Anticancer Potentials of Phytochemicals from Some Indigenous Food and Medicinal Plants of West Africa
Received: 09-Aug-2017 / Accepted Date: 12-Oct-2017 / Published Date: 22-Oct-2017 DOI: 10.4172/2472-0429.1000124
Abstract
A selected number of potential anticancer agents in phytochemicals isolated from some indigenous food and medicinal plants of the West African sub-region, has been reviewed. The attempt has benefited from a store of knowledge on the characterized and identified phytochemicals from indigenous plants by organic chemists, in the past six or so decades. Such compounds as satisfied the structure-activity-relationships with known anticancer active agents were selected and profiled and cover phytochemical classes as: alkaloids, flavonoids, terpenoids, and ‘miscellaneous’, the latter class comprising compounds considered chemically inappropriate for the previous classes. Anticancer activities covered include: induction of carcinogen-metabolizing enzymes, selective cytotoxicity to tumor cells, reversal of multidrug resistance in cancer cells, and inhibition of metastasis. Food and medicinal uses of the source plants have also been described.
Keywords: Anticancer; phytochemicals; West Africa; Alkaloids; Flavonoids; Terpenoids
Introduction
Many indigenous West African plants have been employed as local remedies for various human ailments in the traditional medicine of the region, and as nutritional sources for countless generations [1-3]. Isolation and characterization of the chemical structures for phytochemicals from useful indigenous plants have however been undertaken only for a minimal fraction, largely because the modern techniques of chromatography and mass spectroscopy have been exploited by African scientists relatively recently [4-6]. The significance of the latter development has been to greatly enhance an understanding of the molecular mechanisms of action for these isolated compounds as pharmacological agents. In particular, such knowledge has been applied in the present discussion to identify phytochemicals from indigenous West African plants with potential anticancer activities, based on their structure-activity relationships to known active compounds – an outcome totally unanticipated by the organic chemists who merely isolated and characterized these phytochemicals, or whose main focus was on other biological activities.
That a selected few plants would yield the interesting examples discussed herein, underscores the fact that a programmed, methodical investigation of the vast flora of the region, in particular, the prodigiously rich tropical rain forests, would uncover a resource of unimaginable magnitude and inestimable value as anticancer agents. After all, it is on record, that the first universally acclaimed and potent anticancer agents were discovered from plants of the African continent and include such famous examples as vincristine and vinblastine, both isolated from the Madagascan periwinkle plant, Catharanthus roseus G. Don (Syn. Vinca roseus Linn). These compounds were touted as:“ the most successful of the plant-derived antitumor agents” [7]. Another significant example is maytansine, isolated from the Ethiopian shrub Maytenus serrata (Celestraceae) and was one of the most promising antitumor agents discovered by the commendable program for extensive screening of antitumor principles from plants, by the National Cancer Institute of the USA [8]. The Kenyan plant species Maytenus buchananii is reported to be a richer source of maytansine, yielding more than seven times the amount from M. serrata [7]. A Maytenus species indigenous to West Africa and commonly found in Nigeria is M. senegalensis (syn. Gymnosporia senegalensis), (Celestraceae). It is very likely that a phytochemical investigation of the Nigerian plant species may yield maytansinoids, as is the case with its near relatives in East Africa.
The profiled phytochemicals are grouped as potential “Cancer Chemopreventive” and “Cancer Chemotherapeutic” agents and cover many of the known phytochemical classes such as alkaloids, flavonoids, terpenoids and ‘miscellaneous’, the latter class comprising compounds considered chemically inappropriate for the previous classes. Anticancer activities covered include: induction of carcinogenmetabolizing enzymes, selective cytotoxicity to tumor cell lines, reversal of multidrug resistance in cancer cells, and inhibition of metastasis, with a few specific examples selected to illustrate the point. In some instances, anticancer potentials of the phytochemicals have been discerned from relevant experimental data. A significant example in this regard would be the case of the biflavonoid, kolaviron which had hitherto been known as a potent hepatoprotective agent [9,10] and which, in the author’s research experience, gave indications of a potential human cancer chemopreventive agent against aflatoxin B1- induced hepatocarcinogenesis [11]. The dietary applications as well as traditional medicinal uses of the source plants, as practiced in the specific local environments of origin, have also been described. Such identified compounds however, still await relevant biological activity tests, before confirmation of suggested anticancer potentials.
