Research Article Open Access
Activation of Complement System during Ship Voyage and Winter-over Expedition in Antarctica
Anand Prakash Yadav1, Kamla Prasad Mishra1, Himanshi Tanwar1, Narendra Kumar Sharma2, Sudipta Chanda1, Shashi Bala Singh3 and Lilly Ganju1*
1Immunomodulation Laboratory, Defence Institute of Physiology & Allied Sciences, Lucknow Road, Timarpur, Delhi-110054, India
2Peptides and proteomics division, Defence Institute of Physiology & Allied Sciences, Lucknow Road, Timarpur, Delhi 110054, India
3Defence Institute of Physiology & Allied Sciences, Lucknow Road, Timarpur, Delhi 110054, India
- *Corresponding Author:
- Lilly Ganju
Sc. ‘F’, Immunomodulation Laboratory
Defence Institute of Physiology & Allied Sciences
Lucknow Road, Timarpur
Delhi-110054, India
Tel: +911123883163
Fax: +911123932869
E-mail: lilly.ganju65@gmail.com
Received date: September 04, 2015; Accepted date: November 02, 2015; Published date: November 06, 2015
Citation: Yadav AP, Mishra KP, Tanwar H, Sharma NK, Chanda S, et al. (2015) Activation of Complement System during Ship Voyage and Winter-over Expedition in Antarctica. J Marine Sci Res Dev 5:171. doi:10.4172/2155-9910.1000171
Copyright: © 2015 Yadav AP, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Marine Science: Research & Development
Abstract
The Antarctic milieu comes with extreme cold, intense UV radiation, white-outs, varying magnetic fields, sleep deprivation, altered circadian biorhythms, memory loss, isolation,-causing significant physiological and psychological stresses. These conditions closely resemble space travel, with striking similarity in character of peculiar climate, remoteness, constrained living spaces, disturbed sleep cycles, and environmental stress. Due to its extreme weather conditions, and unique geo-location around the South Pole, it is one of the last frontiers for the human race. Thus the geographic isolation and prolonged stressful stay at Antarctica makes it interesting to study the body responses of expeditioners under extreme environmental conditions. Two teams participated in the study, in 28th Indian Scientific Expedition to Antarctica. The first team included members for short “summer” expedition, while the second team as “Winter team”, included members exposed for long term to Antarctic environment. The present study was performed to study the changes occurring in proteomic profile related to immune system. Blood samples were collected in chronological sequence, including i) baseline control ii) during ship journey and iii) at Maitri (Antarctica), during the months of March, May, August and November. The sera samples collected were subjected to differential expression of protein analysis by 2D Gel electrophoresis followed by MALDI-TOF analysis. Catecholamine was also estimated as a measure of stress. Results of 2D gel and MALDI-TOF-TOF analysis revealed significantly increased level of Complement Factor 3 during ship journey and during peak winter in August. Further detailed analysis of complement pathway by ELISA revealed activation of complement system during the acute and chronic stress of Antarctica.
Keywords
Antarctica; Proteomics; Complement system
Introduction
It is only since last 50 years, that humans have expressed their interest in Antarctica for permanent inheritance [1]. Earlier Antarctica was visited only by expeditioners and logistic personnel in groups, and these groups constituted the total population of about 2500 per year in summers, which would go down to 800 in winters. Whereas with the advancement in the communication technology and facilities the place has become an attraction for adventurous tourists who make the number to go up to 26,000 in summers, but during winters the number remains approximately the same [2]. Many aspects of Antarctic life have become the topic of medical research over recent years and several articles have been published on the lifestyle of Antarctic expeditioners [3]. However, interest has also grown in what is hiding in nature at the bottom of the Earth. Humans have learnt a great deal about the provision of health care in this remote environment and at the same time have also used Antarctica as a laboratory, to expand the knowledge of physiology, psychology and other body systems of mankind [4].
The Antarctic stations offer a unique research environment, in the field of biomedical research, providing the real opportunity for evaluation of human physiology and body responses to adverse conditions in isolated environments [5-8]. These stations built within Antarctica provide simulated environment to prepare space crews for their missions [9], to study atmospheric, geological and biomedical research sciences as an analogue of space on earth [10]. These stations also simulate the typical environment in submarines, and underwater habitats and provide a real-life and real-time observations of human behaviour and experience similar to these conditions.
The visiting population is always fit; usually young, consumes the same diet, experiences the same environment, isolated from the rest of the world for long periods, and is subjected to great seasonal differences in temperature and sunlight in terms of light and dark cycles [11,12]. However, a healthy, transient population participating in wintering over in Antarctica, generally comes from the densely populated regions, and suddenly gets confined to this extreme environment [11]. These expeditioners are totally physically isolated for periods of 8 to 13 months [13,14]. Such population remains away from urban life and thus not exposed to pollution and other health affecting agents such as infectious organisms and parasites [15-17].
Past 30 years of research in the field of psychoneuroimmunology, have convincingly established that stressful experiences alter features of the immune response as well as confer vulnerability to adverse medical outcomes that are either mediated by or resisted by the immune system. However, studies have not yet explored the link between different body systems. It is interesting to determine the changes in various immune system pathways leading to susceptibility to disease onset under stressful conditions.
Complement proteins have many beneficial effects in terms of immunologic defence. However inappropriate activation of the cascade can lead to tissue damage [18,19], which eventually culminates into autoimmune conditions in which the cascade is activated by “self”; this can be observed in the inflamed synovium of patients with rheumatoid arthritis [20] and systemic lupus erythematosus [19]. There is preliminary evidence that psychological stress may also perturb the complement cascade, even in healthy individuals [21]. Naturalistic stressors have been associated with increases in the levels of C3 and C4, which are the two key components of the complement cascade. In students who perceived the examination as highly stressful both C3 and C4 were elevated immediately before an academic examination, which were normal 6 weeks before the examination [22]. Complement levels have also been investigated in men who were subjected to repeated simulations of emergency evacuation procedures for sea-based oil platforms; this was considered a short-term naturalistic stressor [23].
The stress and complement proteins constitute an important part of the immune system. Hence in this study the differential protein expression and detailed study of complement activation pathways during the short summer stay and long winter stay, was evaluated to understand the effect of Antarctic condition on immune system of the Indian expeditioners.
