal size and normal reaction and she presented dysmetric saccades and limitation of abduction of the right eye, bulbar weakness, and dysphagia. Bilateral intention tremor was observed as well as left dysmetria on the finger-to-nose test. Barré, Mingazinni and Babinski tests were negative, masseter reflex was absent. Finally, Gordon Holmes rebound was observed on the right and examination of the sensory system was normal. Investigation was initiated to identify a cause.
Investigations
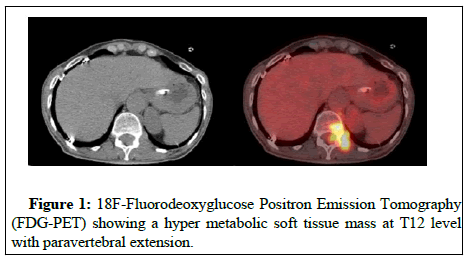
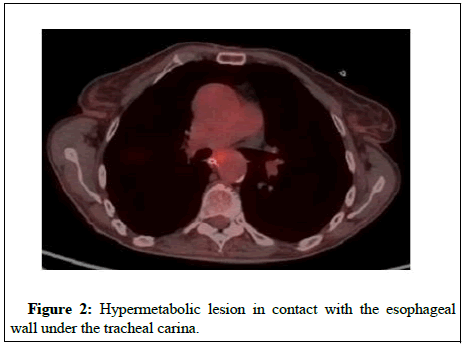
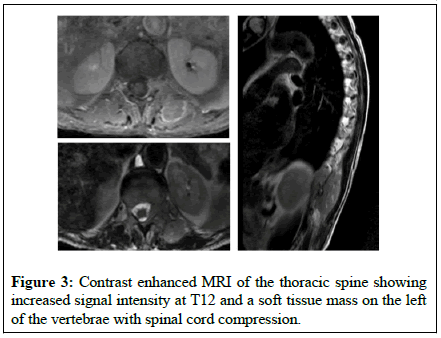
The patient underwent laryngoscopy which revealed paralysis of inferior laryngeal nerve while laryngeal polyps were removed (DAY 5). A subsequent esophagoscopy revealed gastropathy and dysmobility of the upper third of esophagus (DAY 7). CT of the neck and thorax did not reveal any important pathological signs (DAY 9). Brain MRI revealed microvascular brain disease (DAY 12). A Cerebrospinal Fluid (CSF) examination disclosed an elevated level of total protein with no atypical cells or organisms (DAY 15). A diagnosis of PNS was then considered. An analysis of antineuronal antibodies present in the serum showed a strong positivity for anti-Hu antibodies and anti- Zic4 antibodies, anti-AChR or anti-MuSK antibodies were not detected. Mammogram revealed no pathological findings (DAY 21). 18F- Fluorodeoxyglucose Positron Emission Tomography (FDG-PET) showed a hypermetabolic soft tissue mass at T12 level with paravertebral extension (Figure 1). Another hypermetabolic lesion was found in contact with the esophageal wall under the tracheal carina (DAY 23) (Figure 2). A subsequent Tc 99m bone scintigraphy showed increased tracer uptake at T12 and skull (possibly because of hyperostosis frontalis interna) (DAY 28) and contrast enhanced MRI of the thoracic spine showed increased signal intensity at T12 and a soft tissue mass on the left of the vertebrae which caused spinal cord compression (DAY 30) (Figure 3). Finally, CT scan of the thoracic spine revealed a mass with soft tissue density from T11 till S1 causing spinal cord compression (DAY 36). Biopsy of the above-mentioned lesion confirmed soft tissue malignancy of metastatic Small Cell Lung Cancer (SCLC) (DAY 47).
Treatment
According to the findings, PNS associated with SCLC was suspected and the patient was treated with Intravenous Immunoglobulin (IVIg 400 mg/kg per day for five days) and methylprednisolone (1 gm daily for five days) without significant improvement of symptoms. Patient died on day 56.
Discussion
Anti-Hu antibodies are a polyclonal IgG type of antibody that bind to intracellular antigens found in neural as well as tumour cells, thus leading to immune cross-reactivity. Small cell carcinoma of lung associated with paraneoplastic sensory neuronopathy and/or paraneoplastic encephalomyelitis with the presence of anti-Hu antibodies has been termed “anti-Hu” syndrome [4,5]. It is estimated that approximately 85% of anti-Hu positive cases are associated with SCLC [6]. As in other Hu-associated neurological syndromes, patients rarely improve despite treatment of the tumor and immunotherapy and medulla involvement may also contribute to the short overall survival [7,8].
Paraneoplastic Cerebellar Degeneration (PCD) is a heterogeneous group of disorders characterized by subacute cerebellar ataxia, specific tumour types and (often) associated antineuronal antibodies [9,10]. PCD is caused by immune-mediated injury to cerebellar Purkinje cells. It is associated with multiple malignancies but, most commonly, breast and pelvic malignancies. PCD has also been reported in patients with Hodgkin lymphoma, gastric cancer, prostate cancer, and small cell lung cancer.