Cancer Chemopreventive Agents
A. Induction of carcinogen metabolizing enzymes
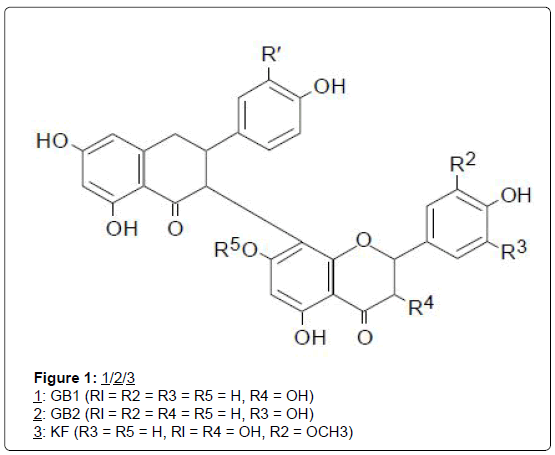
From fresh seeds of an indigenous plant, Bitter kola: Garcinia kola Heckel (Guttiferae), was isolated kolaviron (KV), a mixture of three biflavonoids; GB-1 (1), GB-2 (2) and kolaflavanone, KF (3), [12]. Previous studies had demonstrated that kolaviron reduced hepatotoxicity in experimental animals [12-14]. A subsequent study by this author, indicated that it could inhibit aflatoxin B1 (AFB1)-induced genotoxicity in the human liver-derived HepG2 cells [11]. Kolaviron, at the highest tested concentration of 90 μM reduced AFB1-induced unscheduled DNA synthesis, measured as 3H-thymidine incorporation into HepG2 cells, by 53%. The latter study further demonstrated that kolaviron’s mechanism of action involved an induction of the AFB1- detoxicating enzymes such as cytochrome P450 3A4, and glutathione S-transferases A1-1, A2-2, M1-B, indicated by a three-fold increase in the transcripts for the enzymes, and a two-fold increase for GST A1-1 and A2-2 protein. Kolaviron therefore appeared to have a chemopreventive potential in inhibition of human AFB1-induced genotoxicity and possibly, carcinogenesis (Figures 1 and 2).
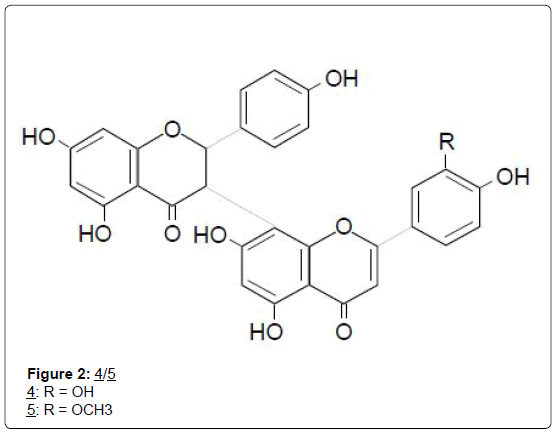
Bitter kola (Garcinia kola) seed from which kolaviron was obtained, is an edible nut frequently consumed as “kola”, or guest snack both at homes and at social gatherings in the southern parts of Nigeria. Two biflavonoids structurally similar to kolaviron were also isolated from the stem bark of G. densivenia [15] and named, morelloflavone (4), and its methyl ether derivative, O-methylfukugetin (5). Many other biflavonoids have been isolated from Garcinia species of the Douala- Edea Forest Reserve of West Cameroun, some of which could display cancer chemopreventive activity similar to kolaviron [16]. These compounds are yet to be investigated for possible anticancer activity.
In a follow-up study, the potentials of kolaviron as a pleiotropic inducer of hepatoprotective genes, especially in aflatoxin B1 toxication of human hepatocytes, was evaluated by gene microarray assay [17]. This work employed human primary hepatocytes (HPH) separately treated with AFB1 (2.0 μM /L) and KV (90 μM /L) then a combined pretreatment with kolaviron before AFB1, while gene expression analysis employed the Affymetrix Gene 1.0ST array containing the entire human genome. Results suggested a central role for the human“Inhibitor of DNA binding 1, ID1 gene” (Gene ID: 3397; MIM: 600349), which was significantly up-regulated to about three-fold message levels, in all three treatments, as well as a few other genes that were separately regulated [17].
Cancer Chemotherapeutic Agents
A. Cytotoxicity to tumor cells
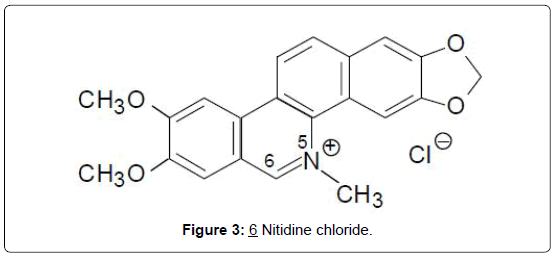
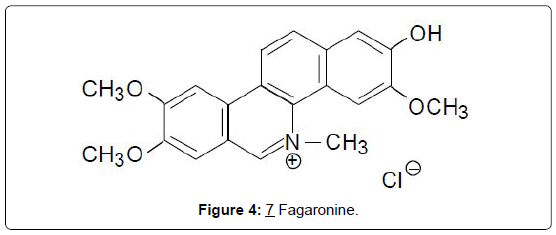
i) Alkaloids: Benzophenanthridine alkaloids: The benzophenanthridine alkaloids, nitidine chloride, (6), and a new derivative, 6-methoxy-5,6-dihydronitidine, were isolated from Fagara macrophylla [18]. Both compounds were shown to be about equipotent in the P-388 mouse leukemia system, giving high T/C values of 240 and 260% respectively, at doses of 30-50 mg/kg. Another derivative fagaronine, (7), was isolated from the Ghanaian and Nigerian plant, Fagara zanthoxyloides Lam. (Rutaceae) [19]. Fagaronine (7), like its relative, nitidine, is active in both the P-388 and L-1210 leukemia test systems [20]. A latter study suggests that the structural determinants for antitumor activities in benzophenanthridines, include the existence of a planar, cationic resonance hybrid between the keto-amine and zwitterionic forms in neutral media, and a phenolic OH at C-7 [21]. It therefore becomes necessary to review the activities of the subsequently discovered derivatives as described below (Figures 3 and 4).