Materials and Methods
The study was approved by Institutional Human Ethics Committee (IHEC) of DIPAS (DRDO, MOD, India); and a written consent was obtained from each individual included in the study. Necessary permissions from the National Centre for Ocean and Antarctic Research (NCAOR) and the Ministry of Earth Sciences, Government of India, was obtained to perform the study. The project was sanctioned by DRDO (Ministry of Defence, India).
Subjects
The 28th Indian Scientific Expedition to Antarctica consisted of two groups with thirty summer (27 males and 3 females; median age: 37) and 22 winter members (all males; median age: 36 years), having both civilian scientists and service personnel.
The team had undergone pre-departure clinical, psychological, and laboratory examinations at All India Institute of Medical Sciences, New Delhi, India, to ensure a healthy population for the expedition.
During the ship journey, after crossing 40°S latitude, 24 out of the 30 volunteers suffered from seasickness, when the ship underwent a significant ‘Rolling and Pitching’ in the turbulent ocean. Thereafter, during the stay at Maitri, most of the members suffered with memory loss and sleep deprivation. However, during the entire duration of the expedition, members were provided with food native to them. None of the subjects used any drugs that could significantly affect the physiological or biochemical parameters, or had symptoms indicative of any infection.
Blood collection
For both short and long term exposure study, blood samples were collected at different stages of the expedition-viz. the pre-exposure blood was collected at Delhi two months before the departure of the expedition in October, as base line control for both the teams. For second time blood was collected on board to Antarctica in January, immediately after the start of the journey, which continued for 30 days. Third blood collection was performed after being off board at Maitri station after 30days i.e., in the month of March. For long term exposure study, additional blood samples were collected subsequently in four different stages of the after one, three, six and nine months viz. in the months of March, May, August and November.
Blood was drawn (10 ml) after overnight fasting and in the morning between 6 and 7 AM, and collected in unheparinised vials; incubated for 1 hr at 37°C. The clear serum was collected in small aliquots and stored at -70°C, for further analysis.
Two dimensional gel electrophoresis
Sample preparation for 2D gel electrophoresis: The sera samples were depleted of the high abundant proteins using Proteoprep- Blue albumin & IgG depletion kit (Sigma Aldrich, St. Louis, MO), according to the manufacturers protocol. The depleted sera samples were precipitated using 10% TCA in acetone. The precipitated protein pellet was dissolved in lysis buffer (8M urea, 2M thio-urea, 40mM Tris-HCl, 4% (w/v) CHAPS, and 0.5% Igepal CA-630) and the protein concentration was determined using bicinchoninic acid assay (BCA assay) (Sigma, USA). The depletion pattern was quantitatively and qualitatively determined on 10% SDS PAGE.
Iso-electric Focussing (IEF) using the protean IEF cell: Immobilized linear pH gradient strips (17 cm, pH 3-10, Bio-Rad), were rehydrated with the individual sera samples, {500 μg of protein, in 325 μl of rehydration buffer solution containing 7 M urea, 2 M thiourea, 1.2%, w/v CHAPS, 0.4 % w/v ABS-14, 20 mM dithiothreitol (DTT) , 0.25%, v/v, pH 3-10 ampholytes and 0.005% w/v bromophenol blue (BPB)}, for 18 h without current (passive rehydration). IEF was conducted using a Protean IEF Cell (Bio-Rad), according to following IEF conditions: Procedure: 250 V for 1 h (slow ramping), changing the wicks every 30 min (to assist removal of ionic contaminants), 1000V for 1h, linear ramping 10000 V to over 3 h and a constant of 10000V until approximately 60 kVh was reached. Strips were removed and stored at-80°C until run on the second dimension.
Second-dimensional SDS-PAGE: Prior to SDS-PAGE, the IPG strips were equilibrated with equilibration solutions twice for 15 min with gentle shaking. The first equilibration solution contained 50 mM of Tris-HCl, pH 8.8, 6M urea, 30% v/v glycerol, 2% w/v SDS, 1% w/v DTT and 0.01% w/v BPB. In the second equilibration solution, DTT was replaced with 2.5% (w/v) iodoacetamide. The equilibrated IPG strips were slightly rinsed with milli-Q water, blotted to remove excess equilibration buffer and then applied to SDS-PAGE gels (20 cm × 20 cm × 1 mm 8-19% polyacrylamide (30% (w/v) acrylamide, 0.8% (w/v) bis-acrylamide, using a PROTEAN II XL system (Bio Rad), at 10 mA per gel for 30 min followed by 35 mA per gel for 12 h.
Protein visualization: Proteins were visualized using modified silver stain procedure compatible with Mass Spectrometry. Following sliver staining method, the gels were fixed in 50% v/v methanol, 12% v/v acetic acid and 0.05% v/v formaldehyde for at least 2 h. The fixed gels were rinsed with 50% v/v ethanol three times for 20 min each, then again sensitized with 0.02% w/v sodium thiosulfate followed by three washings with milli-Q water each for 20 secs. The gels were immersed in 0.1% w/v silver nitrate and 0.075% v/v formaldehyde for 20 min and rinsed with milli-Q water twice for 20 secs each and developed with 6% sodium carbonate and 0.05% v/v formaldehyde. Finally, the reaction was terminated by fixing with 50% v/v methanol and 12% v/v acetic acid. The stained gels were imaged using an Investigator TM ProPic II Genomics Solutions.
In-gel digestion: The silver stained differentially expressed gel spots were excised manually and were subjected to destain with 30mM potassium ferricyanide and 100 mM sodium thiosulfate (1:1). The destained gel spots were washed thrice with the milli Q water and subsequently with 10mM ammonium bicarbonate and acetonitrile (1:1). The gel spots were rehydrated with10 mM ammonium bicarbonate, dehydrated again with acetonitrile, and dried down by speed Vac (Heto MAXI dry plus, UK). Freshly prepared trypsin (20 ng/ml) (sequencing grade, Promega, Madison, WI), was added to the dried gel spots and incubated for 30 min at 4°C and overnight at 37°C. The tryptic digested peptides were extracted by sonicating with 50% acetonitrile and 0.1% TFA and were dried in Speed Vac.