Sudden death observations represent exceptional complications of paraneoplastic syndrome suggesting that clinicians should investigate for malignancy in patients presenting with a subacute cerebellar syndrome [11]. The low sensitivity and specificity of identified neuronal autoantibodies means that clinicians must retain a high index of suspicion even with an initially negative result, and a highly suggestive clinical presentation, exclusion of alternate pathology and response to immunotherapy should support a presumptive diagnosis.
However, multiple cranial nerve palsies and concurrent cerebellar degeneration are uncommon in paraneoplastic syndromes. Furthermore, cranial nerve involvement is an infrequent finding in anti-Hu syndrome. In patients with neurological symptoms of unknown cause detection of Zic4 antibodies predicts a neoplasm, usually a SCLC and suggests that the neurological disorder is paraneoplastic [12,13].
Conclusion
Detection of Zic4 antibodies often associates with anti-Hu or CRMP5 antibodies. Patients with isolated Zic4 antibodies are more likely to develop cerebellar dysfunction than those with concurrent immunities. In the case presented herein, both cranial neuropathy and cerebellar degeneration developed. Simultaneously, both anti-Hu and anti-Zic4 antibodies were detected in the patient's serum at a high titer, which is a highly uncommon finding.
Acknowledgement
We would like to thank the patient’s family for their cooperation.
Statement of Ethics
The patient’s family provided both oral and written informed consent for the publishing of this report.
Disclosure Statement
The authors have no conflicts of interest to declare.
Funding Sources
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors’ Contributions
Corresponding author: Ioannis Kourtesis, Writing - Original Draft Maria Orianou: Writing- Reviewing and Editing Yannis Asteris: Data Curation Gerasimos Georgatos: Software, Maria Maltezou: Conceptualization
References
- Aly R, Emmady PD (2020) Paraneoplastic Cerebellar Degeneration. StatPearls
- Bataller L, Wade DF, Graus F, Stacey HD, Rosenfeld, et al. (2004) Antibodies to Zic4 in Paraneoplastic Neurologic Disorders and Small-Cell Lung Cancer. Neurol 62: 778-782
- Elrington GM, Murray NM, Spiro SG, Davis JN (1991) Neurological Paraneoplastic Syndromes in Patients with Small Cell Lung Cancer. A Prospective Survey of 150 Patients. J Neurol Neurosurg and Psychiatry 54: 764-767.
- Honnorat J, Antoine JC (2007) Paraneoplastic Neurological Syndromes. Orphanet J Rare Dis :1-8.
- Li J, Lin W (2018) Various Clinical Features Of Patients With Anti-Hu Associated Paraneoplastic Neurological Syndromes: An Observational Study. Medicine 97: e0649–e0649.
- Mawhinney E, Gray OM, cVerry FM, McDonnell GV (2010) Paraneoplastic Sensorimotor Neuropathy Associated with Regression of Small Cell Lung Carcinoma. BMJ Case Rep.
- Mirouse A, Gobert D, Chamouard JM, Lord L, Mekinian A, et al. (2014) Sudden death occurring after anti-Hu associated paraneoplastic cerebellar degeneration and dysautonomia revealing a small cell lung carcinoma. Rev Med Interne 35: 757–759.
- Saiz A, Stourac P, Giometto B, Grisold W, Honnarat J, et al. (2009) Anti-Hu-Associated Brainstem Encephalitis. Journal of neurology, neurosurgery, and psychiatry 80: 404–407.
- Shams’ ili S, Grefkens J, de Leeuw B, Van den Bent M, Hooijkaas H, et al. (2003) Paraneoplastic Cerebellar Degeneration Associated With Antineuronal Antibodies: Analysis Of 50 Patients. Brain 126: 1409-1418.
- Sharobeam A, Ray J, Dong J, Chong V (2017) Subacute Cerebellar Degeneration Due To A Paraneoplastic Phenomenon Associated With Metastatic Merkel Cell Carcinoma: A Case Report. Case Rep Oncol 10: 764-768.
- Shibata M, Uchida M, Tsukagoshi S, Yamaguchi K, Yamaguchi A, et al. (2015) Anti-Hu Antibody-Associated Paraneoplastic Neurological Syndrome Showing Peripheral Neuropathy And Atypical Multifocal Brain Lesions. Internal Medicine 54: 3057-3060.
- Soomro Z, Youssef M, Yust-Katz S, Jalali A, Patel AJ, et al. (2020) Paraneoplastic Syndromes In Small Cell Lung Cancer. J Thorac Dis 12: 6253-6263.
- Zekeridou A, Majed M, Heliopoulos I, Lennon VA (2019) Paraneoplastic Autoimmunity And Smallâ€Cell Lung Cancer: Neurological And Serological Accompaniments. Thoracic cancer 10: 1001-1004.
Citation: Kourtesis I, Orianou , Asteris Y, Georgatos G, Malteou M (2021) Anti-Hu Antibody- and Anti-Zic4 Antibody-Positive Paraneoplastic Neurological Syndrome Presenting with Cerebellar Degeneration and Cranial Neuropathy: A Case Report. J Clin Exp Neuroimmunol 6:134.
Copyright: © 2021 Kourtesis I, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Share This Article
Recommended Journals
Open Access Journals
Article Usage
- Total views: 3360
- [From(publication date): 0-2021 - Apr 16, 2025]
- Breakdown by view type
- HTML page views: 2419
- PDF downloads: 941