One such alkaloid bearing structural similarities to a known antitumor compound is palmatine, (8), extracted among other alkaloids, from roots of the West African climbing shrub, Rhigiocarya racemifera [22]. Palmatine (8), is structurally similar to coralyne sulfoacetate, (9), the latter being one of the related alkaloids of nitidine chloride, which showed antileukemic activity [23].
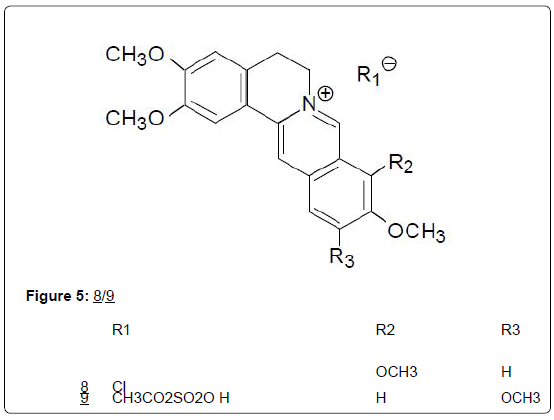
A decoction of the stem bark of Fagara macrophylla is taken as an antiseptic for the genitourinary tract, and is also applied as a rubefacient in toothaches, in the folk medicine of Nigeria [16]. Extracts of the roots are also used for coughs and colds. R. racemifera Miers (Menispermaceae), is a climbing shrub indigenous to the forests of Ghana and other parts of West Africa, whose roots are added to palm wine, and extracts of the plant used medicinally as nasal drops, and as an aphrodisiac (Figure 5) [22].
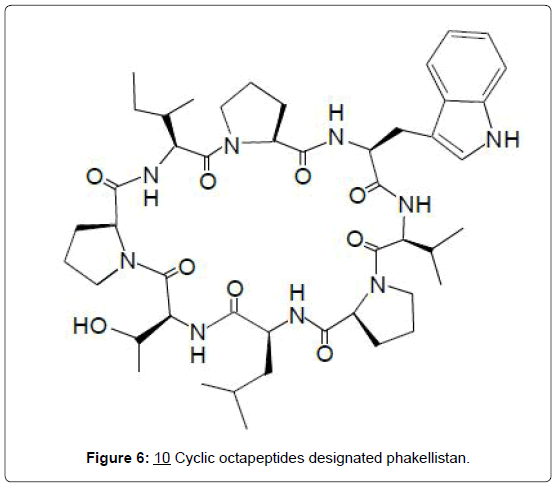
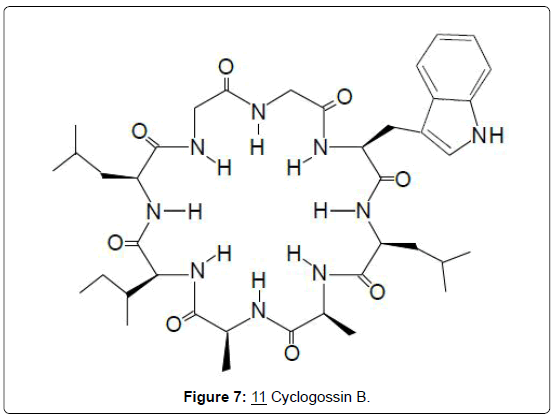
Peptide alkaloids: For peptide alkaloids, the isolation of 2 new cyclic octapeptides designated phakellistan 10 (10) and phakellistan 11 from the marine sponge Phakellia spp. has been reported [24]. Both compounds demonstrated significant cytotoxic activity against the P-388 leukemia cell line, having ED50 values of 2.1 and 0.2 μg/ mL, respectively. A structurally related cyclic octapeptide named cyclogossin B (11), was subsequently isolated from latex of the plant Jatropha gossypifolia (Euphorbiaceae) [25]. Cyclogossin B is yet to be investigated for possible cytotoxic activity. Structure-activity relationships of cytotoxic cyclic octapeptides have not been determined but on the basis of the close structural similarity between phakelistan 10 (10) and cyclogossin B (11), it is reasonable to expect that the latter may also display some cytotoxic activity.
The fresh latex of Jatropha gossypifolia is reportedly applied to infected wounds and ulcers and a decoction of the plant is used to treat fever in Togo and Senegal, where the plant bears the vernacular name of “lumulum” (Figures 6 and 7) [25].
Other cyclopeptide alkaloids have been isolated from local plants, including a number of such compounds from the root bark of Hymenocardia acida, beginning with the first such isolate named hymenocardine [26]. Subsequently, hymenocardine was re-isolated along with its derivatives including one with a hydroxyl- in place of a carbonyl group that was named hymenocardinol, a hymenocardine N-oxide, and a new cyclopeptide alkaloid containing an unusual histidine moiety named hymenocardine-H [27]. In a previous work, it had been reported that the methanol extract from the same part of the plant exhibited cytotoxic activity against a panel of human tumor cell lines [28].