Identification of differentially expressed proteins: The extracted peptides were resuspended in 5 μL of 50% v/v acetonitrile: 0.1% v/v TFA. The resolubilized peptides were mixed with saturated solution of cyano-4-hydroxy-cinnamic acid matrix (1:1) (CHCA, Bruker Daltonics), and were spotted onto the MALDI target plate and allowed to air dry. The spectra were acquired by MALDI-TOF/TOF (Ultraflex III, Bruker Daltonics, Bremen, Germany) in the positive ion mode at the accelerating voltage of 25 kV under the Flex Control software (Version 3.0, Bruker Daltonics). Before the analysis, the instrument was calibrated with mass standards: bradykinin (m/z 757.39), angiotensin II (m/z 1046.54), angiotensin I (m/z 1296.68), substance P (m/z 1347.73), bombysin (m/z 1619.82), ACTH fragment 1-17 (m/z 2093.08), ACTH fragment 18-39 (m/z 2465.39), and somatostatin (m/z 3147.47). For peptide mass fingerprinting (PMF), the generated peptide mass list was exported to Biotools (Version 3.1, Bruker Daltonics) using Flex Analysis 3.0 and submitted to in-house licensed version of the MASCOT database search engine (Matrix Science, Boston, MA). The SWISS-PROT database was used with the following search parameters; Human taxonomy with-100 ppm peptide mass tolerance, 1 maximum missed cleavage sites permitted, and carbamidomethyl (cysteine) as fixed modification. Proteins that returned MOWSE scores over a threshold 56 were considered significant. For each identified protein, at least one peptide was selected for confirmation by MS/MS analysis. The MS/MS spectra were acquired in LIFT mode using the same spot on the target and fragment mass tolerance of -0.8 Da. All the peptide mass values are monoisotopic.
Enzyme linked immunosorbent assay for components of complement system
C3, C4 (Assaypro, Inc.), C3a, C4a, C5a (QND biosystems, Inc.), and Complement Factor B (Qayee bio, China) were estimated by ELISA. Breifly, the Microtiter plate provided was pre-coated with an antibody specific to human C3, C4, C3a, C4a, C5a and Complement Factor B components. Standards and samples were added to the plate followed by a biotin conjugated antibody preparation specific for C3, C4, C3a, C4a, C5a and Complement Factor B components respectively. A solution of avidin conjugated to HRP was added. Colour was developed using TMB as substrate. The substrate initiated a peroxidase-catalyzed color change, which was stopped within 15 min by acidification with stop solution. Absorbance was measured at 450 nm.
Estimation of catecholamine
Catecholamine was estimated by a kit following manufacturer instructions (Cusabio biotech CO., LTD, China). The Microtiter plate provided in the kit was pre-coated with an antibody specific to Catecholamine. Standards and samples were added to the plate followed by a biotin conjugated antibody preparation specific for Catecholamine. Then a solution of Avidin conjugated to HRP was added. The Penultimate step included addition of TMB substrate. The final step included termination of reaction by addition of 1N sulphuric acid solution. Absorbance was measured by ELISA reader (Biotek, USA) at 450 nm.
Statistical analysis
For MALDI TOF analysis: MASCOT probability-based MOWSE (molecular weight search) score calculated for PMF. Protein score is-10*Log (p), where p is the probability that the observed match is a random event and greater than 56 are significant (p<0.05) was considered significant.
For ELISA results, values are represented as mean ± standard error (SE). Comparison between results was performed using the Repeated Measure ANOVA with time as the factor. Results of all the parameters were compared at different time points with each other. The alpha value of ≤ 0.05 was considered significant. Entire analysis was conducted using SPSS 16 software.
Result
Summer study
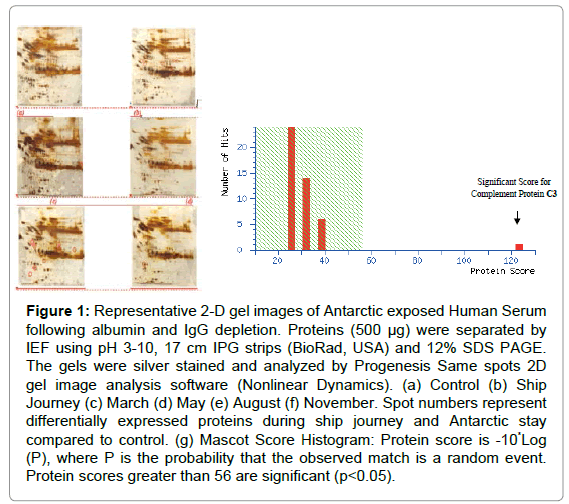
2-D Gel Electrophoresis and MALDI TOF analysis: The sera proteomes of Antarctic exposed summer and winter members were resolved by 2-DGE and compared with baseline control to identify differentially expressed proteins. Comparative gel analysis resulted in the identification of differentially expressed spots (Figure 1a-1f), which were further excised from gels and identified using MALDI-TOF/TOF and MS/MS analysis (Figure 1g). In the MALDI-TOF analysis the Mascot Score is considered significant if the protein score is greater than 56. The Concise Protein Summary Report is as follows: CO3_HUMAN Mass: 188569 Score: 123 Expect: 1e-08 Matches: 25 Complement C3 OS=Homo sapiens GN=C3 PE=1 SV=2.
Figure 1: Representative 2-D gel images of Antarctic exposed Human Serum following albumin and IgG depletion. Proteins (500 μg) were separated by IEF using pH 3-10, 17 cm IPG strips (BioRad, USA) and 12% SDS PAGE. The gels were silver stained and analyzed by Progenesis Same spots 2D gel image analysis software (Nonlinear Dynamics). (a) Control (b) Ship Journey (c) March (d) May (e) August (f) November. Spot numbers represent differentially expressed proteins during ship journey and Antarctic stay compared to control. (g) Mascot Score Histogram: Protein score is -10*Log (P), where P is the probability that the observed match is a random event. Protein scores greater than 56 are significant (p<0.05).
Estimation of various component of complement system: Based on the findings of MALDI-TOF analysis, the complement activation was further comprehensively elucidated for the understanding of the mechanism by ELISA.
Summer study estimations
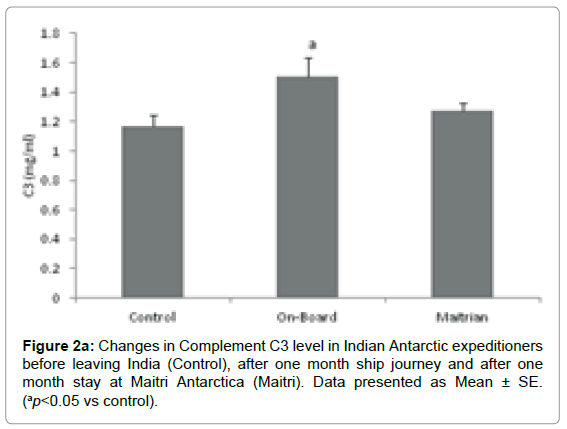
Complement C3: Complement C3 levels were determined in the stressful environmental conditions of southern ocean and Antarctica. The mean level of Complement C3 was 1.165 ± 0.0756 mg/ml before proceeding to Antarctica which is considered as control. The levels increased to 1.504 ± 0.129 mg/ml (ap=0.036; ap<0.05 vs control), after being one month on-board in southern ocean. The Complement C3 levels reduced to 1.271 ± 0.0534 mg/ml after being one month off-board at Maitri as compared to control. On the whole, stressful environmental conditions lead to increased levels of Complement C3 Figure 2a.
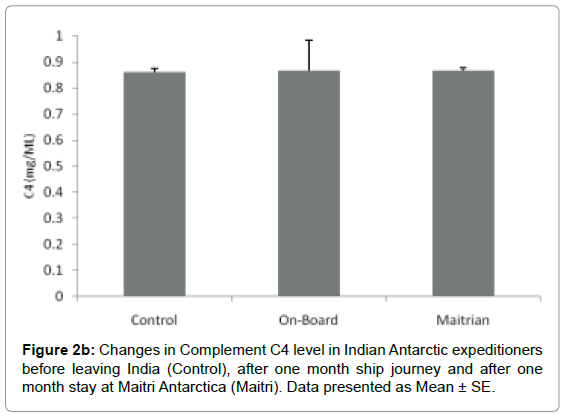
Complement C4: The level of Complement C4 was 0.862 ± 0.0115 mg/ml before proceeding to Antarctica. The levels increased to 0.868 ± 0.116 after being one month on-board which was not significant. The C4 levels remained 0.868 ± .099 mg/ml after being one month offboard at Maitri as compared to control. Levels of C4 were not altered significantly during ship journey and at Maitri as compared to the levels of control Figure 2b.
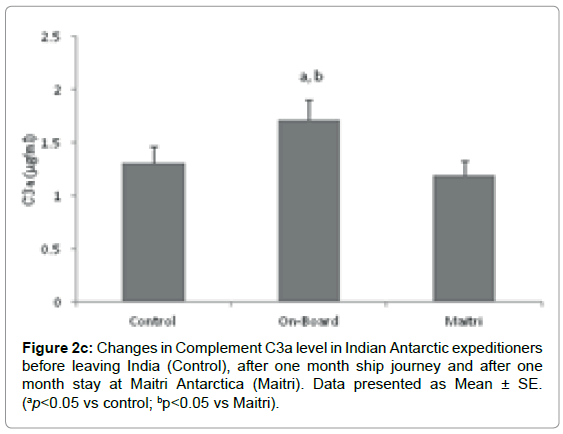
Complement C3a component: C3a levels were determined in the stressful environmental conditions of southern ocean and Antarctica. The mean level of C3a was 1.310 ± 0.156 μg/ml before proceeding to Antarctica. The levels increased to1.716 ± 0.19μg/ml (ap=0.045; ap<0.05 vs control; bp=0.033, bp<0.05 vs Maitri) after being one month onboard in southern ocean. The C3a levels reduced to 1.196 ± 0.134 μg/ ml after being one month off-board at Maitri as compared to control Figure 2c.
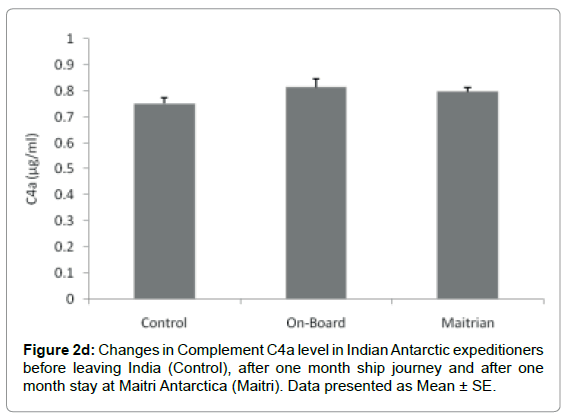
Complement C4a component: The level of C4a was 0.75 ± 0.023 μg/ml before proceeding to Antarctica. The levels increased to 0.814 ± 0.031 μg/ml after being one month on-board which was not significant. The C4a level decreased to 0.796 ± 0.018 μg/ml after being one month off-board at Maitri Figure 2d.
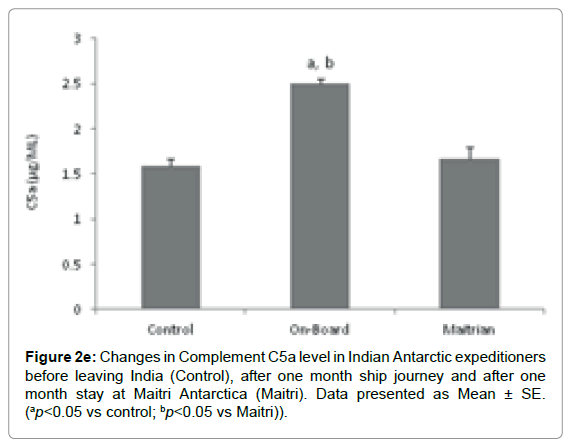
Complement C5a component: The level of C5a was 1.58 ± 0.077 μg/ml before proceeding to Antarctica. The levels increased significantly to 2.508 ± 0.049 μg/ml (ap=0.004; ap<0.05 vs control) after being one month on-board as compared to control. Further, the C5a level decreased to 1.66 ± 0.129 μg/ml (bp=0.011; bp<0.05 vs Maitri) after being one month off-board at Maitri. (Figure 2e).