Hymenocardia acida Tul. (Phyllanthaceae) is a shrub or small tree indigenous to the African Savannah where the leaves and roots of the plant are used to treat malaria in traditional African medicine [29]. The roots of the plant are also said to be used as antimalarial in Nigeria [30].
ii) Terpenoids: Of the numerous reports of tumor cytotoxic terpenoids, one example selected from indigenous West African plants would serve to illustrate the potency of anticancer activity that may be found in this group.
a) Sesquiterpenoids: The cytotoxic and antitumor sesquiterpenes have been found almost exclusively among the germacrane, guaiane, eudesmane and elemane sub-groups. Almost without exception, a common structural factor for these active compounds appears to be the possession of an α-methylene-γ-lactone ring. The latter chemical grouping, alone, or in addition to other reactive groups such as - enones, α,β-unsaturated lactones, ketones, and esters or epoxides, have been hypothesized to be subject to nucleophilic attack, resulting in the alkylation of biologically significant macromolecules, and consequently leading to cancer cell death [31].
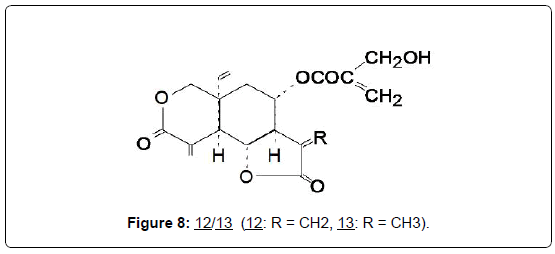
A typical example is the active compound vernodalin (12), from the elemanolide sub-group isolated from the dried leaves of Vernonia amygdalina Del. [32] and having significant in vitro cytotoxic activity against the Walker carcinoma 256 with an ED50 of 1.8 μg/mL. 12 was reisolated from Vernonia amygdalina, along with two other elemanolide lactones, vernodalol and a new compound dihydrovernodalin (13), [33]. The possible anticancer activities of the latter two compounds have not yet been investigated (Figure 8).
A new anticancer sesquiterpene lactone has been isolated from Vernonia amygdalina and named:
“vernodalinol” [34]. The compound which possesses the requisite α-methylene-γ-lactone ring postulated as the structural requirement for anticancer activity in the sesquiterpene lactones, displayed modest cell growth inhibitory activity (inhibition of DNA synthesis) in the human mammary adenocarcinoma cell line MCF-7, where values of 34% and 40% (P <0.025) growth inhibition were obtained at concentrations of 25 and 50 μg/mL, respectively [34].
Vernonia amygdalina (bitter leaf) is a highly valued food item as the leaves are a popular vegetable for soups among the Igbo of southern Nigeria. Medicinal applications of V. amygdalina include use of the twigs as “chewing stick”, or local toothbrush, which are also chewed as a stomachic tonic and appetizer. A decoction of the leaves is taken as an antipyretic and laxative, and for coughs [16].
iv) Miscellaneous: This category covers phytochemicals possessing significant in vitro cytotoxicities against tumor cell lines, and which, for reasons of proper nomenclature, may not be included in the other classical groups. For reasons of space and brevity, only one example is discussed here.
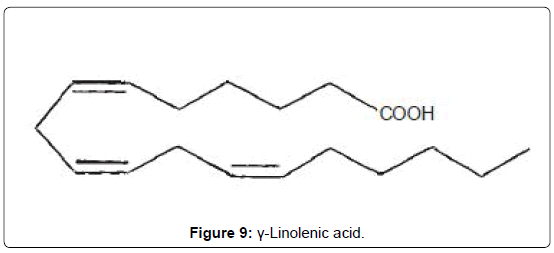
Gamma-linolenic acid: Patients with malignant primary gliomas or mammary adenocarcinomas have a poor prognosis as common therapeutic approaches are relatively ineffective. Interestingly, an edible, essential polyunsaturated fatty acid (PUFA), γ-linolenic acid (GLA, 14) was found to be selectively cytotoxic to gliomas and other tumor types [35,36], while normal cells are largely unaffected. Although the mechanism for this selective cytotoxicity is poorly understood, free radical generation with lipid peroxidation from PUFA supplementation is generally believed to play a role [35,36]. However, the latter assumption is flawed on many counts, not least of which is the fact that higher members of the PUFAs, containing higher unsaturation and presumably better able to generate the assumed toxic free radicals, are ineffective (Figure 9).
This author proposed a plausible mechanism for GLA-induced cytotoxicity to tumor cells, particularly gliomas and mammary adenocarcinomas [37]. The article was published in 2001 and titled: “Repression of cellular anaplerosis as the hypothesized mechanism of GLA-induced toxicity to tumor cells” [37]. About four years later, workers in another laboratory [38] independently provided corroboratory experimental support for this hypothesis, thus strengthening the theory and conferring scientific legitimacy on the postulate. There is hope that this postulate would serve as a basis for the design of new therapeutic agents against the dreaded gliomas and metastatic mammary carcinomas.
GLA, the tumor cell cytotoxic agent, was subsequently shown to be present in large amounts, as its biochemical precursor, linoleic acid, in the commonly eaten (South-Eastern Nigeria) oil bean seed (‘ugba’ or ‘upkaka’: Pentaclethra macrophylla) in a related article co-published by this author [39].
In a related anticancer activity of a dietary linolenic acid obtainable from indigenous plants, dietary supplementation with 0.01% and 0.1% oil from bitter melon (Momordica charantia) which is rich in cis(c)9, trans(t)11, t13-conjugated linolenic acid (CLN), significantly reduced the incidence of azoxymethane (AOM)-induced colonic aberrant crypt foci (ACF) in male 344 rats [40]. The authors suggested that the mechanism of CLN suppression of AOM-induced colon carcinogenesis might, in part be through modification of lipid composition in the colon and liver and/or increased expression of PPARgamma protein level in the colon mucosa.
Momordica charantia (Cucurbitaceae) is a plant of common use in the traditional medicine of West Africa as an antimalarial [41], in the treatment of measles [42] in Nigeria, and for the treatment of gastrointestinal and viral disease in Togo [43].
B. Reversal of multi-drug resistance (MDR)
An illustrative example will be cited here, from the alkaloid class of phytochemicals.
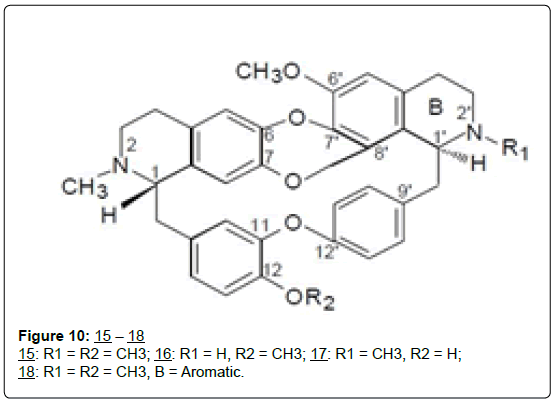
The bisbenzylisoquinoline alkaloids: Figure 10 Two known bisbenzylisoquinoline alkaloids, isotrilobine (15), and trilobine (16), were isolated from an extract of the vines, Stephania japonica [44]. Using the bicinchoninic acid assay, both compounds were shown to be active in reversing doxorubicin resistance in the adriamycinresistant human breast adenocarcinoma cell line, MCF-7/ADR, with isotrilobine (15) being more active than trilobine (16) and equipotent with the reference compound verapamil [44]). Since the only structural difference between (15) and (16) is the methyl group at N-2’, the greater efficacy of 15 suggests that a tertiary amine is preferred at this position to a secondary amine. The authors also suggested that the increased lipophilicity induced by presence of an extra methyl group in 15 may contribute to the enhanced activity, since MDR inhibitors are known to be lipophilic.
Other bisbenzylisoquinoline alkaloids, cocsuline (17) and trigilletimine (18), have been reported from extracts of the whole plant of Triclisia dictyophylla Diels. (Menispermaceae), [45], and from leaves of T. gilletti (DeWild.) Staner, were isolated four more such compounds named obamegine, stebisimine, gilletine and isogilletine-N-oxide, the last two described as new bisbenzylisoquinoline dibenzo-p-dioxin alkaloids [46]. The remarkable structural similarity between these Triclisia isolates and the MDR-active compounds isotrilobine (15) and trilobine (16) raises the hope that some may be found active, if tested. Of particular interest in this regard is cocsuline (17), which is almost identical with the highly active isotrilobine (15), and differing only in the possession of a hydroxyl, rather than a methoxy substituent at position 12. Similarly, trigilletimine (18) may display a higher activity than trilobine (16) since the only structural difference between the two is that the “B” ring in the former is aromatic which should stand it in good stead. For instance, possession of a terminal aromatic group, as well as an internal π-environment are two of the structural requirements for reversal of MDR-activity in these compounds [44].
A bisbenzylisoquinoline alkaloid named ‘cycleanine’ has been isolated from ethanol extract of Triclisia subcordata and shown to potently (IC50<2.4 μg/mL) inhibit tumor cell growth in the ovarian cancer, Ovcar-8 and A2780 cell lines, using the Sulforhodamine B assay [47]. The IC50 of cycleanine on human normal ovarian surface epithelial cells was 35 ± 1 μM, thus suggesting modest selectivity toward cancer cells. Mechanism of action for cycleanine was further indicated to be through apoptosis as shown by activation of caspases 3/7 and cleavage of poly(ADP-ribose) polymerase [47].
From leaves of another indigenous West African plant, Tiliacora funifera, was isolated a new imino bisbenzylisoquinoline alkaloid named, ‘tiliafunimine’ [48]. The authors also described two other bisbenzylisoquinoline alkaloids isolated from the same source as, isotetrandrine and thalrugosine. These compounds are yet to be investigated for possible multidrug resistance activitiy Triclisia subcordata Oliv (Menispermeaceae) was described by the authors as a medicinal plant traditionally used for the treatment of various diseases in West Africa [47], while Tiliacora funifera (Menispermaceae) is a woody climber of Ghana and West Africa that has been used as a medicinal in the treatment of numerous ailments [48].