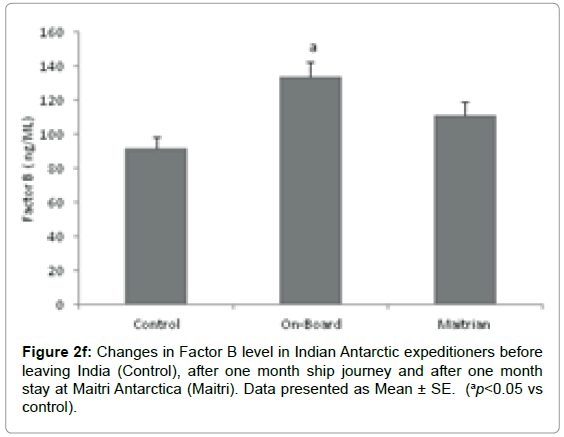
Complement Factor B: The mean level of Factor B was 91.5 ± 7.1 ng/ ml before proceeding to Antarctica which is considered as control. The level significantly increased to 133.7 ± 9.13 ng/ml (ap=0.004; ap<0.05 vs control), after being one month on-board in southern ocean. The level came down to 111.4 ± 7.8 ng/ml after being one month off-board at Maitri as compared to control. On the whole, stressful environmental conditions lead to increased levels of Factor B. (Figure 2f).
Winter study estimations
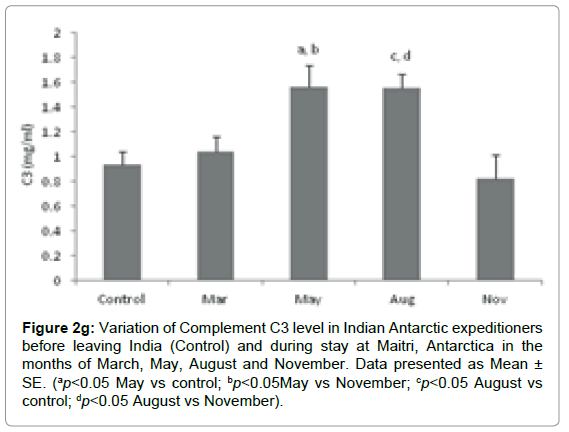
Complement C3: The Complement C3 level in base line-control was 0.935 ± 0.104 mg/ml which increased to1.045 ± 0.115 mg/ml in the month of March. It was further increased to 1.566 ± 0.173 ng/ml (ap=0.035; ap < 0.05 vs control; bp=0.028; bp<0.05 vs November) and 1.554 ± 0.1099 mg/ml (cp=0.041; cp < 0.05 vs control; dp=0.033; dp<0.05 vs November) in the months of May and August which reduced to 0.828 ± 0.189 mg/ml in the month of November Figure 2g.
Figure 2g: Variation of Complement C3 level in Indian Antarctic expeditioners before leaving India (Control) and during stay at Maitri, Antarctica in the months of March, May, August and November. Data presented as Mean ± SE. (ap<0.05 May vs control; bp<0.05May vs November; cp<0.05 August vs control; dp<0.05 August vs November).
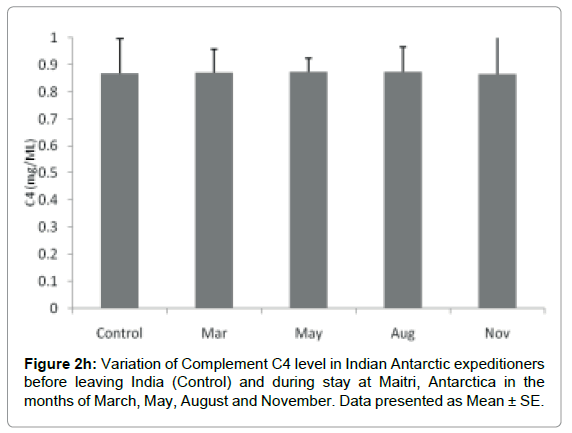
Complement C4: The level of Complement C4 was not significantly altered during the isolation at Antarctica. The Complement C4 level in base line-control was 0.868 ± 0.0127 mg/ml which increased to 0.872 ± .0086 mg/ml in the month of March. It was decreased to 0.873 ± 0.005 mg/ml and 0.875 ± 0.009 mg/ml in the months of May and August respectively in comparison to control. Further the level decreased to 0.865 ± 0.015 mg/ml in the month of November. The levels were significantly not altered, Figure 2h.
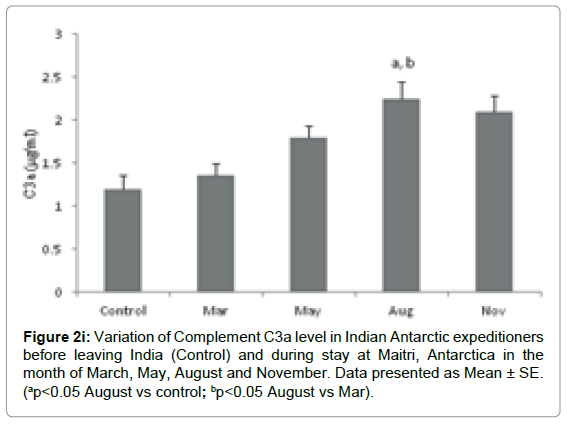
Complement C3a component: The level of C3a was significantly altered during the isolation at Antarctica. C3a level in base line-control was 1.198 ± 0.16 μg/ml which increased to1.354 ± 0.146 μg/ml in the month of March. It was further significantly increased to 1.798 ± 0.139 μg/ml and 2.249 ± 0.194 μg/ml (ap=0.003, ap<0.05 vs control & bp=0.014, bp<0.05 vs March) in the months of May and August respectively in comparison to control. The level insignificantly decreased to 2.093 ± 0.194μg/ml in the month of November, Figure 2i.
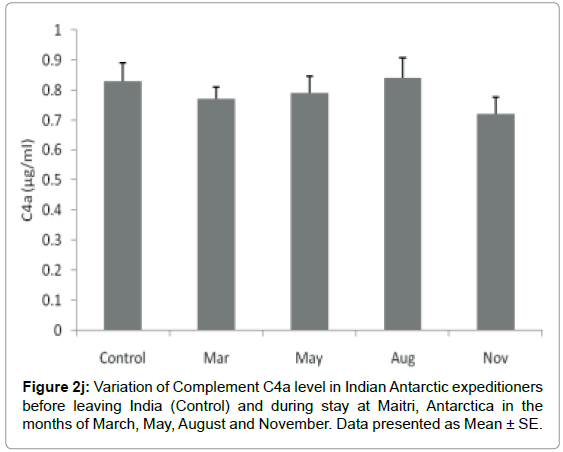
Complement C4a component: The level of C4a was not altered significantly during the isolation at Antarctica. The C4a level in base line-control was 0.83 ± 0.06 μg/ml which was 0.77 ± 0.04 μg/ml in the month of March. The levels were 0.79 ± 0.056 μg/ml and 0.84 ± 0.066 μg/ml in the months of May and August respectively. Further the level insignificantly decreased to 0.72 ± 0.055 μg/ml in the month of November Figure 2j.