C. Inhibition of metastasis
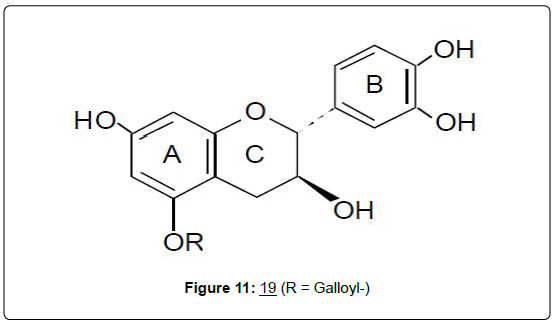
A significant example in this category would be the report of work involving the phytochemical, (+)-catechin (19). This compound has been, almost exclusively, the only flavan investigated for its effects in cancer. Prominent among the anticancer effects reported are its antioxidant effect in inhibiting the auto-oxidation of linoleic, and methyllinoleic acids [49], and the in vitro inhibition of metastasis as described below. In the latter study, the anti-metastatic potential of some phytochemicals, including (+)-catechin, against the highly invasive, Dunning rat prostate adenocarcinoma cells (R3327-5´A), was investigated [50]. Among the invasive parameters studied, were: matrigel membrane invasion by the trans-well assay; gel zymography assay and Northern analysis, for the matrix metalloproteinases (MMP- 2 and MMP-9)’s activity and gene transcription respectively; as well as Western immunoblot assay, for gene expression analysis of the membrane-associated proteins α-, β- and γ-catenins. Results indicated that catechin at 25 μM, significantly decreased matrigel membrane invasion of the prostate tumor cells by 24% and also inhibited γ-catenin protein levels by 58% (P<0.01), (Figure 11) [50].
A significant Nigerian food source from which (+)-catechin (19), and its derivatives have been isolated is described here. 19 was obtained from five edible species of Dioscorea (yams), indigenous to Nigeria [51]. The five yam species, and their cultivars or Nigerian (Igbo) names were given as Dioscorea alata L, two cultivars (Ominelu and UM 680); D. bulbifera L, (Adu); D. cayanensis Lam. (Oku); D. dumetorum Pax. (Ona); and D. rotundata Poir., five cultivars (Abii, Ekpe, Nwopoke, Obiaturugo and Okwocha), [51]. Yams are a starchy staple and highly prized foodstuff in Nigeria and neighboring countries. Often, the wealth of a farmer in the Igbo-speaking areas of Nigeria is determined by the quantity of yams stocked.
In an analysis for secondary plant metabolites contents of kola nuts (Cola nitida) popularly consumed in Nigeria and other West African countries, (+)-catechin was shown to be present in the highest amount among the phytochemicals found [52]. Three varieties of kola nuts (Cola nitida alba, Cola nitida rubra A. Chev, and Cola acuminata Schott & Endl) were analyzed and the phytochemical contents given in decreasing order of quantity as: (+)-catechin (27-37 g/kg), caffeine (18- 24 g/kg), (-)-epicatechin (20-21 g/kg), procyanidin B 1 [epicatechin- (4beta-->8)-catechin] (15-19 g/kg), and procyanidin B2 [epicatechin- (4beta-->8)-epicatechin] (7-10 g/kg) [52,53]. Kola nuts would therefore constitute a significant source of the anticancer compound (+)-catechin, especially for people in the northern parts of Nigeria where kola nut consumption is a very popular pastime.
Conclusion
The objective of this review has been to primarily highlight the existence of promising anticancer potentials inherent in phytochemicals from indigenous West African plants of common usage, as local food, or in the traditional medicine of the area. Ethnopharmacologists and other scientists in related fields on the African continent are hereby exhorted to spear-head more of such studies aimed at espousing the immense benefits accruable from a revival of interest in this relatively neglected pool of valuable resource.
It is however important to stress that efforts should be made to isolate and characterize the specific active principles responsible for anticancer activity. The common practice of ascribing activities to crude extracts, while understandably attributable to a paucity of research facilities, would hamper a desirable appreciation of mechanistic insights and deter necessary contribution to the international knowledge base on this subject. This discussion in which the chemical structures of active agents are presented and evaluated, illustrates the advantages to be gained in the prescribed approach.
Finally, the author once more wishes to draw attention to the imminent threat posed by rapid deforestation of tropical forests, and the looming danger that rare curative plants would be lost forever, if immediate preventive action is not taken.
References
- Sawadogo WR, Schumacher M, Teiten MH, Dicato M, Diederich M (2012) Traditional West African pharmacopeia, plants and derived compounds for cancer therapy. Biochem Pharmacol. 84:1225-1240.
- Lifongo LL, Simoben CV, Ntie-Kang F, Babiaka SB, Judson PN (2014) A bioactivity versus ethnobotanical survey of medicinal plants from Nigeria, west Africa. Nat Prod Bioprospect. 4:1-19.
- Tsouh Fokou PV, Nyarko AK, Appiah-Opong R, Tchokouaha Yamthe LRÂ (2015) Ethnopharmacological reports on anti-Buruli ulcer medicinal plants in three West African countries. J Ethnopharmacol 172:297-311.
- Avoseh O, Oyedeji O, Rungqu P, Nkeh-Chungag B, Oyedeji A (2015) Cymbopogon species; ethnopharmacology, phytochemistry and the pharmacological importance. Molecules 20:7438-7453.
- Boucherle B, Haudecoeur R, Queiroz EF, De Waard M, Wolfender JL, et al. (2016) Nauclea latifolia: biological activity and alkaloid phytochemistry of a West African tree. Nat Prod Rep 33:1034-1043.
- Costa R, Albergamo A, Pellizzeri V, Dugo G (2017) Phytochemical screening by LC-MS and LC-PDA of ethanolic extracts from the fruits of Kigelia africana (Lam) Benth. Nat Prod Res 31:1397-1402.