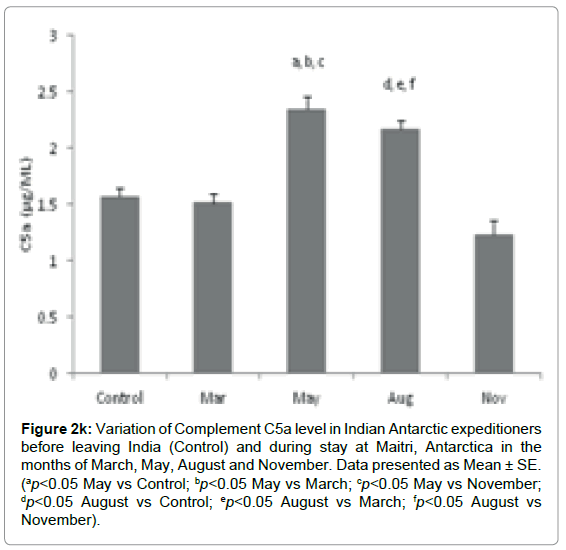
Complement C5a component: The level of C5a was significantly altered during the isolation at Antarctica. The Complement C5a level in base line-control was 1.56 ± 0.078 μg/ml which came down to 1.50 ± 0.090 μg/ml in the month of March. It was increased to 2.34 ± 0.11 μg/ ml (ap=0.022, ap<0.05 vs Control; bp=0.018, bp<0.05 vs March; cp=0.008, cp<0.05 vs November) and 2.165 ± 0.086 μg/ml (dp=0.032, dp<0.05 vs Control; ep= 0.031, ep<0.05 vs March; fp=0.023, fp<0.05 vs November) in the months of May and August respectively, in comparison to control. The level decreased to 1.23 ± 0.122 μg/ml in the month of November Figure 2k.
Figure 2k: Variation of Complement C5a level in Indian Antarctic expeditioners before leaving India (Control) and during stay at Maitri, Antarctica in the months of March, May, August and November. Data presented as Mean ± SE. (ap<0.05 May vs Control; bp<0.05 May vs March; cp<0.05 May vs November; dp<0.05 August vs Control; ep<0.05 August vs March; fp<0.05 August vs November).
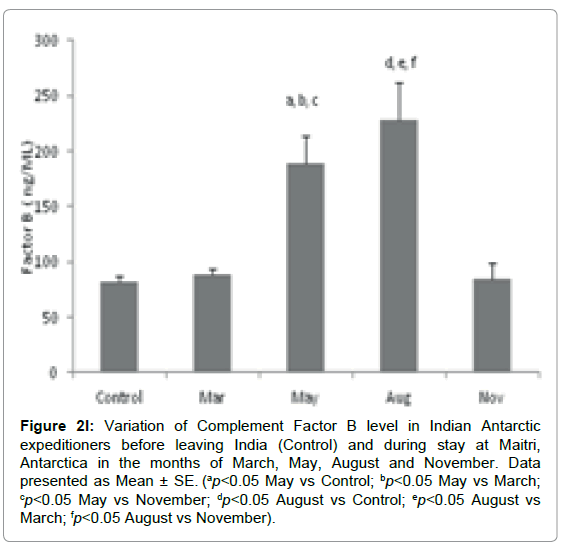
Complement factor B: The level of Factor B was significantly altered during the isolation at Antarctica. The Complement Factor B level in base line-control was 82 ± 4.9 ng/ml which increased to 88 ± 5.2 ng/ml in the month of March. It was further increased significantly to189 ± 24.8 ng/ml (ap=0.012, ap<0.05 vs Control; bp=0.017, bp<0.05 vs March; cp=0.022, cp<0.05 vs November) and 227.9 ± 33.8 ng/ml (dp=0.003, dp<0.05 vs Control; ep=0.004, ep<0.05 vs March & fp=0.009, fp<0.05 vs November) in the months of May and August respectively in comparison to control and March. Further the level insignificantly decreased to 84.3 ± 14.4 ng/ml in the month of November Figure 2l.
Figure 2l: Variation of Complement Factor B level in Indian Antarctic expeditioners before leaving India (Control) and during stay at Maitri, Antarctica in the months of March, May, August and November. Data presented as Mean ± SE. (ap<0.05 May vs Control; bp<0.05 May vs March; cp<0.05 May vs November; dp<0.05 August vs Control; ep<0.05 August vs March; fp<0.05 August vs November).
Estimation of catecholamines in summer study
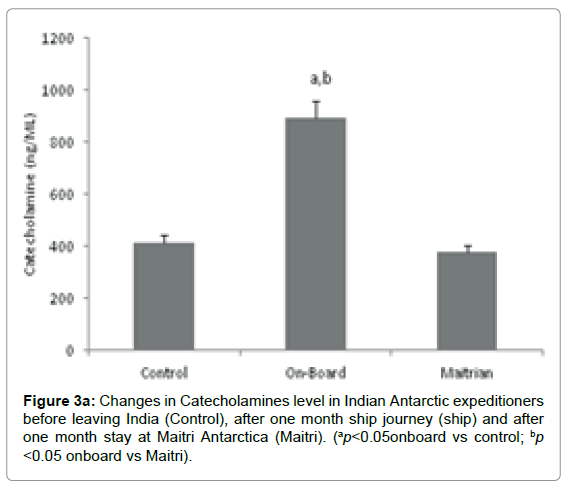
The level of Catecholamine was 415.4 ± 28.7 mg/dl before proceeding to Antarctica. The levels significantly increased to 893.3 ± 64 mg/dL (ap<0.001, ap <0.05 vs control; bp<0.001, bp <0.05 vs Maitri) after being one month on-board. However, the Catecholamines levels decreased to 377.4 ± 27 mg/dL after being one month off-board at Maitri as compared to control Figure 3a.