- Sneden AT (1984) Novel antitumor agents from plants. In: Ottenbrite RM, Butler GB(editors)Anticancer and Interferon Agents: Synthesis and Properties. Marcel Decker Inc.: New York and Basel.
- Komoda Y, Kishi T (1980) Maytansinoids. In: Cassady JM, Douros JD (editors)Anticancer Agents based on Natural Products Models.Academic Press.
- Iwu MM, Igboko OA, Onwuchekwa UA, Okunji CO (1987) Evaluation of the antihepatotoxic activity of the biflavonoids of Garcinia kola seed. J Ethnopharmacol 21:127-138.
- Farombi EO (2000) Mechanisms for the hepatoprotective action of kolaviron: studies on hepatic enzymes, microsomal lipids and lipid peroxidation in carbon tetrachloride-treated rats. Pharmacol Res 42:75-80.
- Nwankwo JO, Tahnteng JG, Emerole GO (2000) Inhibition of aflatoxin B1 genotoxicity in human liver-derived HepG2 cells by kolaviron biflavonoids and molecular mechanisms of action. Eur J Cancer Prev 9:351-361.
- Iwu MM (1985) Antihepatoxic constituents of Garcinia kola seeds. Experientia 41:699-700.
- Iwu MM, Igboko OA, Elekwa OK, Tempesta MS (1990) Prevention of thioacetamide-induced hepatotoxicity by biflavanones of Garcinia kola. Phytother Res 4:157-159.
- Braide VB (1991) Antihepatotoxic biochemical effects of kolaviron, a biflavonoid of Garcinia kola seeds. Phytother Res 5:35-37.
- Waterman PG, Crichton EG (1980) Xanthones and biflavonoids from Garcinia densivenia stem bark. Phytochem 19:2723-2726.
- Nwankwo JO (2011) In: Potential anticancer and antiviral agents from West African phytochemicals. Flavonoids pp: 56-59
- Itokawa H, Oshida Y, Ikuta A, Inatomi H, Ikegami S (1981) Flavonol glycosides from the flowers of Cucurbita pepo. Phytochem 20:2421-2422.
- Wall ME, Wani MC (1987) Plant antitumor agents, 27. Isolation, structure, and structure activity relationships of alkaloids from Fagara macrophylla. J Nat Prod 50:1095-1099.
- Cordell GA (1977) Recent experimental and clinical data concerning antitumor and cytotoxic agents from plants. In: “New Natural Products and Plant Drugs with Pharmacological, Biological or Therapeutical Activity†Proc 1st Intl Congress Med Plant.
- Res Section A.Wagner H, Wolff P(editors) Springer-Verlag, Berlin, Heidelberg, New York.
- Nakanishi T, Suzuki M, Saimoto A, Kabasawa T (1999) Structural Considerations of NK109, an Antitumor Benzo[c]phenanthridine Alkaloid. J Nat Prod 62:864-867.
- Dwuma-Badu D, Ayim JSK, Withers SF, Agyemang NO, Ateya AM, et al. (1980) Constituents of West African Medicinal Plants. XXVII. Alkaloids of Rhigiocarya racemifera and Stephania dinklagei. J Nat Prod 43:123-129.
- Suffness M, Douros J (1980) In: Cassady JM, Douros JD (editors).Anticancer agents based on natural products models. Acad Press: NY; p:14.
- Pettit GR, Tan R, Ichihara Y, Williams MD, Doubek DI, et al. (1995) Antineoplastic Agents, 325. Isolation and Structure of the Human Cancer Cell Growth Inhibitory Cyclic Octapeptides Phakellistatin 10 and 11 from Phakellia sp. J Nat Prod 58:961-965.
- Auvin-Guette C, Baraguey C, Blond A, Pousset JL, Bodo B (1997) Cyclogossin B, a cyclic octapeptide from Jatropha gossypifolia. J Nat Prod 60:1155-1157.
- Pais M, Marchand J, Ratle G, Jarreau FX (1968) Peptidic alkaloides. VI. Hymenocardine, alkaloid from Hymenocardia acida Tul. Bulletin de la Société Chimique de France 7:2979-2984.
- Tuenter E, Exarchou V, Baldé A, Cos P, Maes L, et al. (2016) Cyclopeptide alkaloids from Hymenocardia acida. J Nat Prod. 79:1746-1751.
- Muanza DN, Euler KL, Williams L, Newman DJ (1995) Screening for antitumor and anti-HIV activities of nine medicinal plants from Zaire. Int J Pharmacognosy 33:98-106.
- Tuenter E, Bijttebier S, Foubert K, Breynaert A, Apers S, et al. (2017) In vitro and in vivo study of the gastrointestinal absorption and metabolisation of Hymenocardine, a cyclopeptide alkaloid. Planta Med 83:790-796.
- Ainslie J (1937) A list of plants used in native medicine in Nigeria. Imperial Forestry Institute, University of Oxford p: 107
- Cassady JM, Suffness M (1980) In: Cassady JM, Douros JD (editors) Antitumor Agents based on Natural Product Models. Academic Press:NY; pp: 201-269.
- Kupchan SM, Hemingway RJ, Karim A, Werner D (1969) Tumor inhibitors. XLVII. Vernodalin and vernomygdin, two new cytotoxic sesquiterpene lactones from Vernonia amygdalina del. J Org Chem 34:3908-3911.