Estimation of catecholamines in winter study
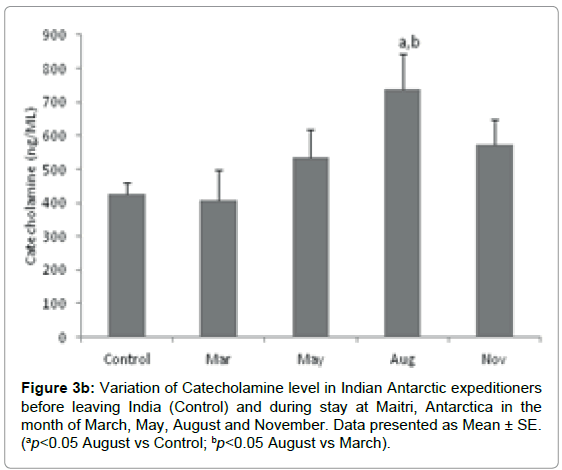
Catecholamine was measured in the sera samples of Antarctic wintering personnel at different time points as shown in Figure 3b.The level of Catecholamine was significantly altered during the isolation at Antarctica, as the level in base line-control was 427.69 ± 32.2 ng/ ml which decreased to 408.55 ± 90.06 ng/ml, in the month of March. It was further increased to 536.37 ± 80.35 ng/ml and 738.547 ± 103.3 ng/ml (ap=0.009, ap<0.05 vs Control; bp=0.006, bp <0.05 vs March) in the months of May and August respectively. However the level was decreased to 573.322 ± 74.5 ng/ml in the month of November. The data indicates that the level of catecholamines was increased significantly in the months of May, August and November as compared to the base line level.
Discussion
In today’s world humans rarely face a stimulus which evokes fightor- flight responses, which were very common to our ancestors, such as predation or inclement weather without protection. However, human physiological response continues to reflect the demands of earlier environments. Threats that do not require a physical response (e.g., academic exams) may therefore have physical consequences, including changes in the immune system. Indeed, over the past 30 years, more than 300 studies have been performed on stress and immunity in humans, and together they have shown that psychological challenges are capable of modifying various features of the immune response.
Our previous metabonomics based study showed that during ship voyage and long term exposure to extreme environmental conditions prevailing in Antarctica triggered significant alterations in the concentrations of various metabolites and changes in different metabolic pathways in the expeditioners [24]. These findings further motivated us to study expression of different stress proteins and proteins related to immune system in particular the complement system ship journey and long Antarctic stay. This study finds its uniqueness as one of the first study which focuses on complement system which play significant role in immunomodulation under stressful environment of Antarctica.
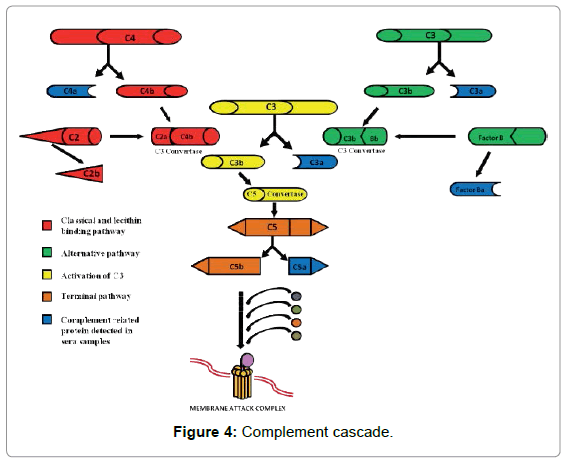
Analysis of differential expression of specific protein was performed using, 2D gel electrophoresis of sera samples. Several differentially expressed spots were identified and subjected to MALDI-TOF analysis which revealed predominant expression of human Complement Protein C3. This alteration in expression of C3 protein indicated a breakthrough in activation of complement proteins during the expedition. More comprehensive and in depth assessment of activation of complements indicated expression of C3a, C4a, C5a and Factor B in sera samples of expeditioners (Figure 4). C3 was significantly increased during the ship journey in summer team. Naturalistic stressors have been associated with increases in the levels of C3 and C4 [25-27], which are two key components of the complement cascade. Activation of complement in response to acute stress in men who were subjected to repeated simulations of emergency evacuation procedures for seabased oil platforms [28]. Maes et al [22] have reported the activation of complements in response to different psychological stress. The increase in the levels of C3 and C3a indicates the activation of complement cascade in response to ship borne journey in the southern Ocean. Significant increase in the levels of C5a indicates that the activation of complement has proceeded down the terminal pathway. At the same time significant increase in the levels of factor B indicates that the activation of the complement system was through alternative pathway. However, after the summer team reached Maitri the levels of C3, C3a, C5a and Factor B decreased significantly. Thus, these results indicate that ship borne journey in southern ocean activated complement system through alternative pathway. In long term winter study, levels of C3 were found to be increased during the months of May and August indicating activation of complement during this period also. Several studies have shown that psychological and physiological stresses can activate complement cascade. [22,23,28,29]. However, C5a was found to be increased only in the month of August indicating that terminal pathways were activated during the month of August. It has been reported that the third quarter of expedition, is the most stressed phase of expedition and activation of complement could be correlated to the multiple stresses encountered by the expeditioners during this period. Stress marker like catecholamine has also shown that this was the most stressed phase of the expedition. It also indicates that acclatimization is not achieved in human. This also suggests that prolonged exposure to Antarctic stress is associated with stimulation of sympathetic and Hypothalamus Pituitary Axis (HPA) and maintenance of higher sympathetic and HPA activity might be necessary for acclatimatization of extreme cold climate condition. Thus this indicates that harsh winter conditions could be detrimental factor for activation of complement cascade. However, the effect of activated complement system during the expedition could not be correlated clinically because of the multiple stresses and small population size in Antarctic expedition.
The most pressing question to be answered is that future research needs to address the extent to which this stress induced changes in the immune system and whether it has meaningful implications for disease susceptibility in healthy expeditioners. Further investigations are required to determine the factors that influence the magnitude of the complement response and the clinical implications of the changes that are observed.