- Ganjian I, Kubo I, Fludzinski P (1983) Insect antifeedant elemanolide lactones from Vernonia amygdalina Phytochem. 22:2525-2526.
- Luo X, Jiang Y, Fronczek FR, Lin C, Izevbigie EB, et al. (2011) Isolation and structure determination of a sesquiterpene lactone (vernodalinol) from Vernonia amygdalina extracts. Pharm Biol 49: 464-470.
- Horobin DF (1989) In: Abraham S (editor)Carcinogeneisi and dietary fat. Kluwer Academic Publishers: Boston; pp: 247-262.
- Jiang WG, Bryce RP, Horrobin DF (1998) Essential fatty acids: molecular and cellular basis of their anti-cancer action and clinical implications. Crit Rev Oncol Hematol 27:179-209.
- Nwankwo JO (2001) Repression of cellular anaplerosis as the hypothesized mechanism of gamma-linolenic acid-induced toxicity to tumor cells. Med Hypotheses 56:582-588.
- Menendez JA, Colomer R, Lupu R (2005) Inhibition of fatty acid synthase-dependent neoplastic lipogenesis as the mechanism of γ-linolenic acid-induced toxicity to tumor cells: an extension to Nwankwo’s hypothesis. Med Hypotheses 64:337-341.
- Onwuliri VA, Attah I, Nwankwo JO (2004) Anti-nutritional factors, essential and non-essential fatty acids composition of Ugba (Pentaclethra macrophylla) seeds at different stages of processing and fermentation J Biol Sci. 4:671-675.
- Kohno H, Yasui Y, Suzuki R, Hosokawa M, Miyashita K, et al. (2004) Dietary seed oil rich in conjugated linolenic acid from bitter melon inhibits azoxymethane-induced rat colon carcinogenesis through elevation of colonic PPARgamma expression and alteration of lipid composition. Int J Cancer 110:896-901.
- Olasehinde GI, Ojurongbe O, Adeyeba AO, Fagade OE, Valecha N, et al. (2014) In vitro studies on the sensitivity pattern of Plasmodium falciparum to anti-malarial drugs and local herbal extracts. Malar J 13:63.
- Sonibare MA, Moody JO, Adesanya EO (2009) Use of medicinal plants for the treatment of measles in Nigeria. J Ethnopharmacol 122:268-72.
- Beloin N, Gbeassor M, Akpagana K, Hudson J, de Soussa K, et al. (2005) Ethnomedicinal uses of Momordica charantia (Cucurbitaceae) in Togo and relation to its phytochemistry and biological activity. J Ethnopharmacol 96:49-55.
- Hall AM, Chang CJ (1997) Multidrug-Resistance Modulators from Stephania japonica. J Nat Prod 60:1193-1195.
- Spiff AI, Zabel V, Watson WH, Zemaitis MA, Ateya AM, et al. (1981)Constituents of West African Medicinal Plants. XXX. Tridictyophylline, A New Morphinan Alkaloid From Triclisia dictyophylla. J Nat Prod 44:160-165.
- Owusu PD, Slatkin DJ, Knapp JE, Schiff PL (1981) Constituents of West African Medicinal Plants. XXVIII. Additional Alkaloids of Triclisia gilletti. J Nat Prod 44:61-66.
- Uche FI, Drijfhout FP, McCullagh J, Richardson A, Li WW (2016) Cytotoxicity effects and apoptosis induction by bisbenzylisoquinoline alkaloids from Triclisia subcordata. Phytother Res 30:1533-1539.
- Ayim JS, Dwuma-Badu D, Fiagbe NY, Ateya AM, Slatkin DJ, et al. (1977) Constituents of West African medicinal plants. XXI. Tiliafunimine, a new imino bisbenzylisoquinoline alkaloid from Tiliacora funifera. Lloydia 40:561-565.
- Torel J, Cillard J, Cillard P (1986) Antioxidant activity of flavonoids and reactivity with peroxy radical. Phytochem 25:383-385.
- Nwankwo JO (2002) Anti-metastatic activities of all-trans retinoic acid, indole-3-carbinol and (+)-catechin in Dunning rat invasive prostate adenocarcinoma cells. Anticancer Res 22:4129-4135.
- Ozo ON, Caygill JC, Coursey DG (1984) Phenolics of five yam (dioscorea) species. Phytochem 23:329-331.
- Atawodi SE, Pfundstein B, Haubner R, Spiegelhalder B, Bartsch H, et al. (2007)Content of polyphenolic compounds in the Nigerian stimulants Cola nitida ssp. alba, Cola nitida ssp. rubra A. Chev, and Cola acuminata Schott & Endl and their antioxidant capacity. J Agric Food Chem 55:9824-9828.
Citation: Nwankwo JO (2017) Anticancer Potentials of Phytochemicals from Some Indigenous Food and Medicinal Plants of West Africa. Adv Cancer Prev 2: 124. DOI: 10.4172/2472-0429.1000124
Copyright: ©2017 Nwankwo JO. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Share This Article
Recommended Conferences
42nd Global Conference on Nursing Care & Patient Safety
Toronto, CanadaRecommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 7343
- [From(publication date): 0-2018 - Apr 03, 2025]
- Breakdown by view type
- HTML page views: 6282
- PDF downloads: 1061