Acknowledgements
The authors record their sincere thanks to the 28th Indian Antarctic Expeditionary Team members who participated voluntarily and allowed their blood samples to be taken for this study. The authors are also thankful to the Directors of DIPAS, DRDO, for the support provided for this study. Special thanks to National Centre for Antarctic and Ocean Research (NCAOR), India for providing all the facilities required for the study.
References
- Lugg D, Shepanek M (1999) Space analogue studies in Antarctica. Acta Astronaut 44:693-699.
- Shearer WT, Lee BN, Cron SG, Rosenblatt HM, Smith EO, et al. (2002) Suppression of human anti-inflammatory plasma cytokines IL-10 and IL-1RA with elevation of proinflammatory cytokine IFN-γ during the isolation of the Antarctic winter. J Allergy Clin Immunol 109: 854-857.
- Olson JJ (2002) Antarctica: a review of recent medical research.Trends PharmacolSci 23: 487-491.
- Carrère S, Evans GW, Stokols D (1991) Winter-Over Stress: Physiological and Psychological Adaptation to an Antarctic Isolated and Confined Environment. From Antarctica to Outer Space 229-237.
- Tingate TR (1997) Antarctic isolation: immune and viral studies. Immunol Cell Biol 75: 275-283.
- Dantzer R, Mormede P (1995) Psychoneuroimmunology of stress. In Stress, the Immune System and Psychiatry 5: 48-83.
- Palinkas LA (2000) Summary of research issues in behaviour and performance in isolated and confined extreme (ICE) environments. Aviat Space Environ Med 71: 48-50.
- Farrace S (1999) Endocrine and psycho-physiological aspects of human adaptation to the extreme. Physiol Behav 66: 613-620.
- Harrison AA (1991) Antarctica: Prototype for outer space. NASA-TM-108095. Scott Polar Research Institute, University of Cambridge 1-13.
- Rivolier J, Bachelard C (1988) EuropeanSpace Agency Report, Paris.
- Roy DD, Deb NC (1999) Role stress profiles of scientists and defense personnel in 15th Antarctic Expedition. 15th IEA scientific report 13: 371-375.
- Palinkas LA (1992) Going to extremes: the cultural context of stress, illness and coping in Antarctica. Soc Sci Med35: 651-664.
- Sauer J (1999) Performance Evaluation in analogue space environments: adaptation duringan 8-month antarctic wintering-over expedition. Aviat Space Environ Med 70: 230-235.
- Shirai T, Magara KK, Motohashi S, Yamashita M, Kimura M, et al. (2003) TH1-biased immunity induced by exposure to Antarctic winter. J Allergy Clin Immunol 111: 1353-1356.
- Harrison AA, Clearwater YA, McKay CP (1991)From Antarctica to Outer Space-Life in Isolation and Confinement, Springer 3: 451-457.
- Williams DL (1986) Cell mediated immunity in healthy adult in Antarctica and the sub Antarctic. J Clin Lab Immunol 20: 43-49.
- Muller HK (1995) Cell mediated immunity in Antarctic wintering personal; 1984-1992.Immunol Cell Biol 73: 316-320.
- Nakagawa K, Sakiyama H, Tsuchida T, Yamaguchi K, Toyoguchi T, et al. (1999) Complement C1s activation in degenerating articular cartilage of rheumatoid arthritis patients: immunohistochemical studies with an active form specific antibody. Ann Rheum Dis 58: 175-181.
- Walport MJ (2002) Complement and systemic lupus erythematosus. Arthritis Res 4: 279-293.
- Neumann E, Barnum SR, Tarner IH, Echols J, Fleck M, et al. (2002) Local production of complement proteins in rheumatoid arthritis synovium. Arthritis Rheum46: 934-945.
- Clancey WJ (2001) A framework for analog studies of Mars surface operations: Using the Flashline Mars Arctic Research Station.USA, Nasa/Ames Research Center 1-18.
- Maes M, Hendriks D, Gastel AV, Demedts P, Wauters A, et al. (1997) Effects of psychological stress on serum immunoglobulin, complement, and acute phase protein concentrations in normal volunteers. Psychoneuroendocrinology 22: 397-409.
- Endresen IM, Relling GB, Tonder O, Myking O, Walther BT, Ursin H (1991) Brief uncontrollable stress and psychological parameters influence human plasma concentrations of IgM and complement component C3. Behav Med 14: 167-176.
- Yadav AP, Chaturvedi S, Mishra KP, Pal S, Ganju L, et al. (2014) Evidence for altered metabolic pathways during environmental stress: 1H-NMR spectroscopy based metabolomics and clinical studies on subjects of sea-voyage and Antarctic-stay. Physiol Behav 135: 81-90.
- Pawlak CR, Witte T, Heiken H, Hundt M, Schubert J, et al. (2003) Flares in patients with systemic lupus erythematosus are associated with daily psychological stress. Psychother Psychosom 72: 159-165.
- Harrington L, Affleck G, Urrows S, Tennen H, Higgins P, et al. (1993) Temporal covariation of soluble interleukin-2 receptor levels, daily stress, and disease activity in rheumatoid arthritis. Arthritis Rheum 36: 199-203.
- McFarlane AC, Kalucy RS and Brooks PM (1987) Psychological predictors of disease course in rheumatoid arthritis. J Psychosom Res 31: 757-764.
- Lachmann PJ, Hughes-Jones NC (1984) Initiation of complement activation. Springer Semin Immunopathol 7: 143-162.
- Segerstrom SC, Miller GE (2004) Psychological stress and the human immune system: a meta-analytic study of 30 years of inquiry. Psychol Bull 130: 601-630.
Relevant Topics
- Algal Blooms
- Blue Carbon Sequestration
- Brackish Water
- Catfish
- Coral Bleaching
- Coral Reefs
- Deep Sea Fish
- Deep Sea Mining
- Ichthyoplankton
- Mangrove Ecosystem
- Marine Engineering
- Marine Fisheries
- Marine Mammal Research
- Marine Microbiome Analysis
- Marine Pollution
- Marine Reptiles
- Marine Science
- Ocean Currents
- Photoendosymbiosis
- Reef Biology
- Sea Food
- Sea Grass
- Sea Transportation
- Seaweed
Recommended Journals
Article Tools
Article Usage
- Total views: 15660
- [From(publication date):
December-2015 - Apr 03, 2025] - Breakdown by view type
- HTML page views : 14779
- PDF downloads : 881